Revolutionizing Asymmetric Synthesis: Scalable Transfer Hydrogenation For Complex Chiral Intermediates
Revolutionizing Asymmetric Synthesis: Scalable Transfer Hydrogenation For Complex Chiral Intermediates
The landscape of asymmetric synthesis is undergoing a significant transformation driven by the need for safer, more efficient, and cost-effective manufacturing routes for chiral intermediates. Patent CN1257468A introduces a groundbreaking methodology for catalytic transfer hydrogenation that leverages specialized metal cyclopentadienyl complexes coordinated with defined bidentate ligands. This technology represents a pivotal shift away from traditional high-pressure hydrogenation methods, offering a robust alternative for the production of optically active compounds. By utilizing preferred metals such as rhodium, ruthenium, and iridium in conjunction with chiral diamines and aminoalcohols, this process achieves exceptional stereoselectivity and reaction rates. For R&D directors and process chemists, the implications are profound: the ability to generate high-purity chiral alcohols and amines without the logistical and safety burdens of gaseous hydrogen infrastructure. This report analyzes the technical merits of this invention and its potential to redefine supply chain reliability for complex pharmaceutical intermediates.

The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the asymmetric reduction of prochiral ketones and imines has relied heavily on methods that, while effective, suffer from significant operational inefficiencies. Prior art, such as the benzene-ligand ruthenium catalysts described by Noyori and others, often necessitates extended reaction times ranging from 15 to over 90 hours to achieve acceptable conversion. These prolonged cycles are not merely an inconvenience; they represent a substantial bottleneck in commercial manufacturing, limiting reactor throughput and increasing energy consumption. Furthermore, these conventional systems are frequently sensitive to the electronic nature of the substrate's aryl groups, requiring precise tuning for different batches. Perhaps most critically, the slow kinetics can lead to the gradual decomposition of the catalytic complex and a concomitant loss of optical purity in the product due to back-hydrogenation or racemization over time. The reliance on specific, often expensive, phosphorus or nitrogen-containing ligands in earlier iterations also added layers of complexity to catalyst preparation and recovery, complicating the path to cost reduction in pharmaceutical manufacturing.
The Novel Approach
The methodology disclosed in CN1257468A addresses these historical pain points through the strategic implementation of pentamethylcyclopentadienyl (Cp*) ligands coordinated with transition metals. This structural modification fundamentally alters the catalytic profile, enabling reaction times that are drastically shorter, often completing within minutes to a few hours rather than days. The use of Cp* ligands imparts superior stability to the metal center, resisting decomposition even under the thermal stress required to drive rapid kinetics. This innovation allows for the use of simple, inexpensive hydrogen donors such as secondary alcohols (e.g., isopropanol) or formic acid-triethylamine mixtures, effectively bypassing the need for high-pressure hydrogen gas. The result is a process that is not only faster but inherently safer and more adaptable to a wide range of substrates, including sensitive imines and sterically hindered ketones. This approach facilitates the commercial scale-up of complex asymmetric syntheses by simplifying the engineering requirements and enhancing the overall robustness of the chemical transformation.
Mechanistic Insights into Cp*-Metal Catalyzed Transfer Hydrogenation
At the heart of this technology lies a sophisticated bifunctional catalytic mechanism where the metal center and the chiral ligand cooperate to facilitate hydride and proton transfer. The catalyst typically features a metal atom (M), such as Rh(III) or Ir(III), bound to the electron-rich Cp* ring and a chelating bidentate ligand (A-E-B). In the active state, the ligand—often an aminoalcohol or diamine—participates directly in the bond-breaking and bond-forming events. For instance, when using isopropanol as the hydrogen donor, the metal center activates the alcohol to form a metal-hydride species, while the basic nitrogen or oxygen atom on the ligand assists in proton shuttling. This concerted action ensures that the hydride is delivered to the carbonyl or imine carbon with high stereochemical fidelity, dictated by the chiral environment of the ligand backbone. The specific geometry of the Cp* ligand creates a steric pocket that further enforces selectivity, minimizing the formation of unwanted enantiomers and ensuring that the final product meets stringent purity specifications required for API intermediates.
