Advanced Biocatalytic Resolution of Ethyl 2-Bromoisovalerate for Scalable Agrochemical Manufacturing
The pharmaceutical and agrochemical industries are constantly seeking more efficient pathways to produce chiral intermediates, particularly for pyrethroid insecticides like flumethrin. A significant breakthrough in this domain is documented in patent CN110066835B, which discloses a highly stereoselective application of a specific lipase for the resolution of racemic ethyl 2-bromoisovalerate. This biological catalysis method represents a paradigm shift from traditional chemical synthesis, offering a direct route to obtain the critical (R)-enantiomer with exceptional purity. By leveraging recombinant DNA technology, the inventors have engineered a robust biocatalyst that operates under mild conditions, effectively bypassing the need for expensive chiral pool starting materials. For R&D directors and procurement specialists, this technology signals a viable pathway to drastically lower production costs while adhering to stringent environmental standards. The ability to achieve an enantiomeric excess value of greater than 99 percent underscores the precision of this enzymatic system, making it a cornerstone for modern green chemistry initiatives in the agrochemical sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of flumethrin and related pyrethroids has relied heavily on the use of D-valine as a chiral starting material to introduce the necessary stereochemistry. While this chemical approach can directly yield chiral products, it suffers from significant economic and logistical drawbacks, primarily due to the exorbitant cost of D-valine compared to its racemic counterparts. Furthermore, traditional chemical resolution methods often involve harsh reaction conditions, the use of toxic heavy metal catalysts, and complex purification steps that generate substantial hazardous waste. These factors not only inflate the overall manufacturing budget but also complicate regulatory compliance regarding environmental discharge and worker safety. The reliance on scarce natural chiral sources creates supply chain vulnerabilities, where price volatility of raw materials can severely impact the profitability of the final agrochemical product. Consequently, there is an urgent industrial demand for alternative synthetic routes that decouple production from expensive chiral precursors while maintaining high optical purity.
The Novel Approach
The innovative strategy outlined in the patent data utilizes a recombinant lipase derived from Aspergillus oryzae WZ007 to catalyze the kinetic resolution of inexpensive racemic ethyl 2-bromoisovalerate. This biocatalytic method elegantly solves the cost issue by starting with cheap, readily available racemic esters rather than costly chiral acids. The engineered enzyme exhibits remarkable substrate specificity, selectively hydrolyzing one enantiomer while leaving the desired (R)-ethyl 2-bromoisovalerate intact or forming it with high fidelity depending on the specific reaction design. Unlike chemical catalysts, this biological system operates in aqueous buffers at moderate temperatures, eliminating the need for extreme heat or pressure and significantly reducing energy consumption. The process achieves a conversion rate of 50.8 percent with a mass yield reaching 96.4 percent, demonstrating that high efficiency does not require compromising on sustainability. This approach not only simplifies the synthetic route but also aligns perfectly with the principles of green chemistry, offering a cleaner and more economically attractive alternative for large-scale manufacturing.
Mechanistic Insights into Lipase-Catalyzed Enantioselective Hydrolysis
The core of this technological advancement lies in the unique structural properties of the lipase encoded by SEQ ID NO.2, which facilitates a highly precise stereoselective hydrolysis mechanism. The enzyme's active site is geometrically configured to accommodate the specific spatial arrangement of the (S)-enantiomer of the substrate, thereby catalyzing its hydrolysis while sterically hindering the reaction of the (R)-enantiomer, or vice versa depending on the specific kinetic resolution strategy employed. This molecular recognition is driven by a network of hydrogen bonds and hydrophobic interactions within the enzyme-substrate complex, ensuring that only the target isomer undergoes transformation. The genetic engineering of the host E. coli Rosetta strain allows for the intracellular overexpression of this lipase, resulting in a high concentration of active biocatalyst that drives the reaction equilibrium towards the desired product. Understanding this mechanism is crucial for process optimization, as it highlights the importance of maintaining the enzyme's tertiary structure through controlled pH and temperature conditions to preserve its chiral discrimination capabilities.
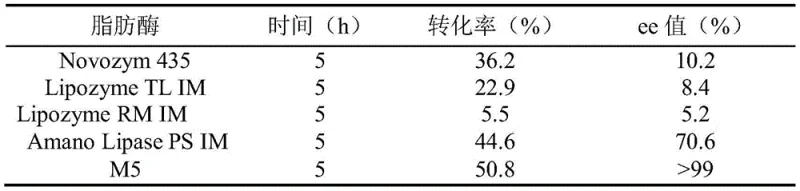
Furthermore, the control of impurities in this biocatalytic process is inherently superior to chemical methods due to the high regioselectivity and chemoselectivity of the lipase. In traditional chemical synthesis, side reactions such as elimination or non-specific hydrolysis can lead to a complex mixture of by-products that are difficult and costly to separate. However, the enzymatic pathway minimizes these side reactions, as the biocatalyst is evolved to perform a single specific transformation with high fidelity. The result is a reaction profile that yields a much cleaner crude product, significantly reducing the burden on downstream purification units. This purity advantage is critical for agrochemical intermediates, where trace impurities can affect the efficacy and safety of the final pesticide formulation. The data indicates that the process consistently delivers an enantiomeric excess value exceeding 99 percent, confirming that the mechanistic design effectively suppresses the formation of the unwanted optical isomer.
