Advanced Synthesis of Fluorinated Azobenzene Photochromic Compounds for High-Performance Optical Materials
Advanced Synthesis of Fluorinated Azobenzene Photochromic Compounds for High-Performance Optical Materials
The rapid evolution of optoelectronic technologies demands materials with precise control over light-matter interactions, driving significant innovation in the field of photochromic compounds. Patent CN101503376A introduces a groundbreaking class of azobenzene derivatives featuring fluorine-terminated chiral carbon groups, addressing critical limitations found in traditional photochromic systems. This technological advancement represents a pivotal shift for manufacturers seeking reliable electronic chemical supplier partnerships, as it delivers compounds with markedly improved spectral differentiation between cis and trans isomeric states. The integration of perfluorinated chains onto the azobenzene backbone not only enhances the magnitude of UV-Vis absorption changes upon irradiation but also ensures exceptional thermal stability at ambient conditions. For R&D directors and procurement specialists in the display and data storage sectors, this development offers a robust pathway toward next-generation optical switching materials that maintain performance integrity over extended operational lifecycles without degradation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional azobenzene-based photochromic materials have long struggled with inherent physicochemical deficiencies that restrict their utility in high-performance commercial applications. The primary bottleneck lies in the insufficient spectral contrast between the stable trans-isomer and the metastable cis-isomer generated upon photo-excitation, resulting in weak signal modulation that is often inadequate for precision optical recording or switching. Furthermore, conventional derivatives frequently exhibit poor thermal stability, leading to spontaneous relaxation or degradation at room temperature, which compromises the longevity and reliability of devices such as liquid crystal displays or optical memory units. The synthetic routes for modifying these core structures often involve harsh conditions or expensive catalysts that introduce complex impurity profiles, necessitating costly downstream purification processes that erode profit margins. Additionally, the lack of structural rigidity in standard alkyl-substituted azobenzenes can lead to unpredictable phase behavior in polymer matrices, limiting their compatibility with advanced composite materials used in modern optoelectronics.
The Novel Approach
The innovative strategy outlined in the patent data overcomes these historical barriers by strategically incorporating a fluorine-terminated chiral carbon moiety into the molecular architecture of the azobenzene core. This structural modification fundamentally alters the electronic environment surrounding the azo linkage, thereby amplifying the dipole moment changes associated with photo-isomerization and yielding a much more distinct separation in absorption spectra. 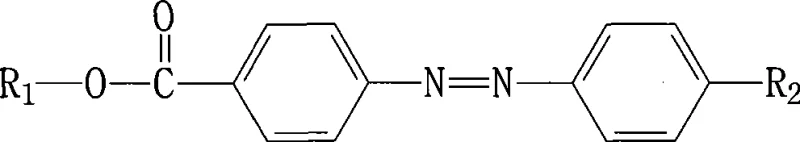 By leveraging the unique electronegativity and steric properties of the perfluorinated chain, the novel compounds achieve a state of enhanced kinetic stability, effectively locking the desired isomeric form in place under ambient storage conditions. This approach eliminates the need for cryogenic storage or specialized handling protocols, significantly simplifying logistics for supply chain managers responsible for hazardous material transport. The versatility of the synthetic design allows for the attachment of various functional groups at the R2 position, enabling customization for specific refractive index or solubility requirements without sacrificing the core photochromic efficiency. Consequently, this method provides a scalable and economically viable route for cost reduction in electronic chemical manufacturing, delivering high-purity intermediates ready for immediate integration into sophisticated optical device assemblies.
By leveraging the unique electronegativity and steric properties of the perfluorinated chain, the novel compounds achieve a state of enhanced kinetic stability, effectively locking the desired isomeric form in place under ambient storage conditions. This approach eliminates the need for cryogenic storage or specialized handling protocols, significantly simplifying logistics for supply chain managers responsible for hazardous material transport. The versatility of the synthetic design allows for the attachment of various functional groups at the R2 position, enabling customization for specific refractive index or solubility requirements without sacrificing the core photochromic efficiency. Consequently, this method provides a scalable and economically viable route for cost reduction in electronic chemical manufacturing, delivering high-purity intermediates ready for immediate integration into sophisticated optical device assemblies.
