Revolutionizing OLED Manufacturing: Efficient One-Step Synthesis of Donor-Acceptor Luminescent Materials
Revolutionizing OLED Manufacturing: Efficient One-Step Synthesis of Donor-Acceptor Luminescent Materials
The landscape of organic electroluminescence is undergoing a significant transformation driven by the need for more efficient, cost-effective, and scalable manufacturing processes for display technologies. Patent CN101967373B introduces a groundbreaking class of donor-acceptor type organic electroluminescent materials, designated generally as DCD(n-R)SC, which challenge long-standing technical biases in molecular design. Traditionally, high-performance red-emitting dopants relied heavily on nitrogen-containing groups as electron donors, a constraint that often complicated synthesis and limited structural diversity. This innovation demonstrates that aromatic groups without amine functionalities can serve as effective electron donors, achieving comparable or superior luminescent performance while drastically simplifying the synthetic pathway. For R&D directors and procurement strategists in the display industry, this represents a pivotal shift towards more robust supply chains and reduced production complexity.
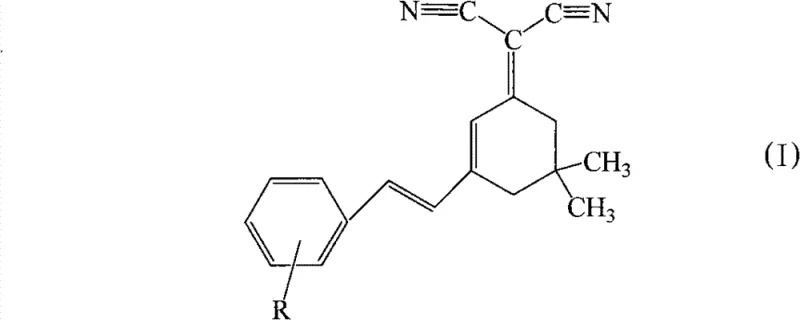
The core of this technology lies in its versatile molecular architecture, expressed by general formula (I), where the substituent R can be varied to include hydroxyl groups, alkyl chains, alkoxy groups, or halogens. This modularity allows for fine-tuning of the electronic properties and emission wavelengths, which are consistently observed in the long-wave region greater than 535nm. By decoupling the requirement for amine-based donors, the invention opens new avenues for material stability and film-forming properties, critical factors for the longevity and efficiency of OLED devices. The ability to synthesize these complex conjugated systems in fewer steps directly addresses the industry's pain points regarding yield loss and purification bottlenecks associated with legacy materials like DCJTB.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the gold standard for red-emitting OLED dopants has been materials like DCJTB, a classic doped red light-emitting material developed by Kodak. While DCJTB offers excellent comprehensive performance, its manufacturing process is notoriously cumbersome, typically requiring more than five distinct organic reaction steps to complete. This multi-step synthesis not only increases the consumption of solvents and reagents but also significantly elevates the risk of generating symmetrical by-products that are structurally similar to the target molecule. Consequently, the separation and purification of the final product become extremely difficult and costly, often resulting in low overall yields that hinder large-scale commercial application. Furthermore, the raw materials required for these traditional pathways are often rare or expensive specialty chemicals, creating supply chain vulnerabilities and driving up the unit cost of the final electroluminescent material, which is a major barrier for mass-market display adoption.
The Novel Approach
In stark contrast, the methodology described in patent CN101967373B utilizes a streamlined one-pot Knoevenagel condensation reaction that merges three readily available starting materials: an aromatic aldehyde derivative, malononitrile, and isophorone. This novel approach eliminates the need for intermediate isolation and complex protection-deprotection sequences, collapsing what was once a five-step process into a single, continuous operation. The reaction proceeds efficiently in a mixed solvent system of acetic acid, acetic anhydride, piperidine, and DMF, with temperatures carefully controlled between 70°C and 82°C to optimize conversion rates. Experimental data from the patent indicates that this method can achieve impressive yields, reaching as high as 93.4% for specific derivatives like DCD(4-OH)SC, compared to the significantly lower yields typical of the DCJTB route. This drastic reduction in process complexity translates directly into lower operational expenditures and a much smaller environmental footprint due to reduced waste generation.
Mechanistic Insights into Knoevenagel Condensation and Intramolecular Charge Transfer
The success of this synthetic route relies on the precise orchestration of a Knoevenagel condensation mechanism facilitated by a basic catalyst system involving piperidine and acetic anhydride. In this reaction, the active methylene group of the malononitrile-isophorone intermediate acts as a nucleophile, attacking the carbonyl carbon of the aromatic aldehyde derivative. The subsequent elimination of water leads to the formation of a highly conjugated carbon-carbon double bond system that links the electron-rich aromatic donor unit with the electron-deficient dicyanomethylene acceptor unit. This extended pi-conjugation is essential for lowering the energy gap between the highest occupied molecular orbital (HOMO) and the lowest unoccupied molecular orbital (LUMO), thereby enabling the desired long-wavelength emission in the orange-red spectrum. The use of acetic anhydride likely serves to activate the aldehyde or sequester water, driving the equilibrium towards product formation and preventing hydrolysis of the sensitive nitrile groups.
From an electronic structure perspective, these materials function as intramolecular charge transfer (ICT) compounds, where the excitation involves the movement of electron density from the aromatic donor to the polynitrile acceptor. A key mechanistic breakthrough here is the validation that non-coplanar aromatic structures without nitrogen atoms can effectively serve as electron donors. Previous consensus suggested that nitrogen-containing groups were necessary to prevent excessive molecular aggregation and quenching at high concentrations. However, the steric bulk provided by the gem-dimethyl groups on the cyclohexene ring of the isophorone moiety effectively prevents close packing of the dye molecules, maintaining high fluorescence quantum yields even in the solid state. This structural insight allows chemists to explore a broader chemical space for donor units, potentially accessing new color coordinates and stability profiles that were previously inaccessible with amine-based designs.
