Revolutionizing Sitagliptin Intermediate Production with Advanced AcATA Mutant Technology
The pharmaceutical industry is constantly seeking more efficient and sustainable pathways for the production of critical drug intermediates, and the technology disclosed in patent CN111549008B represents a significant leap forward in the enzymatic synthesis of sitagliptin intermediates. This patent details a novel amine transaminase AcATA mutant that has been specifically engineered through site-directed mutagenesis to overcome the limitations of wild-type enzymes, offering a robust solution for the asymmetric synthesis of chiral amines. By targeting the 122nd amino acid position and mutating methionine to histidine, valine, or phenylalanine, the inventors have created a biocatalyst that demonstrates exceptional catalytic efficiency and stability under industrial conditions. For R&D directors and procurement specialists, this innovation signals a shift away from resource-intensive chemical methods towards a greener, more cost-effective biocatalytic platform that aligns with modern regulatory and environmental standards. The ability to achieve high conversion rates with reduced reaction times directly translates to improved throughput and lower operational expenditures for manufacturers scaling up production of this key diabetes medication precursor.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional chemical synthesis routes for sitagliptin intermediates have long been plagued by inherent inefficiencies and environmental concerns that pose significant challenges for large-scale manufacturing. Conventional methods often rely on the use of expensive transition metal catalysts, such as ruthenium or platinum complexes, which not only drive up raw material costs but also introduce the risk of heavy metal contamination in the final product. These chemical processes typically require harsh reaction conditions, including high pressures and extreme temperatures, which demand specialized equipment and rigorous safety protocols, thereby increasing capital investment and operational complexity. Furthermore, the stereo-selectivity of chemical hydrogenation can be inconsistent, often necessitating multiple recrystallization steps to achieve the required enantiomeric purity, which inevitably leads to substantial yield losses and increased waste generation. The reliance on toxic reagents and the generation of hazardous by-products also complicate waste treatment procedures, making it difficult for manufacturers to comply with increasingly stringent environmental regulations without incurring significant additional costs.
The Novel Approach
In stark contrast to these legacy methods, the novel biocatalytic approach utilizing the AcATA mutant offers a streamlined and highly selective pathway that addresses the core pain points of traditional synthesis. By leveraging the power of protein engineering, this method employs a genetically modified enzyme that operates under mild aqueous conditions, eliminating the need for high-pressure reactors and toxic organic solvents commonly associated with chemical catalysis. The mutant enzyme demonstrates a remarkable affinity for the specific ketone substrate, facilitating a direct transamination reaction that constructs the chiral center with exceptional precision and minimal by-product formation. This biological specificity ensures that the reaction proceeds with high stereo-selectivity, drastically reducing the need for downstream purification steps and maximizing the overall yield of the desired intermediate. For supply chain managers, this translates to a more predictable and reliable production process that is less susceptible to the fluctuations in raw material availability and pricing that often affect petrochemical-derived reagents, ensuring a more stable supply of high-quality intermediates.
Mechanistic Insights into AcATA-Catalyzed Transamination
The core of this technological breakthrough lies in the precise structural modification of the amine transaminase enzyme, specifically the single-point mutation at the 122nd position of the amino acid sequence. Through advanced molecular docking and homology modeling, researchers identified that the native methionine residue at this position created steric hindrance or suboptimal electronic interactions that limited the enzyme's catalytic turnover. By substituting this residue with histidine, valine, or phenylalanine, the spatial configuration of the active site was optimized to better accommodate the bulky sitagliptin precursor ketone, thereby enhancing the binding affinity and catalytic efficiency. The optimal mutant, M122H, exhibits an enzyme activity of up to 610 U/g, which is a staggering 5.5-fold increase compared to the wild-type enzyme, allowing for rapid conversion of the substrate even at high concentrations. This enhanced activity is crucial for industrial applications where high substrate loading is necessary to minimize reactor volume and maximize productivity, demonstrating how rational protein design can unlock significant performance gains in biocatalytic processes.
