Advanced Synthesis of Arylbis(triflyl)methane for High Performance Lewis Acid Catalyst Manufacturing
The chemical industry continuously seeks robust methodologies for synthesizing highly functionalized fluorinated intermediates, particularly those serving as precursors for superacidic catalysts. Patent CN1481357A introduces a groundbreaking approach for the efficient production of various arylbis(perfluoroalkylsulfonyl)methanes, compounds that have historically been difficult to synthesize due to steric and electronic constraints. This innovation specifically addresses the challenges associated with introducing bulky aryl groups and electrophilic aryl groups into the bis(trifluoromethanesulfonyl)methane framework. By leveraging sodium trifluoromethanesulfinate as a stable trifluoromethanesulfonyl source, the process circumvents the safety hazards and handling difficulties of gaseous reagents used in prior art. For R&D Directors and Procurement Managers, this represents a significant shift towards safer, more reliable supply chains for high-purity fluorinated intermediates. The ability to produce compounds like pentafluorophenylbis(trifluoromethanesulfonyl)methane in high yield opens new avenues for developing advanced Lewis acid catalysts essential in pharmaceutical and agrochemical manufacturing.
Historically, the synthesis of phenylbis(trifluoromethanesulfonyl)methane and its derivatives relied on methods that were either dangerously hazardous or inefficiently low-yielding. One conventional route involved the reaction of benzylmagnesium chloride with trifluoromethanesulfonyl fluoride, a gas with a boiling point of minus twenty-one degrees Celsius, requiring specialized cryogenic handling and posing significant safety risks in a production environment. Another method utilized photochemical reactions between iodobenzene bis(triflyl methide) and benzene, which necessitated large excesses of solvent and failed completely when attempting to synthesize derivatives with electron-withdrawing groups like fluorobenzene. These limitations severely restricted the structural diversity and commercial viability of these potent organic acids. The novel approach detailed in the patent data overcomes these barriers by employing a nucleophilic substitution strategy that is both operationally simpler and chemically more versatile, enabling the synthesis of previously inaccessible arylbis(perfluoroalkylsulfonyl)methanes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
The traditional pathways for constructing the bis(trifluoromethanesulfonyl)methane motif were fraught with technical and safety impediments that hindered widespread adoption in fine chemical manufacturing. The reliance on trifluoromethanesulfonyl fluoride gas not only introduced severe safety protocols regarding leak detection and containment but also limited the reaction scope due to the gas's high reactivity and difficulty in stoichiometric control. Furthermore, the photochemical methods suffered from poor atom economy and required extensive purification steps to remove unreacted benzene solvent, driving up production costs and waste generation. When attempting to introduce electron-deficient aryl groups, which are crucial for tuning the acidity and catalytic properties of the final molecule, these conventional methods often failed to produce any desired product, resulting in zero yield. This lack of reliability made it nearly impossible for supply chain heads to guarantee consistent availability of these critical intermediates for downstream catalyst production, creating bottlenecks in the development of new pharmaceutical processes.
The Novel Approach
The innovative methodology described in the patent data utilizes easily available aryl halomethanes as starting materials, reacting them with sodium trifluoromethanesulfinate in the presence of a phase transfer catalyst like tetrabutylammonium iodide. This nucleophilic substitution proceeds efficiently in propionitrile solvent under reflux conditions, generating arylmethyltrifluoromethylsulfone intermediates in high yields, often exceeding ninety percent. Subsequent deprotonation with tert-butyllithium followed by reaction with trifluoromethanesulfonic anhydride allows for the precise installation of the second trifluoromethanesulfonyl group. This stepwise liquid-phase synthesis eliminates the need for hazardous gases and enables the successful incorporation of bulky and electrophilic aryl groups that were previously unattainable. For procurement teams, this translates to cost reduction in fine chemical manufacturing by utilizing stable, shelf-stable reagents and standard reactor equipment, significantly lowering the barrier to entry for producing these high-value specialty chemicals.