Mechanistic Insights into Fluorine-Enhanced Photo-Isomerization
The fundamental mechanism driving the superior performance of these materials revolves around the reversible photo-induced trans-cis isomerization of the central diazene (-N=N-) bridge, a process that is critically modulated by the attached fluorinated side chains. Upon exposure to ultraviolet radiation at wavelengths around 365nm, the molecule absorbs photon energy to overcome the rotational barrier of the nitrogen double bond, transitioning from the thermodynamically stable trans configuration to the higher energy cis form.  The presence of the bulky, electron-withdrawing perfluoroalkyl group exerts a profound influence on the potential energy surface of this transition, effectively widening the energy gap between the ground and excited states which results in the observed large shift in absorption maxima. Unlike simple alkyl chains that may allow for free rotation and rapid thermal relaxation, the rigid fluorinated tail imposes steric constraints that kinetically trap the cis-isomer, preventing premature reversion and ensuring the retention of the optical state until triggered by visible light greater than 440nm. This bistable behavior is essential for non-volatile optical data storage applications where information must be retained without continuous power input. Furthermore, the chiral center introduced within the fluorinated segment adds a layer of stereochemical complexity that can be exploited for circularly polarized light manipulation, opening new avenues for 3D display technologies and advanced security printing inks that require high-fidelity pattern reproduction.
The presence of the bulky, electron-withdrawing perfluoroalkyl group exerts a profound influence on the potential energy surface of this transition, effectively widening the energy gap between the ground and excited states which results in the observed large shift in absorption maxima. Unlike simple alkyl chains that may allow for free rotation and rapid thermal relaxation, the rigid fluorinated tail imposes steric constraints that kinetically trap the cis-isomer, preventing premature reversion and ensuring the retention of the optical state until triggered by visible light greater than 440nm. This bistable behavior is essential for non-volatile optical data storage applications where information must be retained without continuous power input. Furthermore, the chiral center introduced within the fluorinated segment adds a layer of stereochemical complexity that can be exploited for circularly polarized light manipulation, opening new avenues for 3D display technologies and advanced security printing inks that require high-fidelity pattern reproduction.
From an impurity control perspective, the reaction mechanism relies on a clean nucleophilic acyl substitution that minimizes the formation of side products commonly associated with radical-based functionalization methods. The use of thionyl chloride for activating the carboxylic acid precursor ensures quantitative conversion to the reactive acyl chloride intermediate, which then reacts selectively with the fluorinated alcohol in the presence of a mild base like triethylamine. This controlled pathway prevents the formation of azo-reduction byproducts or hydrolysis impurities that could otherwise act as quenchers for the photochromic effect, thereby preserving the quantum yield of the isomerization cycle. The high degree of regioselectivity achieved in this synthesis guarantees a consistent impurity profile across different production batches, a critical factor for pharmaceutical and electronic grade intermediates where trace contaminants can disrupt device performance. By understanding these mechanistic nuances, process engineers can optimize reaction parameters such as temperature ramps and stoichiometric ratios to maximize yield while maintaining the stringent purity specifications required for high-end optical applications.
How to Synthesize Fluorinated Azobenzene Derivatives Efficiently
The practical realization of these advanced materials involves a streamlined two-stage synthetic protocol that balances reaction efficiency with operational safety and scalability. The process begins with the activation of the azobenzene carboxylic acid precursor, followed by a coupling reaction with the specialized fluorinated alcohol to install the critical chiral end-group. This methodology avoids the use of exotic reagents or extreme pressure conditions, making it highly suitable for adaptation in standard multipurpose chemical reactors found in modern fine chemical facilities. 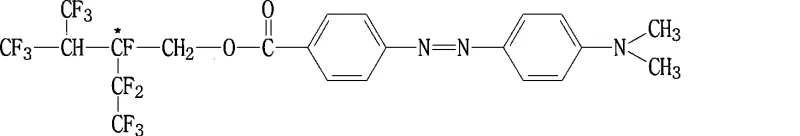 The following guide outlines the standardized operational procedure derived from the patent examples, providing a clear roadmap for technical teams aiming to replicate this high-value synthesis. Adherence to the specified temperature gradients and addition rates is paramount to controlling the exothermic nature of the acyl chloride formation and ensuring the integrity of the sensitive azo linkage throughout the transformation.
The following guide outlines the standardized operational procedure derived from the patent examples, providing a clear roadmap for technical teams aiming to replicate this high-value synthesis. Adherence to the specified temperature gradients and addition rates is paramount to controlling the exothermic nature of the acyl chloride formation and ensuring the integrity of the sensitive azo linkage throughout the transformation.
- Activate the carboxylic acid precursor (4-carboxy-4'-N,N-dimethylazobenzene) using thionyl chloride and DMF catalyst under controlled temperature gradients ranging from 0°C to 90°C.