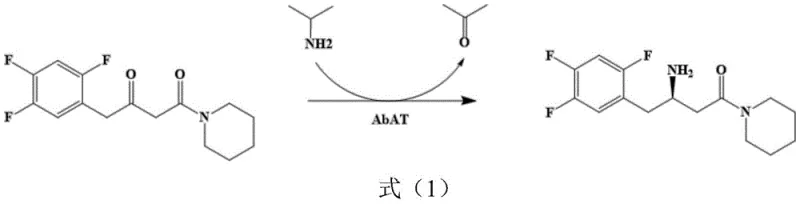
Furthermore, the mechanism of impurity control in this enzymatic system is inherently superior to chemical alternatives due to the high specificity of the biocatalyst. Unlike chemical catalysts that may promote side reactions such as over-reduction or non-specific bond cleavage, the AcATA mutant selectively targets the carbonyl group of the ketone substrate for amination, leaving other functional groups intact. This selectivity is further supported by the use of isopropylamine as an amino donor, which drives the equilibrium towards product formation while generating acetone as a benign by-product that can be easily removed. The reaction conditions, maintained at a moderate temperature of 45-50°C and a pH of 8.0-9.0, ensure that the enzyme remains stable throughout the process while minimizing the degradation of sensitive intermediates. For quality control teams, this means a cleaner reaction profile with fewer unknown impurities, simplifying the analytical validation process and ensuring that the final intermediate meets the strict purity specifications required for pharmaceutical grade materials without extensive chromatographic purification.
How to Synthesize Sitagliptin Intermediate Efficiently
The implementation of this synthesis route involves a series of well-defined bioprocessing steps that leverage the high activity of the AcATA mutant to ensure consistent and scalable production outcomes. The process begins with the cultivation of recombinant Escherichia coli expressing the mutant enzyme, followed by induction and harvesting of the wet cells which serve as the whole-cell biocatalyst. These cells are then suspended in a buffered reaction system containing the ketone substrate, isopropylamine, and the essential PLP coenzyme, where the biotransformation occurs under controlled agitation and temperature. Detailed standardized synthesis steps are provided below to guide technical teams in replicating these high-efficiency results. This structured approach ensures that the critical parameters influencing enzyme activity and substrate conversion are tightly controlled, resulting in a robust process capable of delivering high-purity intermediates suitable for subsequent API synthesis steps.
- Cultivate recombinant E. coli expressing the AcATA mutant (e.g., M122H) in LB medium with ampicillin, induce with IPTG at 28°C, and harvest wet cells.
- Prepare the reaction system with substrate (50g/L), isopropylamine (80g/L), PLP coenzyme, and buffer at pH 8.5, maintaining temperature at 45-50°C.
- After 24 hours, adjust pH to 1.5, filter cells, extract with dichloromethane, and purify to obtain the intermediate with >99% purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this enzymatic technology offers profound advantages that extend beyond mere technical performance, directly impacting the bottom line and supply chain resilience for pharmaceutical manufacturers. The elimination of expensive noble metal catalysts and the reduction in solvent usage contribute to a significant decrease in raw material costs, while the milder reaction conditions lower energy consumption and equipment maintenance requirements. Additionally, the high conversion rate and selectivity of the AcATA mutant minimize waste generation and simplify downstream processing, leading to substantial cost savings in waste treatment and purification operations. For procurement managers, this translates into a more cost-efficient supply chain that is less vulnerable to the volatility of precious metal markets and regulatory changes regarding hazardous chemical usage, ensuring long-term economic viability for the production of sitagliptin intermediates.
- Cost Reduction in Manufacturing: The transition to this biocatalytic route fundamentally alters the cost structure of sitagliptin intermediate production by removing the dependency on high-cost transition metal catalysts and reducing the number of synthetic steps required. The high enzyme activity allows for lower catalyst loading and shorter reaction times, which increases reactor throughput and reduces utility costs associated with heating and cooling. Furthermore, the simplified work-up procedure, which avoids complex extraction and recrystallization cycles needed to remove metal residues, significantly lowers labor and material costs associated with downstream processing. These cumulative efficiencies result in a leaner manufacturing process that delivers substantial cost savings without compromising on the quality or purity of the final product, making it an attractive option for cost-sensitive generic drug production.
