Advanced Synthesis of Potent Tetrahydrocarbazole Derivatives for Next-Generation Anti-Tuberculosis Therapeutics
Advanced Synthesis of Potent Tetrahydrocarbazole Derivatives for Next-Generation Anti-Tuberculosis Therapeutics
The global fight against tuberculosis (TB) faces a critical challenge with the rising prevalence of multidrug-resistant strains, necessitating the urgent development of novel therapeutic agents with distinct mechanisms of action. Patent CN114560804A introduces a groundbreaking class of tetrahydrocarbazole derivatives, defined by the general Formula I, which demonstrate exceptional efficacy against Mycobacterium tuberculosis. These compounds are designed to overcome the limitations of current first-line treatments like isoniazid, offering superior potency and a reduced risk of resistance. The molecular architecture features a fused tetrahydrocarbazole core substituted with specific aryl or heteroaryl groups at the nitrogen and aromatic positions, optimizing their interaction with biological targets such as dihydrofolate reductase (DHFR). 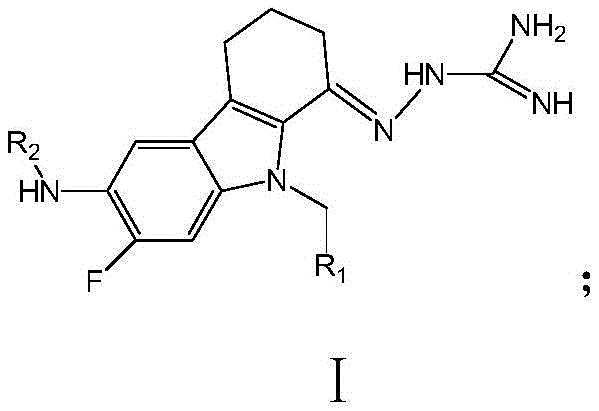
For pharmaceutical R&D teams and procurement strategists, understanding the synthetic accessibility of these high-value intermediates is paramount. The disclosed preparation method is not merely a laboratory curiosity but a robust, scalable process engineered for industrial viability. By leveraging a sequence of substitution, reduction, and transition-metal catalyzed coupling reactions, the synthesis achieves high target product yields with minimal side reactions. This technical breakthrough positions these tetrahydrocarbazole derivatives as a reliable pharmaceutical intermediate supplier option for companies aiming to diversify their anti-infective pipelines. The stability of the yield and performance across different batches ensures a consistent supply chain, crucial for long-term drug development projects.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing complex heterocyclic scaffolds often suffer from harsh reaction conditions, poor regioselectivity, and the generation of difficult-to-remove impurities that compromise the final API quality. In the context of anti-tuberculosis agents, many existing synthetic routes rely on multiple protection-deprotection steps or utilize expensive, toxic reagents that hinder commercial scalability. Furthermore, conventional methods frequently struggle to introduce diverse functional groups at specific positions on the carbazole ring without affecting other sensitive moieties, limiting the structure-activity relationship (SAR) exploration necessary for optimizing potency. The inability to consistently achieve high purity without extensive downstream processing leads to increased manufacturing costs and extended lead times, creating bottlenecks for reliable agrochemical intermediate supplier networks and pharma partners alike.
The Novel Approach
The methodology outlined in CN114560804A represents a significant paradigm shift by employing a streamlined, convergent synthesis strategy that maximizes atom economy and operational simplicity. Instead of linear, step-intensive sequences, this novel approach constructs the core scaffold efficiently and then diversifies it through modular coupling reactions. The use of a composite catalyst system in the substitution steps allows for milder conditions and higher selectivity, drastically reducing the formation of by-products. This innovation directly addresses the pain points of cost reduction in pharmaceutical intermediate manufacturing by minimizing waste and solvent usage. Moreover, the process is designed to be robust against variations in raw material quality, ensuring that the final tetrahydrocarbazole derivatives maintain stringent purity specifications required for clinical applications, thereby enhancing the overall reliability of the supply chain.
Mechanistic Insights into Palladium-Catalyzed Cross-Coupling and Scaffold Construction
The heart of this synthesis lies in the precise construction of the fluorinated nitro-tetrahydrocarbazole core, achieved through an acid-catalyzed cyclization reminiscent of the Fischer indole synthesis but optimized for this specific substrate. As illustrated in the reaction scheme, 4-nitro-5-fluorophenylhydrazine reacts with 1,2-cyclohexanedione under acidic conditions to form the tricyclic ketone scaffold. 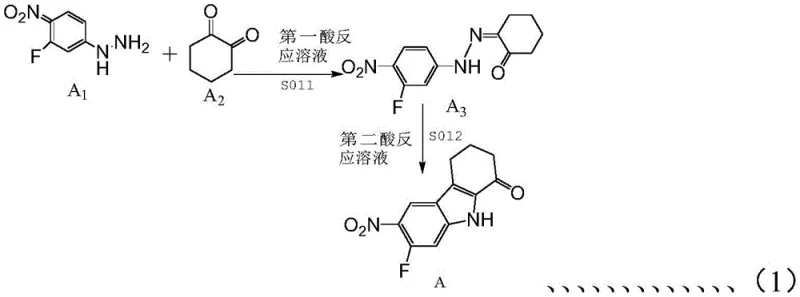
This step is critical as it establishes the stereochemistry and electronic properties of the core ring system. The subsequent N-alkylation and nitro-reduction steps are carefully tuned to preserve the integrity of the fluorine substituent, which is essential for the compound's metabolic stability and binding affinity. The process avoids over-reduction or dehalogenation, common pitfalls in similar chemistries, by utilizing specific reducing agents like iron powder in controlled acidic media. This level of control ensures that the intermediate amines are generated in high purity, ready for the subsequent coupling reactions without the need for exhaustive purification, thus streamlining the overall workflow.
