Scaling Up Ethyl 6-Oxo-8-Chlorooctanoate Production via Advanced Continuous Flow Technology
Introduction to Next-Generation Synthesis Technology
The pharmaceutical industry is constantly seeking more efficient and safer pathways for producing critical intermediates, and the preparation of ethyl 6-oxo-8-chlorooctanoate stands as a prime example of this evolution. As detailed in patent CN116332761A, a groundbreaking methodology has been established that transitions the synthesis from traditional batch processing to a sophisticated continuous flow system using pipeline reactors. This compound serves as an indispensable precursor in the manufacture of alpha-lipoic acid, a potent antioxidant widely utilized in treating metabolic disorders such as diabetes and cardiovascular diseases. The shift to flow chemistry addresses long-standing challenges regarding reaction efficiency, safety hazards associated with gaseous reagents, and environmental sustainability. By leveraging precise control over reaction parameters, this new approach not only maximizes yield but also ensures a consistently high-purity profile essential for downstream pharmaceutical applications.
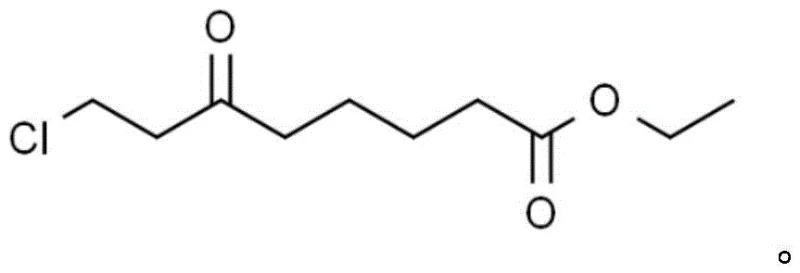
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of ethyl 6-oxo-8-chlorooctanoate relied heavily on stirred tank reactors where ethyl 6-chloro-6-oxohexanoate was reacted with ethylene gas in the presence of aluminum trichloride. This traditional gas-liquid two-phase reaction suffered from inherent inefficiencies due to poor mass transfer between the gaseous ethylene and the liquid reaction mixture. The limited contact area often resulted in incomplete conversions, necessitating longer reaction times and leading to unstable product quality with variable impurity levels. Moreover, handling large volumes of flammable ethylene gas in open or semi-open batch systems posed significant safety risks, including the potential for explosive atmospheres if ventilation failed. These operational bottlenecks made it difficult to achieve the consistency required for modern Good Manufacturing Practice (GMP) standards, often resulting in higher waste generation and increased costs for purification.
The Novel Approach
In stark contrast, the novel approach outlined in the patent utilizes a dual-stage pipeline reactor system that fundamentally transforms the reaction dynamics. By forcing the reactants through a confined tubular environment, the system creates a high-shear mixing zone that dramatically improves the dispersion of ethylene gas into the liquid phase containing the substrate and catalyst. This intensified mixing ensures that every molecule of the starting material has optimal access to the reagent, driving the reaction to near-completion within a precisely defined residence time. The continuous nature of the process allows for real-time monitoring and adjustment of critical parameters such as pressure and temperature, eliminating the thermal gradients common in large batch vessels. Consequently, this method delivers a robust, reproducible process that significantly mitigates safety hazards by containing the reactive gases within a closed, pressurized loop, thereby aligning perfectly with the principles of green chemistry and process safety management.
Mechanistic Insights into AlCl3-Catalyzed Alkylation and Hydrolysis
The core chemical transformation involves a Lewis acid-catalyzed alkylation followed by a hydrolysis step, both optimized for continuous operation. In the first stage, aluminum trichloride (AlCl3) acts as a powerful Lewis acid catalyst, activating the carbonyl group of the ethyl 6-chloro-6-oxohexanoate substrate. When introduced into the first pipeline reactor alongside ethylene, the activated complex undergoes an electrophilic addition where the ethylene double bond attacks the electron-deficient center. The high pressure maintained in this reactor, typically between 0.25 MPa and 0.35 MPa, forces the ethylene into solution, increasing its effective concentration and driving the equilibrium forward. The temperature is carefully regulated between 80°C and 100°C to provide sufficient activation energy for the bond formation without triggering thermal decomposition or unwanted polymerization of the olefin. This precise thermal management is crucial for maintaining the integrity of the ester functionality while extending the carbon chain.
Following the alkylation, the reaction stream enters a hydrolysis phase where water is introduced to quench the aluminum complexes and finalize the product structure. This step occurs in a second pipeline reactor operating at slightly elevated temperatures of 100°C to 130°C and pressures of 0.10 MPa to 0.40 MPa. The introduction of water serves a dual purpose: it hydrolyzes the intermediate aluminum species to release the free ketone product and simultaneously deactivates the catalyst to prevent further side reactions. The short residence time in this second reactor, optimized to around 10 minutes, ensures that the hydrolysis is complete before the mixture exits the system. This rapid quenching mechanism is vital for impurity control, as it prevents the prolonged exposure of the sensitive keto-ester product to acidic conditions that could lead to ester hydrolysis or chlorination side products, thereby securing the high purity levels observed in the final organic phase separation.
