Revolutionizing (S)-Nornicotine Production via Engineered Imine Reductase and Immobilization Technology
The pharmaceutical and fine chemical industries are currently witnessing a paradigm shift towards greener, more efficient biocatalytic processes, particularly for the synthesis of chiral amines which serve as critical building blocks for active pharmaceutical ingredients (APIs). A groundbreaking development in this sector is detailed in Chinese Patent CN114317472B, titled "High stereoselectivity imine reductase and preparation method and application thereof." This intellectual property discloses a sophisticated enzymatic route for the asymmetric reduction of myosmine to produce (S)-nornicotine, a pivotal intermediate for smoking cessation therapies and potential treatments for Parkinson's disease. Unlike traditional chemical synthesis which often relies on harsh reducing agents and complex resolution steps, this patent leverages site-directed mutagenesis of wild-type Aeromonas imine reductase to create a biocatalyst with exceptional stereocontrol. The technology addresses the longstanding industry challenge of producing high-optical-purity chiral amines economically, offering a robust alternative that aligns with modern sustainability goals while ensuring the rigorous quality standards required by global regulatory bodies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of (S)-nornicotine and its derivative (S)-nicotine has been plagued by significant inefficiencies inherent to chemical synthesis and plant extraction. Traditional chemical routes typically involve the reduction of myosmine using stoichiometric amounts of chemical reducing agents, which invariably yield a racemic mixture containing both (R) and (S) enantiomers. Separating these enantiomers requires the use of chiral resolving agents, such as dibenzoyl tartaric acid, followed by multiple recrystallization steps to achieve acceptable optical purity. This resolution process is not only material-intensive, consuming large quantities of organic solvents and auxiliary chemicals, but it also inherently caps the theoretical yield at 50% unless dynamic kinetic resolution is employed, which adds further complexity and cost. Furthermore, the presence of residual heavy metals from catalysts and solvent impurities necessitates extensive downstream purification, creating substantial environmental waste and increasing the overall carbon footprint of the manufacturing process. These factors collectively result in a high cost of goods sold (COGS) and supply chain vulnerabilities associated with the sourcing of specialized chiral auxiliaries.
The Novel Approach
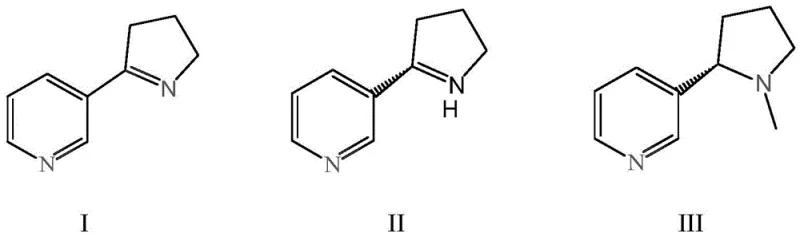
In stark contrast, the biocatalytic methodology outlined in the patent introduces a streamlined, atom-economical pathway that bypasses the need for racemic synthesis and subsequent resolution. By utilizing an engineered imine reductase (IRED) derived from Aeromonas sp., the process directly converts the prochiral substrate myosmine into the desired (S)-nornicotine with exquisite selectivity. The core innovation lies in the specific mutation of the enzyme's active site—altering amino acids at positions 57, 122, 176, and 241—which dramatically enhances its stereoselectivity towards the S-enantiomer. This biological approach operates under mild aqueous conditions, typically at temperatures between 25°C and 35°C and near-neutral pH, eliminating the safety hazards associated with high-pressure hydrogenation or pyrophoric reagents. Moreover, the integration of a cofactor regeneration system using glucose dehydrogenase (GDH) ensures that the expensive NADPH cofactor is continuously recycled in situ, driving the reaction to completion with minimal auxiliary input. This holistic design not only simplifies the operational workflow but also fundamentally alters the economic model of production by enabling higher yields and purities in a single synthetic step.
Mechanistic Insights into Engineered Imine Reductase Catalysis
The catalytic prowess of this system is rooted in the precise molecular engineering of the imine reductase enzyme. The patent describes a site-directed mutagenesis strategy where the wild-type sequence is modified to create Variant II (SEQ ID No.1), which exhibits superior binding affinity and transition state stabilization for the myosmine substrate. Mechanistically, the imine reductase facilitates the transfer of a hydride ion from the reduced nicotinamide adenine dinucleotide phosphate (NADPH) to the C=N double bond of the myosmine imine group. The chirality of the product is dictated by the spatial arrangement of the enzyme's active site pocket, which sterically hinders the approach of the hydride from the Re-face, thereby forcing addition from the Si-face to exclusively generate the (S)-configuration. This enzymatic precision is further supported by a coupled cofactor regeneration cycle. As the IRED oxidizes NADPH to NADP+, the co-immobilized glucose dehydrogenase simultaneously oxidizes glucose to gluconic acid, reducing NADP+ back to NADPH. This closed-loop system ensures that only catalytic amounts of the expensive cofactor are required, making the process economically viable for large-scale applications.
Beyond the primary catalytic cycle, the patent emphasizes the critical role of enzyme immobilization in maintaining long-term stability and operational efficiency. The crude enzyme lysates are treated with flocculants like polyethyleneimine and ammonium sulfate to remove cellular debris and stabilize the protein structure before being covalently bound to activated resin carriers, such as amino resin LX-1000NH or epoxy resins. This immobilization serves a dual purpose: it protects the enzyme from denaturation in the reaction medium and creates a heterogeneous catalyst that can be easily separated from the product stream via simple filtration. The data indicates that the immobilized enzyme retains over 90% of its initial activity even after 20 consecutive reaction cycles, demonstrating remarkable robustness. This stability is crucial for preventing the leaching of protein impurities into the final product, thereby simplifying the purification protocol and ensuring that the final (S)-nornicotine meets the stringent purity specifications required for pharmaceutical intermediates without the need for complex chromatographic separations.