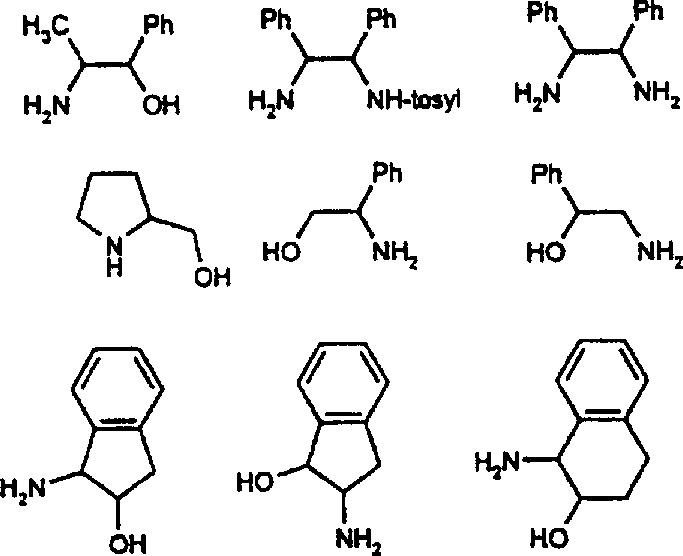
Controlling the impurity profile in such reactions is paramount, and the mechanism described offers inherent advantages in this regard. The rapid turnover frequency of the Cp*-based catalysts means that the substrate spends less time in the reaction mixture, reducing the window for side reactions such as over-reduction or condensation. Additionally, the stability of the catalyst complex prevents the leaching of free metal ions, which can often catalyze non-selective background reactions. The patent data highlights that by carefully selecting the substituents on the ligand backbone (R groups on the A-E-B framework), chemists can fine-tune the electronic and steric properties to suppress specific impurity pathways. For example, the use of N-tosylated diamines versus free aminoalcohols can shift the selectivity profile significantly, allowing for the optimization of enantiomeric excess (ee) for difficult substrates. This level of mechanistic control provides R&D teams with a powerful toolkit for troubleshooting and optimizing processes for new drug candidates.
How to Synthesize Chiral Alcohols Efficiently
The practical implementation of this transfer hydrogenation technology is designed to be straightforward and scalable, making it highly attractive for process development teams aiming to reduce lead time for high-purity chiral alcohols. The general procedure involves the in situ generation of the active catalyst species by mixing a stable metal precursor, such as a dichloride dimer, with the chiral ligand in a solvent like 2-propanol. This mixture is typically heated briefly to ensure complete complexation before the addition of the substrate and a base activator. The reaction proceeds smoothly under mild thermal conditions, often between 35°C and 60°C, and atmospheric pressure. Detailed standard operating procedures for specific substrates, including precise molar ratios and workup protocols, are essential for reproducibility and are outlined in the technical guide below.
- Prepare the active catalyst species by reacting a metal cyclopentadienyl halide dimer (e.g., [Rh(Cp*)Cl2]2) with a chiral bidentate ligand such as an aminoalcohol or diamine in a suitable solvent like 2-propanol under inert atmosphere.
- Activate the catalyst system by adding a base, such as sodium 2-propoxide or triethylamine, to facilitate the formation of the active metal-hydride or metal-amido species required for hydrogen transfer.