How to Synthesize Ethyl 6-Oxo-8-Chlorooctanoate Efficiently
Implementing this continuous flow protocol requires a systematic approach to equipment setup and parameter control to ensure optimal performance. The process begins with the preparation of a homogeneous feed solution where dichloroethane, aluminum trichloride, and the starting ester are mixed at low temperatures to prevent premature reaction. This mixture is then pumped concurrently with ethylene gas into the heated pipeline reactor system. The success of the synthesis hinges on maintaining the specific residence times and pressure differentials described in the patent data. For research and development teams looking to replicate or scale this process, adhering to the precise stoichiometric ratios and thermal profiles is essential to achieving the reported yields exceeding 97%. The detailed standardized synthesis steps for this high-efficiency route are provided in the guide below.
- Prepare a homogeneous mixture of dichloroethane, aluminum trichloride catalyst, and ethyl 6-chloro-6-oxohexanoate at controlled low temperatures.
- Pump the mixed solution and ethylene gas into the first pipeline reactor for alkylation under precise pressure (0.25-0.35 MPa) and temperature (80-100°C) conditions.
- Introduce water to the addition reaction liquid and pass it through a second pipeline reactor for hydrolysis, followed by phase separation to isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this continuous flow methodology represents a strategic opportunity to optimize the cost structure and reliability of the lipoic acid supply chain. The elimination of inefficient batch processing steps directly translates to a reduction in overall manufacturing cycle times, allowing for faster turnover of inventory and improved responsiveness to market demand fluctuations. By adopting a process that inherently minimizes waste and maximizes raw material utilization, manufacturers can offer more competitive pricing structures without compromising on quality. Furthermore, the enhanced safety profile of the pipeline reactor system reduces the regulatory burden and insurance costs associated with handling hazardous gases, contributing to a more sustainable and economically viable production model. These operational efficiencies collectively strengthen the supply chain resilience against disruptions.
- Cost Reduction in Manufacturing: The implementation of continuous flow technology eliminates the need for large, expensive batch reactors and the associated downtime for cleaning and setup between runs. By achieving significantly higher conversion rates and reducing the formation of by-products, the process lowers the consumption of raw materials and solvents per kilogram of finished product. The simplified post-treatment workflow, which involves straightforward phase separation rather than complex distillation or chromatography, further drives down utility and labor costs. This holistic reduction in operational expenditure allows for substantial cost savings that can be passed down the supply chain.
- Enhanced Supply Chain Reliability: The continuous nature of the pipeline reactor system ensures a steady and predictable output of ethyl 6-oxo-8-chlorooctanoate, mitigating the risks of batch-to-batch variability that often plague traditional manufacturing. The robustness of the process against minor fluctuations in feed rates means that production schedules can be maintained with high precision, ensuring on-time delivery for downstream API manufacturers. Additionally, the reduced safety hazards associated with the contained handling of ethylene gas minimize the risk of unplanned shutdowns due to safety incidents, thereby guaranteeing a more consistent and reliable supply of this critical pharmaceutical intermediate.
- Scalability and Environmental Compliance: Scaling up continuous flow processes is inherently more linear and predictable than scaling batch reactions, as it often involves numbering up reactors rather than increasing vessel size, which preserves reaction kinetics. This scalability facilitates rapid capacity expansion to meet growing global demand for lipoic acid derivatives. From an environmental perspective, the process generates significantly less three-waste pollution due to higher atom economy and reduced solvent usage. The closed-loop system prevents the release of volatile organic compounds and hazardous gases, ensuring full compliance with increasingly stringent environmental regulations and supporting corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of ethyl 6-oxo-8-chlorooctanoate. These insights are derived directly from the patented technology and are intended to clarify the advantages of this advanced synthesis route for potential partners and stakeholders. Understanding these details is crucial for evaluating the feasibility of integrating this intermediate into your specific pharmaceutical formulations or supply networks.
Q: What are the primary advantages of using a pipeline reactor over traditional batch methods for this synthesis?
A: The pipeline reactor system significantly enhances mass transfer efficiency between the gas (ethylene) and liquid phases, leading to higher reaction yields (up to 97.3%) and superior product purity (99.3%) compared to conventional batch stirring. Furthermore, it drastically reduces safety risks associated with handling excess ethylene gas in large vessels.
Q: How does this process impact the impurity profile of the final intermediate?
A: By strictly controlling residence time and temperature in the continuous flow system, side reactions such as over-alkylation or polymerization are minimized. This results in a much cleaner crude product with fewer by-products, simplifying downstream purification and reducing solvent consumption.
Q: Is this manufacturing method suitable for large-scale industrial production?
A: Yes, the continuous flow design is inherently scalable. It allows for consistent quality control and steady-state operation, making it ideal for meeting the high-volume demands of the global lipoic acid market while maintaining stringent environmental and safety standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ethyl 6-Oxo-8-Chlorooctanoate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving medications like alpha-lipoic acid. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with unwavering consistency. We are committed to delivering products that meet stringent purity specifications, utilizing our rigorous QC labs to verify every batch against the highest industry standards. Our adoption of advanced continuous flow technologies allows us to maintain a competitive edge in both quality and cost, positioning us as a strategic partner for your long-term growth.
We invite you to engage with our technical procurement team to discuss how our optimized manufacturing processes can benefit your specific projects. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic advantages of sourcing from our facility. We encourage you to reach out today to obtain specific COA data and route feasibility assessments tailored to your needs, ensuring a seamless integration of our high-purity ethyl 6-oxo-8-chlorooctanoate into your supply chain.
