Advanced Biocatalytic Synthesis of Chiral Amines Using Mutated Amine Dehydrogenase Enzymes
Introduction to Breakthrough Enzyme Engineering in Chiral Synthesis
The landscape of chiral amine production is undergoing a significant transformation driven by advanced genetic engineering, as exemplified by the innovations disclosed in patent CN110577941B. This pivotal intellectual property details the development of novel amine dehydrogenase variants derived from Bacillus nanhaiensis, specifically engineered through site-directed mutagenesis to overcome the limitations of wild-type enzymes. For R&D directors and process chemists, this represents a critical advancement in accessing high-value chiral building blocks essential for modern pharmaceutical APIs and fine chemicals. The technology leverages specific amino acid substitutions to drastically alter substrate specificity and catalytic efficiency, enabling the direct asymmetric reductive amination of ketones that were previously difficult to convert with high stereoselectivity. By shifting the paradigm from traditional chemical resolution to direct enzymatic synthesis, this approach offers a streamlined pathway to optically pure amines, addressing the growing industry demand for sustainable and efficient manufacturing processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the industrial preparation of chiral amines has relied heavily on chemical synthesis routes such as the reduction of amino acids, imine coupling, or the ring-opening of epoxy compounds, all of which present substantial operational and environmental challenges. These conventional chemical methods often necessitate the use of expensive transition metal catalysts, high-pressure hydrogenation equipment, and harsh reaction conditions that can compromise safety and increase capital expenditure. Furthermore, achieving high enantiomeric purity frequently requires complex downstream resolution steps, such as diastereomeric salt formation or chiral chromatography, which inherently limit the maximum theoretical yield to 50% and generate significant volumes of chemical waste. The reliance on heavy metals also introduces stringent regulatory hurdles regarding residual metal limits in final drug substances, necessitating additional purification stages that drive up production costs and extend lead times for reliable pharmaceutical intermediate suppliers.
The Novel Approach
In stark contrast, the novel biocatalytic approach described in the patent utilizes engineered amine dehydrogenases to facilitate asymmetric reductive amination under mild, aqueous conditions, effectively bypassing the need for hazardous reagents and extreme pressures. This enzymatic route allows for the direct conversion of prochiral ketones into chiral amines with exceptional stereocontrol, often achieving enantiomeric excess values exceeding 90% in a single step without the need for resolution. The use of ammonia or simple amino acids as nitrogen donors simplifies the reaction stoichiometry, while the inherent specificity of the enzyme minimizes the formation of by-products, thereby simplifying the work-up and purification procedures. This shift not only enhances the overall process mass intensity but also aligns with green chemistry principles by reducing the environmental footprint associated with solvent usage and waste generation, making it an attractive option for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Mutated Amine Dehydrogenase Catalysis
The core innovation lies in the precise modification of the enzyme's active site through specific point mutations, including G131L/M, N262V/L, Y285L/M, and M333D, which collectively reshape the binding pocket to accommodate a broader range of ketone substrates. These mutations alter the steric and electronic environment surrounding the catalytic center, enhancing the enzyme's affinity for aliphatic and aryl ketones while maintaining strict control over the stereochemical outcome of the hydride transfer from the NADH cofactor. The mechanism proceeds via the formation of a transient imine intermediate between the ketone substrate and the amino donor, which is subsequently reduced by the enzyme-bound cofactor to yield the chiral amine product with high fidelity. This catalytic cycle is highly efficient, as the enzyme stabilizes the transition state for the pro-R or pro-S hydride attack, ensuring that the resulting amine possesses the desired configuration required for downstream drug synthesis.

Furthermore, the structural flexibility introduced by these mutations allows the enzyme to tolerate diverse functional groups on the substrate, as defined by the general ketone structure where R1 and R2 can vary from alkyl to aryl moieties. This versatility is crucial for industrial applications where a single biocatalyst platform needs to be adapted for multiple product lines without extensive re-engineering. The ability to operate effectively across a wide pH range of 8 to 12 and temperatures up to 70°C demonstrates the robustness of these mutants, making them suitable for large-scale fermentation and whole-cell catalysis processes. By understanding these mechanistic nuances, process developers can optimize reaction parameters such as cofactor regeneration and substrate feeding strategies to maximize space-time yields and ensure consistent product quality.