- Perform nucleophilic substitution by reacting the activated acyl chloride intermediate with dodecafluoroheptanol and triethylamine in DMF solvent at elevated temperatures.
- Isolate the final fluorinated azobenzene product through aqueous workup, pH neutralization, filtration, and recrystallization using chloroform.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this fluorinated azobenzene synthesis route presents a compelling value proposition centered on operational efficiency and risk mitigation. The reliance on commodity chemicals such as thionyl chloride, dimethylformamide, and triethylamine ensures a stable and resilient supply base, shielding manufacturers from the volatility often associated with specialized catalyst markets. The elimination of transition metal catalysts from the process flow removes the necessity for expensive and time-consuming heavy metal scavenging steps, directly translating to substantial cost savings in downstream processing and waste management. Furthermore, the robustness of the final product at room temperature significantly reduces the logistical burden and costs related to cold chain transportation and climate-controlled warehousing, allowing for more flexible distribution networks. The high yield and selectivity reported in the patent examples suggest a process that is inherently waste-minimized, aligning with increasingly stringent environmental regulations and corporate sustainability goals without compromising on output volume.
- Cost Reduction in Manufacturing: The synthetic pathway leverages widely available industrial solvents and reagents, avoiding the premium pricing of rare earth metals or complex organometallic catalysts often required in alternative functionalization strategies. By streamlining the purification process through simple recrystallization rather than column chromatography, the overall production cost per kilogram is drastically lowered, enhancing the margin potential for high-volume commercial orders. The high atom economy of the esterification step ensures that the majority of the input mass is converted into the desired product, minimizing raw material waste and maximizing resource utilization efficiency.
- Enhanced Supply Chain Reliability: The simplicity of the two-step reaction sequence reduces the number of unit operations required, thereby decreasing the probability of equipment failure or process deviations that could lead to batch delays. Since the key fluorinated alcohol intermediate can be sourced from established fluorine chemical suppliers, the risk of raw material shortages is mitigated, ensuring consistent production schedules and reliable delivery timelines for downstream clients. The stability of the intermediate acyl chloride allows for potential semi-batch processing strategies, offering flexibility in production planning to meet fluctuating market demand without the need for extensive inventory holding.
- Scalability and Environmental Compliance: The reaction conditions operate within standard temperature and pressure ranges compatible with existing glass-lined or stainless steel reactor infrastructure, facilitating seamless scale-up from pilot plant to multi-ton commercial production without the need for capital-intensive equipment upgrades. The absence of halogenated byproducts other than the intended fluorine chain simplifies effluent treatment protocols, reducing the environmental footprint and compliance costs associated with hazardous waste disposal. This green chemistry profile makes the process attractive for manufacturing in regions with strict environmental oversight, securing long-term operational licenses and social license to operate.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and application of this fluorinated photochromic technology. These insights are derived directly from the experimental data and structural analysis provided in the patent documentation, offering clarity for stakeholders evaluating this material for integration into their product lines. Understanding these details is crucial for making informed decisions about material selection and process validation.
Q: What distinguishes this fluorinated azobenzene from conventional photochromic materials?
A: Unlike standard azobenzenes which exhibit minimal spectral differences between cis and trans states, this fluorine-terminated variant demonstrates significantly enhanced UV-Vis absorption shifts and superior room temperature storage stability.
Q: Is the synthesis process scalable for industrial production?
A: Yes, the methodology utilizes standard organic synthesis techniques such as acyl chloride formation and esterification, employing common reagents like thionyl chloride and DMF, which facilitates straightforward scale-up from laboratory to commercial manufacturing.
Q: What are the primary application fields for these compounds?
A: These materials are specifically engineered for high-tech sectors including optical data storage, liquid crystal display technologies, nonlinear optical devices, and photomolecular switches due to their reversible isomerization properties.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluorinated Azobenzene Photochromic Compound Supplier
As the global demand for advanced optical materials continues to surge, partnering with an experienced chemical manufacturing expert is essential for navigating the complexities of bringing novel photochromic compounds to market. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to industrial reality is smooth and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of fluorinated azobenzene meets the exacting standards required for high-performance display and data storage applications. We understand the critical nature of supply continuity in the electronics sector and have optimized our logistics to ensure timely delivery of these specialized intermediates.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific volume and quality requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to this fluorine-enhanced platform compared to your current material sources. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to validate the performance characteristics of these compounds against your internal benchmarks with confidence and precision.