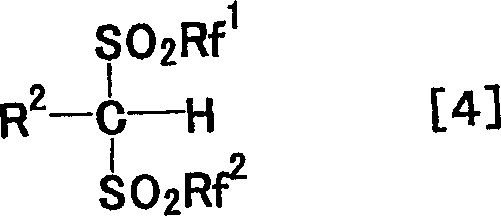
Mechanistic Insights into TfNa-Mediated Sulfonylation and Deprotonation
The core of this synthetic breakthrough lies in the careful manipulation of acidity and nucleophilicity throughout the reaction sequence. The initial step involves the nucleophilic attack of the trifluoromethanesulfinate anion on the aryl halomethane, facilitated by the polar aprotic solvent propionitrile which stabilizes the transition state. The use of tetrabutylammonium iodide enhances the solubility of the inorganic salt in the organic phase, ensuring rapid reaction kinetics even with sterically hindered substrates. Once the arylmethyltrifluoromethylsulfone is formed, the critical deprotonation step requires precise stoichiometric control. Using approximately 2.2 equivalents of tert-butyllithium ensures that the highly acidic product, which is significantly more acidic than the starting sulfone, is immediately converted to its lithium salt. This prevents side reactions such as over-triflylation, where the product might react further with trifluoromethanesulfonic anhydride to form unwanted tris-substituted byproducts. This mechanistic understanding is vital for R&D Directors aiming to replicate the process, as it highlights the importance of base equivalents in maintaining high purity and yield.
Impurity control is further achieved through the specific choice of reagents and reaction conditions that minimize the formation of side products. The patent data indicates that using less than the optimal amount of base, such as 1.2 equivalents, leads to a mixture where the desired product is deprotonated by the starting material's lithium salt, leading to a 1:1 mixture of starting material and tris-substituted impurity. By strictly adhering to the 2.2 equivalent protocol, the reaction drives quantitatively towards the lithium salt of the desired arylbis(trifluoromethanesulfonyl)methane. Subsequent quenching and acidification allow for the isolation of the free acid in high purity. This level of control over the impurity profile is essential for applications in pharmaceutical intermediates, where strict regulatory standards demand minimal levels of unknown impurities. The ability to produce high-purity fluorinated intermediates consistently ensures that downstream catalytic applications perform reliably without interference from trace contaminants.
How to Synthesize Arylbis(trifluoromethanesulfonyl)methane Efficiently
The synthesis of these complex fluorinated structures requires a disciplined approach to reaction conditions and reagent quality to ensure optimal outcomes. The process begins with the preparation of the arylmethyltrifluoromethylsulfone intermediate, which serves as the foundational building block for the final superacidic molecule. Operators must ensure that the propionitrile solvent is dry and that the reaction is conducted under an inert atmosphere to prevent moisture from degrading the sensitive organolithium intermediates in the second step. The detailed standardized synthesis steps involve precise temperature control during the addition of tert-butyllithium, typically at minus seventy-eight degrees Celsius, to manage the exothermic nature of the deprotonation. Following the addition of trifluoromethanesulfonic anhydride, the reaction is allowed to warm to room temperature to complete the sulfonylation. These operational details are critical for scaling the process from laboratory to commercial production while maintaining safety and efficiency.
- React arylhalomethane with sodium trifluoromethanesulfinate in propionitrile with TBAI catalyst under reflux.
- Deprotonate the resulting arylmethyltrifluoromethylsulfone using tert-butyllithium at low temperature.
- React the metal salt intermediate with trifluoromethanesulfonic anhydride to form the final product.
