Advanced Synthesis of Donor-Acceptor OLED Materials: Scaling High-Purity Electroluminescent Compounds
Advanced Synthesis of Donor-Acceptor OLED Materials: Scaling High-Purity Electroluminescent Compounds
The landscape of organic optoelectronics is constantly evolving, driven by the demand for more efficient and cost-effective light-emitting materials. Patent CN101967373B introduces a significant breakthrough in the field of donor-acceptor type organic electroluminescent materials, specifically targeting the limitations of traditional red-emitting dopants. This technology presents a novel class of compounds, generally referred to as DCD(n-R)SC, which are synthesized through a highly efficient one-step reaction pathway. For R&D directors and procurement specialists in the display industry, this patent represents a pivotal shift away from complex, multi-step syntheses towards a streamlined manufacturing process that promises both economic and operational advantages. The core innovation lies in the molecular design that utilizes non-nitrogen-containing aromatic groups as electron donors, challenging the long-held consensus that amine groups are strictly necessary for high-performance luminescence.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industry has relied heavily on 4-(dinitrilemethylene)-2-methyl-6-(4-dimethylamino-styrene)-4H-pyran, commonly known as DCM, and its derivatives like DCJTB as standard red-emitting dopants. While these materials offer excellent comprehensive performance, their preparation methods are notoriously cumbersome and inefficient. The conventional synthesis typically requires more than five distinct organic reaction steps, creating a complex workflow that is difficult to optimize for large-scale production. Furthermore, these multi-step processes are prone to generating symmetrical structural byproducts, which drastically lowers the overall yield and complicates the separation and purification stages. The reliance on rare or difficult-to-source raw materials further exacerbates the production costs, making the commercial scale-up of complex polymer additives and OLED intermediates based on the DCM skeleton a significant financial burden for manufacturers seeking cost reduction in electronic chemical manufacturing.
The Novel Approach
In stark contrast to the legacy methods, the technology disclosed in CN101967373B employs a direct Knoevenagel condensation reaction that simplifies the entire synthetic route into a single, manageable operation. By reacting aromatic aldehyde derivatives with isophorone and malononitrile in a specific mixed solvent system, the process achieves high yields ranging from 65% to over 93% depending on the substituent. This approach eliminates the need for intermediate isolation and reduces the consumption of solvents and reagents associated with multi-step sequences. The simplicity of the reaction conditions, which operate at moderate temperatures between 68°C and 84°C, enhances safety and energy efficiency. Moreover, the purification process is straightforward, utilizing standard recrystallization techniques to achieve purities exceeding 99%, thereby ensuring that the final high-purity OLED material meets the stringent quality requirements of modern display applications without the need for expensive chromatographic separations.
Mechanistic Insights into Knoevenagel Condensation and Intramolecular Charge Transfer
The chemical foundation of this innovation rests on the formation of an intramolecular charge transfer (ICT) compound featuring a distinct electron donor-electron acceptor structural unit. The reaction mechanism involves the condensation of the active methylene group of the isophorone-malononitrile adduct with the carbonyl group of the aromatic aldehyde. This creates a highly conjugated system where the dicyanomethylene group acts as a strong electron acceptor, while the substituted styryl moiety serves as the electron donor. Uniquely, this patent demonstrates that effective luminescence can be achieved even when the donor unit is a simple aromatic group lacking nitrogen atoms, such as hydroxyl-substituted phenyl rings. This finding overturns the previous technical bias that nitrogen-containing groups were essential for reducing molecular interaction at high concentrations and maintaining energy levels suitable for red emission.
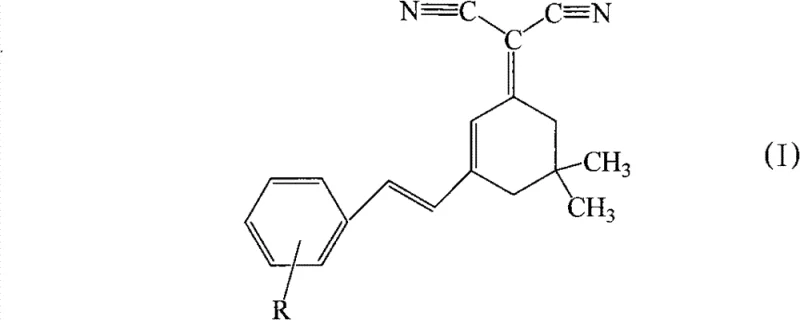
The resulting molecular architecture facilitates efficient radiative decay, evidenced by maximum emission wavelengths consistently greater than 535nm, which falls within the desirable orange-red to red spectrum for full-color displays. The rigidity introduced by the cyclohexene ring, derived from isophorone, contributes to narrower fluorescence emission compared to traditional DCM series compounds, resulting in superior color purity for the end device. Furthermore, the presence of various substituents (R groups such as hydroxyl, alkyl, alkoxy, or halogens) allows for fine-tuning of the electronic properties and solubility profiles. This tunability is crucial for R&D teams aiming to optimize the compatibility of the dopant with various host materials like Alq3, ensuring uniform dispersion and preventing aggregation-induced quenching in the final emissive layer.