How to Synthesize (S)-Nornicotine Efficiently
The practical implementation of this biocatalytic route involves a carefully orchestrated sequence of genetic engineering, fermentation, and process optimization steps designed to maximize space-time yield. The process begins with the construction of recombinant E. coli strains expressing both the mutated imine reductase and glucose dehydrogenase, followed by high-density fermentation to produce sufficient biomass. The subsequent downstream processing involves cell lysis, clarification, and the critical immobilization step onto selected resin carriers to create the final biocatalyst. For R&D teams looking to replicate or adapt this technology, understanding the interplay between substrate loading, buffer composition, and enzyme loading is essential to achieving the reported conversion rates of >99%. The detailed standardized synthesis steps, including specific buffer molarities, agitation speeds, and incubation times required to achieve industrial-grade results, are outlined in the comprehensive guide below.
- Construct recombinant expression vectors containing the mutated imine reductase gene (SEQ ID No.2) and glucose dehydrogenase gene, then transform into E. coli host cells for high-level expression.
- Prepare crude enzyme lysates via ultrasonic disruption, followed by flocculation treatment using ammonium sulfate and polyethyleneimine to clarify the enzyme solution.
- Immobilize the clarified enzymes onto activated amino or epoxy resin carriers (e.g., LX-1000NH), wash thoroughly, and utilize the immobilized biocatalysts in a buffered reaction system with myosmine substrate and glucose for cofactor regeneration.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this immobilized enzyme technology represents a strategic opportunity to optimize cost structures and mitigate supply risks associated with chiral intermediate sourcing. The shift from chemical resolution to biocatalysis fundamentally changes the cost drivers of production, moving away from expensive chiral resolving agents and vast volumes of organic solvents towards renewable biological catalysts and aqueous systems. This transition not only aligns with corporate sustainability mandates but also offers tangible economic benefits through improved material efficiency and reduced waste disposal costs. The ability to operate at high substrate concentrations further amplifies these advantages by increasing the volumetric productivity of existing reactor infrastructure, effectively lowering the capital expenditure required per unit of output.
- Cost Reduction in Manufacturing: The elimination of stoichiometric chiral resolving agents and the associated mother liquor losses inherent in resolution processes leads to a drastic improvement in overall yield. By utilizing an immobilized enzyme system that can be reused for multiple batches, the effective cost of the biocatalyst per kilogram of product is significantly diminished compared to single-use free enzymes or chemical catalysts. Furthermore, the simplified downstream processing, which avoids the need for extensive solvent exchanges and heavy metal scavenging, reduces utility consumption and labor hours, contributing to substantial overall cost savings in API manufacturing.
- Enhanced Supply Chain Reliability: Reliance on complex chemical supply chains for specialized resolving agents can introduce volatility and lead time uncertainties. In contrast, the biocatalytic components—glucose, buffers, and recombinant enzymes—are derived from abundant, commodity-grade raw materials, ensuring a stable and predictable supply base. The robustness of the immobilized enzyme, capable of withstanding repeated use without significant loss of activity, reduces the frequency of catalyst replenishment orders, thereby streamlining inventory management and reducing the risk of production stoppages due to material shortages.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to function effectively from liter-scale laboratory reactors up to 300-liter pilot scales with consistent performance. The aqueous nature of the reaction medium minimizes the generation of hazardous volatile organic compound (VOC) emissions, facilitating easier compliance with increasingly stringent environmental regulations. Additionally, the high conversion efficiency and specificity reduce the formation of by-products, simplifying wastewater treatment requirements and lowering the environmental burden associated with effluent disposal, which is a critical factor for maintaining operational licenses in regulated jurisdictions.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating the feasibility of integrating this technology into their existing portfolios, we have compiled a set of frequently asked questions based on the specific technical disclosures within the patent documentation. These queries address common concerns regarding enzyme stability, substrate tolerance, and product quality, providing clarity on how this biocatalytic platform compares to incumbent technologies. Understanding these nuances is vital for assessing the potential return on investment and the technical readiness level of the process for commercial deployment.
Q: What is the optical purity achievable with this new imine reductase process?
A: According to patent CN114317472B, the engineered imine reductase achieves an enantiomeric excess (ee) value of greater than 99.5% for (S)-nornicotine, significantly surpassing traditional chemical resolution methods which often struggle to exceed 99% without multiple recrystallizations.
Q: How does enzyme immobilization impact production costs?
A: Immobilization allows the biocatalyst to be recovered and reused for at least 20 cycles while retaining over 90% of its initial activity. This drastically reduces the cost per kilogram of product by eliminating the need for fresh enzyme in every batch and simplifying downstream purification by avoiding free protein contamination.
Q: Can this process handle high substrate concentrations suitable for industrial scale?
A: Yes, the immobilized enzyme system demonstrates excellent tolerance to high substrate loading, efficiently converting myosmine concentrations up to 300g/L with conversion rates exceeding 99%, making it highly viable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-Nornicotine Supplier
As the global demand for high-purity chiral intermediates continues to surge, particularly in the sectors of smoking cessation aids and neurological therapeutics, partnering with a technically proficient CDMO is essential for securing a competitive edge. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced fermentation capabilities, allowing us to meet stringent purity specifications and deliver consistent quality batch after batch. We understand the critical importance of supply continuity and are committed to implementing robust process controls that guarantee the availability of high-value intermediates like (S)-nornicotine.
We invite forward-thinking pharmaceutical companies and chemical manufacturers to collaborate with us to leverage this cutting-edge biocatalytic technology. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that will drive innovation and profitability in your supply chain.