Commercial Advantages for Procurement and Supply Chain Teams
The transition from hazardous gas-based chemistry to this stable reagent-based process offers substantial benefits for supply chain reliability and operational safety. By eliminating the need for trifluoromethanesulfonyl fluoride gas, facilities can avoid the high costs associated with specialized gas handling infrastructure, leak detection systems, and rigorous safety training required for toxic gases. This shift significantly reduces the capital expenditure required to manufacture these intermediates, making the technology accessible to a broader range of chemical producers. Furthermore, the use of sodium trifluoromethanesulfinate, a solid salt, simplifies logistics and storage, as it does not require pressurized cylinders or cryogenic containment. For supply chain heads, this means reducing lead time for high-purity fluorinated compounds, as raw materials are more readily available and easier to transport globally without the regulatory burdens associated with hazardous gases. The robustness of the reaction conditions also implies fewer batch failures, ensuring a steady flow of materials for downstream catalyst synthesis.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous gaseous reagents directly lowers the raw material costs and safety compliance overhead associated with production. The high yields reported in the patent data, often exceeding ninety percent for the intermediate step, mean less waste generation and lower disposal costs, contributing to substantial cost savings. Additionally, the use of standard solvents like propionitrile and common reagents like tert-butyllithium allows for procurement from multiple global suppliers, fostering competition and driving down prices. The process efficiency reduces the need for extensive purification steps, saving both time and energy resources during the manufacturing cycle. These factors combine to create a more economically viable production model for high-value fluorinated intermediates.
- Enhanced Supply Chain Reliability: Relying on stable solid reagents rather than cryogenic gases mitigates the risk of supply disruptions caused by transportation delays or cylinder shortages. The synthetic route is robust against variations in raw material quality, ensuring consistent output even when sourcing from different vendors. This reliability is crucial for maintaining continuous production schedules in pharmaceutical and agrochemical manufacturing, where downtime can be extremely costly. The ability to store key intermediates safely allows for better inventory management, enabling companies to buffer against market fluctuations. For procurement managers, this translates to a more predictable and resilient supply chain for critical catalyst precursors.
- Scalability and Environmental Compliance: The liquid-phase nature of the reaction facilitates easy scale-up from kilogram to tonne quantities using standard stainless steel reactors. The process generates fewer hazardous byproducts compared to photochemical methods, simplifying waste treatment and ensuring compliance with increasingly stringent environmental regulations. The high atom economy of the nucleophilic substitution step minimizes the volume of chemical waste, aligning with green chemistry principles. This scalability ensures that the commercial scale-up of complex Lewis acid catalysts can proceed without significant engineering bottlenecks, supporting the growing demand for these materials in advanced organic synthesis.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these novel fluorinated compounds. The answers are derived directly from the experimental data and claims within the patent documentation, providing accurate insights for potential partners. Understanding these details helps stakeholders evaluate the feasibility of integrating this technology into their existing manufacturing workflows. The superior catalytic activity of the resulting metal salts, particularly the scandium derivatives, makes them attractive alternatives to traditional catalysts in various organic transformations.
Q: What are the advantages of this synthesis method over conventional routes?
A: This method avoids the use of hazardous trifluoromethanesulfonyl fluoride gas and achieves significantly higher yields with electron-deficient aryl groups compared to photochemical methods.
Q: Can the resulting metal salts be used as Lewis acid catalysts?
A: Yes, metal salts such as the scandium(III) derivative exhibit superior catalytic activity in benzoylation and Diels-Alder reactions compared to traditional scandium triflate.
Q: Is this process scalable for industrial production?
A: The process utilizes standard reagents and solvents like propionitrile and avoids extreme conditions, making it highly suitable for commercial scale-up of complex fluorinated intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Arylbis(trifluoromethanesulfonyl)methane Supplier
The technical potential of arylbis(trifluoromethanesulfonyl)methane and its metal salts represents a significant opportunity for advancing catalytic processes in the fine chemical industry. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that these complex molecules can be manufactured reliably at the volumes required by global enterprises. Our stringent purity specifications and rigorous QC labs guarantee that every batch meets the exacting standards necessary for pharmaceutical and agrochemical applications. We understand the critical nature of supply continuity and are committed to providing a stable source of these high-performance intermediates to support your R&D and production needs.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can benefit your specific applications. Request a Customized Cost-Saving Analysis to understand the economic impact of switching to this safer, more efficient methodology. Our experts are ready to provide specific COA data and route feasibility assessments tailored to your project requirements. By partnering with us, you gain access to cutting-edge chemical technology backed by a robust supply chain infrastructure designed to support your long-term growth and innovation goals in the competitive global market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