How to Synthesize DCD(n-R)SC Efficiently
The synthesis protocol outlined in the patent provides a robust framework for producing these advanced electroluminescent materials with high reproducibility. The process begins with the preparation of a catalytic mixture comprising acetic acid, acetic anhydride, piperidine, and N,N-dimethylformamide (DMF), which serves as both solvent and reaction medium. Malononitrile and isophorone are introduced first to form the initial adduct under mild stirring conditions before the temperature is elevated. Subsequently, the specific aromatic aldehyde derivative is added to drive the final condensation step. The detailed standardized synthesis steps see the guide below for precise molar ratios and temperature controls required to maximize yield and minimize side reactions.
- Prepare a catalytic mixed solution of acetic acid, acetic anhydride, piperidine, and DMF, then add malononitrile and isophorone at room temperature.
- Heat the mixture to 70-82°C, add the specific aromatic aldehyde derivative, and maintain reaction temperature between 68-84°C for 60-90 minutes.
- Precipitate the product by pouring into hot dilute hydrochloric acid, followed by filtration and multiple recrystallizations using chloroform and ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers tangible benefits that extend beyond mere chemical elegance. The reduction in synthetic steps directly correlates to a significant decrease in manufacturing lead time and operational complexity. By eliminating the need for multiple intermediate isolations and the associated solvent exchanges, the process inherently reduces the consumption of utilities and labor hours. This streamlining of the production workflow translates into substantial cost savings, making the sourcing of reliable OLED material suppliers more economically viable for downstream display manufacturers. Additionally, the use of commodity chemicals like isophorone and malononitrile ensures a stable and continuous supply of raw materials, mitigating the risks associated with sourcing specialized or scarce reagents.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and the reduction of reaction steps from five or more down to a single pot operation drastically simplifies the production economics. Without the need for expensive heavy metal removal processes or complex chromatographic purification, the overall cost of goods sold is significantly optimized. The high atom economy of the Knoevenagel condensation further ensures that raw material utilization is maximized, reducing waste disposal costs and enhancing the overall profitability of the manufacturing process.
- Enhanced Supply Chain Reliability: The reliance on widely available bulk chemicals such as substituted benzaldehydes, isophorone, and malononitrile ensures that the supply chain is resilient against market fluctuations. Unlike proprietary intermediates that may have limited suppliers, these starting materials are produced by numerous global chemical manufacturers, guaranteeing continuity of supply. This accessibility allows for flexible sourcing strategies and reduces the risk of production stoppages due to raw material shortages, thereby securing the delivery schedules for high-purity organic electroluminescent materials.
- Scalability and Environmental Compliance: The process is inherently scalable due to its simple thermal profile and the use of standard precipitation techniques for product isolation. The workup procedure, which involves pouring the reaction mixture into hot dilute hydrochloric acid, is easily adaptable from laboratory to industrial scales without requiring specialized high-pressure or cryogenic equipment. Furthermore, the ability to achieve high purity through recrystallization rather than column chromatography reduces the volume of organic solvent waste generated, aligning with increasingly stringent environmental regulations and sustainability goals in the fine chemical sector.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these donor-acceptor type materials. The answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on performance metrics and processing capabilities. Understanding these details is essential for stakeholders evaluating the feasibility of integrating these materials into existing OLED fabrication lines.
Q: What is the primary advantage of DCD-SC materials over traditional DCM dyes?
A: Unlike traditional DCM dyes which require cumbersome multi-step synthesis and often produce symmetrical byproducts, DCD-SC materials utilize a simplified one-step Knoevenagel condensation. This results in significantly higher yields, easier purification, and avoids the use of rare or expensive raw materials typically associated with pyran-based laser dyes.
Q: What is the emission characteristic of these donor-acceptor compounds?
A: These compounds function as intramolecular charge transfer materials with maximum emission wavelengths greater than 535nm, placing them firmly in the long-wave red to orange-red region. They exhibit excellent film-forming properties and maintain high luminescent performance even without nitrogen-containing groups as electron donors.
Q: How is high purity achieved in the commercial production of these intermediates?
A: High purity, typically reaching 99%, is achieved through a robust purification protocol involving vacuum filtration, washing with deionized water, and repeated recrystallization using a specific 3:1 volume ratio of chloroform to ethyl acetate. This ensures the removal of insoluble impurities and unreacted starting materials.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable DCD-SC Supplier
As the demand for high-performance display technologies continues to surge, the ability to source high-quality organic electroluminescent intermediates becomes a critical strategic priority. NINGBO INNO PHARMCHEM stands ready to support your development and production needs with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with rigorous QC labs and advanced analytical instrumentation to ensure that every batch of DCD-SC derivatives meets stringent purity specifications, including precise control over impurity profiles and optical properties. We understand the critical nature of consistency in the OLED supply chain and are committed to delivering materials that enable superior device performance and longevity.
We invite you to engage with our technical team to discuss how this patented technology can be integrated into your specific application requirements. Whether you need a Customized Cost-Saving Analysis for switching from traditional DCM dopants or require specific COA data and route feasibility assessments for new derivatives, our experts are available to provide comprehensive support. Contact our technical procurement team today to initiate a dialogue on securing a reliable supply of next-generation organic electroluminescent materials that balance performance with economic efficiency.