- Introduce the prochiral substrate (ketone or imine) to the reaction mixture and maintain moderate temperatures (35°C to 60°C) while removing volatile byproducts like acetone to drive the equilibrium towards the desired chiral product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this transfer hydrogenation technology translates into tangible strategic benefits beyond mere chemical efficiency. The elimination of gaseous hydrogen removes the need for specialized high-pressure reactors and the associated safety certifications, significantly lowering capital expenditure (CAPEX) for new production lines. Furthermore, the use of liquid hydrogen donors like isopropanol simplifies logistics and storage, as these materials are readily available commodity chemicals rather than hazardous gases requiring dedicated infrastructure. This shift enhances supply chain reliability by decoupling production from the constraints of hydrogen availability and delivery schedules. The robustness of the catalyst system also implies longer campaign runs with less frequent catalyst replenishment, contributing to substantial cost savings in raw material consumption and waste disposal.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the drastic reduction in cycle times and the elimination of high-pressure equipment. Traditional hydrogenation often requires expensive autoclaves and rigorous safety monitoring, whereas this transfer method operates at ambient pressure. By shortening the reaction time from days to hours, manufacturers can achieve significantly higher throughput per reactor volume, effectively increasing capacity without building new facilities. Additionally, the high turnover numbers reported in the patent data suggest that catalyst loading can be minimized, reducing the cost contribution of precious metals like rhodium and iridium to the final product price. The simplified workup procedures, often involving simple distillation or extraction, further lower operational expenses by reducing solvent usage and energy consumption during purification.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the versatility and stability of the reagents involved. Isopropanol and formic acid are globally sourced commodities with stable pricing and availability, unlike specialized hydrogenation catalysts that may have long lead times. The ability to run reactions under inert atmospheres (nitrogen or argon) rather than explosive hydrogen gas reduces the risk of unplanned shutdowns due to safety incidents. Moreover, the tolerance of the catalyst system to various functional groups means that a single catalytic platform can be applied to multiple intermediates within a portfolio, streamlining inventory management and reducing the complexity of the supply chain. This flexibility allows manufacturers to respond more agilely to fluctuations in demand for specific API intermediates.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this technology offers a greener alternative to traditional reduction methods. The absence of high-pressure hydrogen mitigates the risk of catastrophic failure, aligning with increasingly stringent process safety management (PSM) regulations. The use of secondary alcohols as hydrogen donors generates ketone byproducts (e.g., acetone from isopropanol) which are often easier to recover and recycle than the waste streams associated with stoichiometric reducing agents like borohydrides. The high selectivity of the reaction minimizes the formation of byproducts, reducing the load on wastewater treatment systems and lowering the overall E-factor of the process. This alignment with green chemistry principles not only ensures compliance but also enhances the corporate sustainability profile, a key factor for modern pharmaceutical partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalytic transfer hydrogenation technology. These insights are derived directly from the experimental data and claims within the patent literature, providing a factual basis for decision-making. Understanding these nuances is critical for evaluating the feasibility of adopting this route for your specific product pipeline. We encourage technical teams to review these points in the context of their current manufacturing challenges.
Q: What are the primary advantages of using Cp*-metal catalysts over traditional benzene-ligand systems for transfer hydrogenation?
A: According to patent CN1257468A, Cp*-metal complexes (using pentamethylcyclopentadienyl ligands) demonstrate significantly faster reaction rates and higher turnover numbers compared to traditional benzene-ligand catalysts. They also exhibit greater stability against decomposition during prolonged reaction times, allowing for more robust processing conditions without the loss of optical purity often seen in slower conventional methods.
Q: Is high-pressure gaseous hydrogen required for this asymmetric reduction process?
A: No, this process specifically utilizes transfer hydrogenation, meaning it operates without gaseous hydrogen. Instead, it employs safe and liquid hydrogen donors such as secondary alcohols (e.g., isopropanol) or formic acid/triethylamine mixtures. This eliminates the need for expensive high-pressure autoclaves and enhances overall plant safety by removing explosion hazards associated with H2 gas.
Q: Which types of substrates are most suitable for this catalytic system?
A: The technology is particularly effective for the reduction of prochiral ketones and imines to their corresponding chiral alcohols and amines. Specific examples in the data include acetophenone derivatives, tetralones, and cyclic imines, which are critical building blocks for active pharmaceutical ingredients (APIs) and agrochemical intermediates requiring high enantiomeric excess.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Intermediate Supplier
The technological advancements detailed in CN1257468A underscore the immense potential of Cp*-metal catalyzed transfer hydrogenation for the production of high-value chiral building blocks. At NINGBO INNO PHARMCHEM, we recognize that translating such innovative chemistry from the laboratory to commercial reality requires more than just intellectual property; it demands deep process engineering expertise and a commitment to quality. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this technology are fully realized in your supply chain. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, including precise enantiomeric excess measurements, to guarantee that every batch meets the exacting standards of the global pharmaceutical industry.
We invite you to explore how this advanced catalytic methodology can optimize your manufacturing costs and timelines. Our technical team is ready to conduct a Customized Cost-Saving Analysis tailored to your specific target molecules, evaluating the feasibility of adapting this transfer hydrogenation route to your existing processes. We encourage you to contact our technical procurement team today to request specific COA data from our pilot runs and comprehensive route feasibility assessments. Let us collaborate to secure a more efficient, safe, and reliable supply of your critical chiral intermediates.