A key mechanistic highlight is the employment of a palladium-catalyzed Buchwald-Hartwig amination to install the diverse aryl or heteroaryl groups at the aniline position. This transformation utilizes a composite catalyst system comprising a palladium source, a specialized ligand like binaphthol, and a strong base. 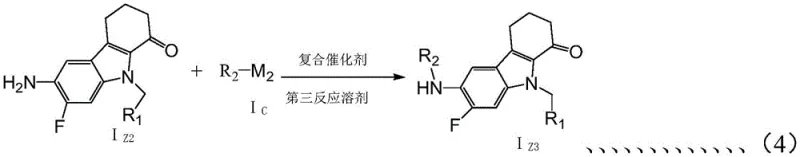
The choice of ligand and base is instrumental in facilitating the oxidative addition and reductive elimination steps of the catalytic cycle, enabling the coupling of sterically hindered or electron-deficient aryl halides that would otherwise be unreactive. This mechanistic sophistication allows for the rapid generation of a library of derivatives with varying electronic and steric properties, accelerating the SAR study process. For R&D directors, this means faster iteration cycles in identifying lead candidates with optimal pharmacokinetic profiles. The robustness of this catalytic system also implies that the reaction can be scaled up with confidence, maintaining high conversion rates and minimizing the residual palladium content in the final product through standard workup procedures.
How to Synthesize Tetrahydrocarbazole Derivatives Efficiently
The synthesis of these potent anti-tuberculosis agents follows a logical, four-step sequence that balances chemical complexity with operational feasibility. Beginning with the construction of the nitro-fluoro scaffold, the process moves through selective alkylation and reduction before culminating in the final condensation with aminoguanidine bicarbonate. Each step has been optimized to maximize yield and minimize impurity profiles, ensuring that the final product meets the rigorous standards expected of high-purity pharmaceutical intermediates. The detailed标准化 synthesis steps见下方的指南 provide a comprehensive roadmap for replicating this chemistry in a GMP-compliant environment.
- Construct the 6-nitro-7-fluoro-2,3,4,9-tetrahydrocarbazol-1-one scaffold via acid-catalyzed cyclization of phenylhydrazine and cyclohexanedione.
- Perform N-alkylation using benzyl bromide derivatives and cesium carbonate, followed by reduction of the nitro group to an amine.
- Execute a palladium-catalyzed Buchwald-Hartwig amination with aryl halides, concluding with condensation using aminoguanidine bicarbonate.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis route offers substantial strategic advantages for procurement managers and supply chain heads looking to optimize their sourcing strategies for anti-infective APIs. The process is designed with scalability in mind, utilizing readily available starting materials and common solvents that mitigate the risk of supply disruptions. By eliminating the need for exotic reagents or cryogenic conditions, the manufacturing footprint is significantly simplified, leading to enhanced supply chain reliability. This robustness ensures that production schedules can be met consistently, reducing the lead time for high-purity pharmaceutical intermediates and allowing partners to respond more agilely to market demands.
- Cost Reduction in Manufacturing: The streamlined nature of the synthesis, characterized by high yields and fewer purification steps, translates directly into lower production costs. The efficient use of catalysts and the ability to recycle solvents further contribute to economic efficiency. By avoiding complex protection group strategies and minimizing waste generation, the overall cost of goods sold (COGS) is optimized, making these derivatives a cost-effective choice for large-scale API manufacturing without compromising on quality or performance.
- Enhanced Supply Chain Reliability: The reliance on stable, commercially available raw materials reduces dependency on single-source suppliers for niche chemicals. The robustness of the reaction conditions means that the process is less susceptible to minor fluctuations in temperature or reagent quality, ensuring consistent batch-to-batch performance. This reliability is crucial for maintaining continuous production lines and meeting the stringent delivery timelines required by global pharmaceutical clients, thereby strengthening the partnership between manufacturers and their downstream customers.
- Scalability and Environmental Compliance: The synthesis route is inherently green, generating fewer hazardous by-products and utilizing safer reagents compared to traditional methods. This aligns with increasingly strict environmental regulations and corporate sustainability goals. The ease of scale-up from gram to kilogram quantities without significant re-optimization demonstrates the process maturity, allowing for rapid transition from pilot plant to full commercial production. This scalability ensures that supply can easily match demand surges, providing a secure and sustainable source of critical medicinal ingredients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these tetrahydrocarbazole derivatives. The answers are derived directly from the patent data and reflect the practical realities of implementing this chemistry in an industrial setting. Understanding these details helps stakeholders make informed decisions about integrating these intermediates into their development pipelines.
Q: What is the primary biological activity of these tetrahydrocarbazole derivatives?
A: These derivatives exhibit excellent anti-tuberculosis activity, often surpassing isoniazid in potency with low MIC values and minimal drug resistance development against Mycobacterium tuberculosis.
Q: How does the synthesis route ensure high purity for pharmaceutical applications?
A: The process utilizes controlled reaction conditions, specific composite catalysts for coupling, and rigorous purification steps like silica gel column chromatography to minimize side reactions and impurities.
Q: Is this synthesis method scalable for commercial production?
A: Yes, the patent highlights stable yields, easily controlled process conditions, and the use of common solvents and reagents, making it highly suitable for scale-up from laboratory to industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetrahydrocarbazole Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of having a dependable partner for complex pharmaceutical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and efficiency. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical techniques to verify every batch. Our capability to handle complex chemistries, such as the palladium-catalyzed couplings described in this patent, underscores our technical proficiency and dedication to quality.
We invite you to collaborate with us to accelerate your anti-tuberculosis drug development programs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your journey from discovery to commercialization, ensuring a seamless and successful partnership.