- Enhanced Supply Chain Reliability: Implementing this enzymatic process enhances supply chain reliability by diversifying the source of catalytic activity from finite mineral resources to renewable biological systems that can be produced consistently through fermentation. The stability of the engineered enzyme under process conditions ensures batch-to-batch consistency, reducing the risk of production delays caused by catalyst deactivation or variability in raw material quality. Moreover, the use of readily available and inexpensive amino donors like isopropamine ensures that the supply of key reagents remains stable and unaffected by geopolitical tensions that often impact the availability of specialized chemical catalysts. This reliability is critical for maintaining continuous production schedules and meeting the demanding delivery timelines of global pharmaceutical clients who require uninterrupted supply of critical intermediates.
- Scalability and Environmental Compliance: The scalability of this biocatalytic process is demonstrated by its ability to maintain high conversion rates even at elevated substrate concentrations, making it suitable for transfer from laboratory scale to multi-ton commercial production facilities. The aqueous nature of the reaction system and the absence of toxic heavy metals align perfectly with green chemistry principles, facilitating easier compliance with environmental regulations and reducing the burden of hazardous waste disposal. This environmental compatibility not only mitigates regulatory risks but also enhances the corporate sustainability profile of manufacturers, appealing to stakeholders who prioritize eco-friendly production practices. The combination of scalability and environmental stewardship ensures that this technology can support growing market demand for sitagliptin while adhering to the strictest global standards for pharmaceutical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this AcATA mutant technology, providing clarity on its operational benefits and compatibility with existing manufacturing infrastructure. These insights are derived directly from the patent data and are intended to assist decision-makers in evaluating the feasibility of adopting this advanced biocatalytic route for their specific production needs. Understanding these details is essential for aligning technical capabilities with strategic procurement goals and ensuring a smooth transition to this next-generation synthesis method. We encourage technical teams to review these answers thoroughly to gain a comprehensive understanding of how this innovation can be integrated into their current workflows to maximize efficiency and product quality.
Q: What is the primary advantage of the AcATA mutant over wild-type enzymes?
A: The AcATA mutant (specifically M122H) exhibits enzyme activity up to 610 U/g, which is 5.5 times higher than the wild type, enabling a 90% conversion rate in just 24 hours compared to 50% in 48 hours for the wild type.
Q: Does this enzymatic process require expensive transition metal catalysts?
A: No, this biocatalytic route eliminates the need for expensive ruthenium or platinum catalysts used in traditional chemical hydrogenation, significantly reducing raw material costs and heavy metal contamination risks.
Q: What is the optical purity achieved with this method?
A: The process consistently achieves an enantiomeric excess (ee) value greater than 99%, meeting the stringent stereochemical requirements for active pharmaceutical ingredient (API) synthesis without extensive recrystallization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sitagliptin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this enzymatic technology and are fully equipped to leverage it for the commercial production of high-purity sitagliptin intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the AcATA mutant are realized in practical, large-scale manufacturing environments. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the exacting standards required by global regulatory agencies, providing our partners with the confidence they need to integrate our intermediates into their API synthesis pipelines. Our commitment to technical excellence and quality assurance makes us an ideal partner for pharmaceutical companies seeking to optimize their supply chain with cutting-edge biocatalytic solutions.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific volume and quality requirements. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the potential economic benefits of switching to this enzymatic process for your specific application. We encourage you to contact us to索取 specific COA data and route feasibility assessments, allowing you to make informed decisions based on concrete performance metrics and validated process data. Partnering with us ensures access to a reliable supply of high-quality intermediates produced using the most advanced and sustainable technologies available in the industry today.