How to Synthesize (R)-Ethyl 2-Bromoisovalerate Efficiently
To implement this advanced biocatalytic route, manufacturers must follow a standardized protocol that ensures the stability and activity of the recombinant lipase throughout the production cycle. The process begins with the fermentation of the engineered E. coli strain to produce the crude enzyme powder, followed by the careful preparation of the reaction medium to maintain optimal pH levels. It is essential to control the substrate concentration and agitation speed to prevent mass transfer limitations that could hinder the enzyme's access to the hydrophobic ester substrate. The detailed standardized synthesis steps see the guide below.
- Construct recombinant E. coli expressing the specific lipase gene from Aspergillus oryzae WZ007 and cultivate to obtain crude enzyme powder.
- Perform enantioselective hydrolysis of racemic ethyl 2-bromoisovalerate in pH 7.0 phosphate buffer at 35°C with vigorous stirring.
- Acidify the reaction mixture, extract with ethyl acetate, and purify via washing and rotary evaporation to isolate the target (R)-enantiomer.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this lipase-mediated resolution process offers transformative benefits that extend far beyond simple technical feasibility. The primary advantage lies in the drastic reduction of raw material costs, as the process substitutes expensive chiral starting materials with affordable racemic esters that are commodity chemicals. This shift fundamentally alters the cost structure of the intermediate, providing a significant buffer against market fluctuations in chiral pool prices. Additionally, the mild reaction conditions reduce the requirement for specialized high-pressure or high-temperature equipment, lowering capital expenditure and maintenance costs for production facilities. The simplicity of the downstream processing, involving standard extraction and distillation techniques, further streamlines operations and reduces the time required to bring the product to market. These factors collectively enhance the overall economic viability of the supply chain, making the final agrochemical product more competitive in the global marketplace.
- Cost Reduction in Manufacturing: The elimination of expensive chiral precursors like D-valine represents a major cost saving opportunity, as the process relies on cheap racemic feedstocks that are widely available from bulk chemical suppliers. By removing the need for complex chemical resolution agents or precious metal catalysts, the operational expenditure is significantly lowered, allowing for better margin management. The high mass yield reported in the patent data suggests that material utilization is optimized, minimizing waste and maximizing the output per batch. This efficiency translates directly into a lower cost of goods sold, enabling companies to offer more competitive pricing to their customers while maintaining healthy profit margins.
- Enhanced Supply Chain Reliability: Sourcing high-purity chiral intermediates can often be a bottleneck due to limited suppliers and long lead times, but this biocatalytic method mitigates such risks by utilizing a fermentable enzyme system. Since the biocatalyst is produced in-house or sourced from reliable enzyme manufacturers, the dependency on external suppliers for critical chiral building blocks is greatly reduced. The robustness of the recombinant E. coli strain ensures a consistent and scalable supply of the catalyst, preventing production delays caused by raw material shortages. This self-sufficiency in catalyst production strengthens the supply chain resilience, ensuring continuous operation even during periods of market instability or logistical disruptions.
- Scalability and Environmental Compliance: The aqueous nature of the reaction medium and the absence of toxic heavy metals make this process inherently safer and easier to scale up to industrial volumes without encountering significant environmental hurdles. Regulatory compliance is simplified as the waste streams are less hazardous, reducing the costs associated with waste treatment and disposal. The energy efficiency of running reactions at moderate temperatures further contributes to a lower carbon footprint, aligning with corporate sustainability goals and increasingly strict environmental regulations. This eco-friendly profile not only avoids potential fines but also enhances the brand reputation of the manufacturer as a responsible producer of agrochemical ingredients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this lipase resolution technology, based on the specific data and embodiments provided in the patent documentation. These insights are designed to clarify the operational parameters and strategic benefits for stakeholders evaluating this synthesis route. Understanding these details is essential for making informed decisions about process adoption and integration into existing manufacturing lines.
Q: Why is this lipase resolution method superior to traditional chemical synthesis for flumethrin intermediates?
A: Traditional methods often rely on expensive chiral starting materials like D-valine. This biocatalytic approach utilizes cheap racemic esters and achieves >99% ee with high mass yield, significantly reducing raw material costs while eliminating heavy metal catalysts.
Q: What are the critical process parameters for maintaining high enantioselectivity?
A: Temperature control is paramount; the patent specifies 35°C as optimal. Deviations above or below this temperature reduce both conversion rates and enantiomeric excess. Additionally, maintaining pH 7.0 and specific agitation speeds ensures consistent enzyme activity.
Q: Can this enzymatic process be scaled for industrial production?
A: Yes, the use of recombinant E. coli allows for high-density fermentation and large-scale enzyme production. The mild reaction conditions and simple downstream processing make it highly suitable for commercial scale-up compared to complex chemical resolutions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (R)-Ethyl 2-Bromoisovalerate Supplier
At NINGBO INNO PHARMCHEM, we recognize the immense potential of this biocatalytic technology to revolutionize the production of key agrochemical intermediates like (R)-ethyl 2-bromoisovalerate. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch meets the highest international standards for enantiomeric excess and chemical purity. We are committed to delivering high-quality intermediates that empower your downstream synthesis of flumethrin and other valuable pyrethroids.
We invite you to collaborate with us to leverage this cutting-edge resolution technology for your specific production needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your current supply chain structure. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our expertise can optimize your manufacturing process and drive significant value for your organization.