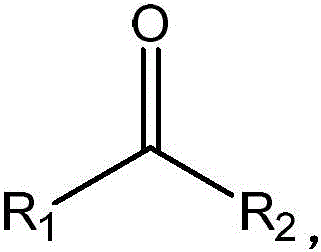
How to Synthesize Chiral Amines Efficiently
Implementing this biocatalytic route requires a systematic approach to strain construction and reaction optimization to fully leverage the potential of the mutated amine dehydrogenase. The process begins with the design of specific primers for PCR-based mutagenesis to introduce the desired amino acid changes into the phenylalanine dehydrogenase gene sequence, followed by transformation into a suitable host organism like E. coli BL21(DE3). Once the recombinant strains are established, cultivation conditions must be carefully controlled to induce high-level expression of the soluble, active enzyme, often utilizing auto-induction media or specific temperature shifts to prevent inclusion body formation. The subsequent catalytic step involves mixing the crude enzyme or whole cells with the ketone substrate, an ammonia source, and a cofactor regeneration system in a buffered solution, where parameters like agitation speed and oxygen transfer are critical for maintaining enzyme stability and activity throughout the reaction duration.
- Construct recombinant expression strains by introducing specific point mutations (e.g., G131L, N262V) into the phenylalanine dehydrogenase gene sequence using PCR-based site-directed mutagenesis.
- Cultivate the transformed E. coli BL21(DE3) strains in LB medium with IPTG induction to express the His-tagged mutant amine dehydrogenase enzyme.
- Perform asymmetric reductive amination by reacting target ketones with an amino donor (ammonia or alanine) and NADH cofactor in a buffered aqueous system at controlled pH and temperature.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this enzymatic technology translates into tangible strategic advantages regarding cost structure, supply reliability, and regulatory compliance. By eliminating the dependency on precious metal catalysts such as rhodium or iridium, which are subject to volatile market pricing and geopolitical supply risks, manufacturers can achieve significant cost reduction in manufacturing operations. The simplified downstream processing, resulting from the high selectivity of the enzyme, reduces the consumption of organic solvents and chromatography materials, further driving down the variable costs associated with production. Additionally, the aqueous nature of the reaction medium mitigates the hazards associated with flammable organic solvents and high-pressure hydrogen gas, leading to lower insurance premiums and reduced safety infrastructure requirements.
- Cost Reduction in Manufacturing: The transition to biocatalysis removes the need for expensive chiral ligands and transition metals, which are major cost drivers in traditional asymmetric synthesis, thereby substantially lowering the raw material bill. The high atom economy of the reductive amination reaction ensures that a greater proportion of the starting materials are converted into the final product, minimizing waste disposal costs and maximizing resource efficiency. Furthermore, the ability to use inexpensive ammonia or alanine as nitrogen sources instead of complex chiral amines for resolution significantly reduces the input cost per kilogram of the final API intermediate.
- Enhanced Supply Chain Reliability: Relying on fermentation-based production decouples the supply chain from the fluctuations of the petrochemical industry, as the primary feedstocks are renewable sugars and simple salts. The robustness of the engineered enzymes allows for consistent batch-to-batch performance, reducing the risk of production delays caused by catalyst deactivation or inconsistent reaction outcomes. This stability ensures a continuous and predictable supply of high-purity chiral amines, which is critical for meeting the rigorous delivery schedules of global pharmaceutical clients.
- Scalability and Environmental Compliance: The process is inherently scalable from laboratory shake flasks to multi-ton industrial fermenters without the need for specialized high-pressure reactors, facilitating rapid commercial scale-up of complex pharmaceutical intermediates. The reduction in hazardous waste generation and the use of biodegradable aqueous systems simplify environmental permitting and compliance with increasingly strict global regulations on industrial emissions. This alignment with sustainability goals enhances the corporate reputation of suppliers and meets the ESG criteria demanded by major multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this amine dehydrogenase technology in industrial settings. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this biocatalytic route into their existing manufacturing portfolios.
Q: What specific mutations improve the catalytic activity of the amine dehydrogenase?
A: The patent identifies key mutations at positions 131 (G to L/M), 262 (N to V/L), 285 (Y to L/M), and 333 (M to D) which significantly enhance selectivity and activity towards aliphatic and aryl ketones.
Q: How does this biocatalytic method compare to traditional chemical synthesis?
A: Unlike chemical methods that often require high pressure, toxic metals, and complex resolution steps, this enzymatic approach operates under mild conditions with high enantiomeric excess (ee) and eliminates heavy metal contamination.
Q: What are the optimal reaction conditions for this enzyme?
A: The enzyme functions effectively within a pH range of 8-12 and temperatures between 10-70°C, utilizing NADH as a cofactor and ammonia or alanine as the nitrogen source.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Amines Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the enzymatic technologies described in patent CN110577941B and are well-positioned to bring these innovations to commercial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced chiral HPLC and GC systems to guarantee that every batch of chiral amine meets the highest standards of optical purity and chemical integrity required by the global pharmaceutical industry.
We invite you to collaborate with us to explore how this advanced biocatalytic route can optimize your supply chain and reduce your overall production costs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific target molecules. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities align with your project goals and timelines.