How to Synthesize DCD(n-R)SC Efficiently
The practical implementation of this synthesis requires careful attention to stoichiometry and temperature control to maximize the yield of the target donor-acceptor compound. The process begins by preparing a catalytic solvent matrix where the volume ratios of acetic acid, acetic anhydride, piperidine, and DMF are strictly maintained to ensure optimal solubility and reaction kinetics. Malononitrile and isophorone are introduced first to form the active intermediate before the aromatic aldehyde is added, a sequence that minimizes side reactions such as self-condensation of the aldehyde. Following the reaction, the crude product is precipitated into hot dilute hydrochloric acid, which helps to neutralize the basic catalyst and crash out the organic product.
- Prepare a catalytic mixed solution of acetic acid, acetic anhydride, piperidine, and DMF, then add malononitrile and isophorone at room temperature.
- Heat the mixture to 70-82°C to activate the intermediate, then introduce the specific aromatic aldehyde derivative for the condensation reaction.
- Quench the reaction into hot dilute hydrochloric acid, collect the precipitate, and purify via repeated recrystallization in chloroform/ethyl acetate to achieve >99% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers profound strategic benefits beyond mere technical performance. The most immediate impact is the substantial reduction in manufacturing costs driven by the simplification of the process flow. By eliminating multiple reaction steps and the associated work-up procedures, facilities can reduce labor hours, energy consumption, and solvent usage, leading to a significantly lower cost of goods sold (COGS). Furthermore, the reliance on commodity chemicals like isophorone and malononitrile, which are produced on a massive global scale for other industries, ensures a stable and resilient supply chain that is less susceptible to the volatility often seen with specialized fine chemical intermediates. This availability allows for better long-term planning and inventory management, reducing the risk of production stoppages due to raw material shortages.
- Cost Reduction in Manufacturing: The consolidation of the synthesis into a single step removes the need for expensive transition metal catalysts and complex purification columns often required in multi-step routes. This simplification means that the capital expenditure for reactor trains can be optimized, as the same vessel can be used for the entire reaction cycle without intermediate cleaning or transfer. Additionally, the high crude purity achieved directly from precipitation reduces the load on downstream purification units, saving significant amounts of recrystallization solvents like chloroform and ethyl acetate. These cumulative efficiencies result in a drastically simplified cost structure that enhances competitiveness in the price-sensitive display market.
- Enhanced Supply Chain Reliability: Sourcing strategies are greatly improved because the key building blocks are not proprietary or single-source restricted. Isophorone and various substituted benzaldehydes are widely available from multiple global suppliers, providing procurement teams with the leverage to negotiate better pricing and terms. The robustness of the reaction conditions, which tolerate moderate temperatures and standard atmospheric pressure, further reduces the dependency on specialized high-pressure or cryogenic equipment, making the technology transferable to a wider range of manufacturing partners. This flexibility ensures continuity of supply even in the face of regional disruptions or logistical challenges.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, moving seamlessly from gram-scale laboratory experiments to tonnage-level commercial production without fundamental changes to the chemistry. The use of aqueous acid quenching and simple filtration aligns well with standard wastewater treatment protocols, minimizing the generation of hazardous heavy metal waste often associated with cross-coupling reactions. This environmental compatibility facilitates easier regulatory approval and permitting for new production lines, accelerating the time-to-market for new OLED formulations. The ability to produce high-purity material with minimal waste supports corporate sustainability goals and reduces the overall environmental liability of the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this donor-acceptor material technology. These answers are derived directly from the experimental data and structural analysis provided in the patent documentation, ensuring accuracy for decision-makers evaluating this route for adoption. Understanding these details is crucial for assessing the feasibility of integrating these materials into existing OLED fabrication lines.
Q: What is the primary advantage of this donor-acceptor material over traditional DCJTB?
A: Unlike DCJTB which requires cumbersome multi-step synthesis with low yields, this patent utilizes a one-step Knoevenagel condensation that significantly simplifies production, reduces raw material costs, and achieves yields up to 93.4% with easier purification.
Q: Can the emission color be tuned using this synthetic route?
A: Yes, by varying the substituent R on the aromatic aldehyde (such as hydroxyl, halogen, or alkoxy groups), the maximum emission wavelength can be adjusted across the orange-red spectrum (>535nm), allowing for precise color coordinate tuning.
Q: Is this material suitable for large-scale commercial production?
A: Absolutely. The process uses commodity chemicals like isophorone and malononitrile, operates at moderate temperatures (70-82°C), and employs simple filtration and recrystallization for purification, making it highly scalable for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable DCD(n-R)SC Supplier
As the demand for high-performance organic electroluminescent materials continues to surge, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM provides the technical assurance needed for successful commercialization. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory curiosity to industrial reality is seamless and efficient. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of verifying the structural integrity and optical properties of every batch, guaranteeing that the materials meet the exacting standards required for next-generation display applications. We understand the critical nature of supply consistency in the electronics sector and have built our operations to prioritize reliability and quality above all else.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific product requirements. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this streamlined manufacturing process for your specific portfolio. We encourage you to reach out for specific COA data and route feasibility assessments to validate the performance of these donor-acceptor compounds in your device architectures. Let us collaborate to drive down costs and accelerate the deployment of advanced lighting and display technologies through superior chemical engineering.
