Revolutionizing Chiral Intermediate Production: Advanced Biocatalysis for (R)-3-Cyclohexene-1-Carboxylic Acid
Revolutionizing Chiral Intermediate Production: Advanced Biocatalysis for (R)-3-Cyclohexene-1-Carboxylic Acid
The pharmaceutical and fine chemical industries are constantly seeking more efficient, sustainable, and cost-effective routes for producing chiral building blocks, particularly those critical for blockbuster drugs. A significant technological breakthrough has been documented in patent CN116396950A, which discloses a novel carboxylesterase mutant specifically engineered for the asymmetric synthesis of (R)-3-cyclohexene-1-carboxylic acid ((R)-CHCA). This compound serves as a pivotal chiral precursor for the synthesis of vital active pharmaceutical ingredients (APIs), including the immunosuppressant tacrolimus, the antiviral oseltamivir phosphate, and various antineoplastic agents. The innovation lies not merely in the discovery of a new enzyme, but in the strategic protein engineering that reverses natural stereoselectivity, offering a robust alternative to traditional chemical synthesis and animal-derived biocatalysts. For R&D directors and procurement strategists, this technology represents a paradigm shift towards high-purity, scalable, and environmentally compliant manufacturing processes that can significantly de-risk the supply chain for complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of optically active 3-cyclohexene-1-carboxylic acid has relied heavily on the Diels-Alder reaction or chemical resolution of racemates, both of which present substantial industrial drawbacks. The Diels-Alder approach, while chemically elegant, involves the use of gaseous butadiene, creating significant safety hazards and operational complexities regarding pressure control and gas handling. Furthermore, the separation of products is notoriously difficult, often resulting in low yields and requiring energy-intensive purification steps that inflate the overall cost of goods sold (COGS). Alternatively, chemical resolution methods typically necessitate at least six rounds of recrystallization in acetone to achieve optical purity, a process that is not only atom-uneconomical with a theoretical yield cap of 50% but also generates massive amounts of solvent waste. Previous biocatalytic attempts using animal-derived enzymes like Pig Liver Esterase (PLE) suffered from high costs, batch-to-batch inconsistency, and the inherent risk of viral contamination, rendering them unsuitable for modern GMP-compliant API manufacturing.
The Novel Approach
The technology described in patent CN116396950A overcomes these historical bottlenecks through the deployment of a recombinant carboxylesterase mutant, designated as AcEst1_M2, which exhibits exceptional catalytic efficiency and inverted stereoselectivity. Unlike its wild-type counterparts that predominantly produce the (S)-enantiomer, this engineered variant selectively hydrolyzes the (S)-ester from a racemic mixture, leaving the desired (R)-3-cyclohexene-1-carboxylic acid ester untouched with extremely high enantiomeric excess. This kinetic resolution strategy allows for the isolation of the target (R)-isomer with optical purity exceeding 99% e.e., bypassing the need for multiple recrystallizations. The process operates under mild physiological conditions (30°C, pH 8.0), utilizing water as the primary solvent, which drastically reduces the environmental footprint compared to acetone-heavy chemical resolutions. By shifting from animal-sourced enzymes to a recombinant E. coli expression system, manufacturers gain access to a stable, scalable, and cost-efficient catalyst supply that is free from biological contaminants.
Mechanistic Insights into Carboxylesterase-Mediated Kinetic Resolution
The core of this technological advancement lies in the precise molecular engineering of the enzyme's active site to accommodate the specific steric and electronic properties of the cyclohexene ring. The mutant enzyme incorporates seven specific amino acid substitutions (T248W/L249A/V254K/T258A/V257F/L297F/N329F) relative to the parent AcEst1 sequence. These mutations are not random; they are strategically located to reshape the substrate-binding pocket, effectively reversing the enzyme's chiral recognition logic. By introducing bulky aromatic residues like Tryptophan and Phenylalanine at key positions, the enzyme creates a steric environment that disfavors the binding orientation of the (R)-ester for hydrolysis, while simultaneously enhancing the binding affinity and catalytic turnover for the (S)-ester. This results in a highly efficient kinetic resolution where the (S)-enantiomer is rapidly converted to the acid, while the (R)-enantiomer remains intact as the ester, ready for subsequent isolation. The ability to tune enzyme specificity through rational design and directed evolution demonstrates a sophisticated level of control over biocatalytic processes, ensuring that the reaction proceeds with minimal formation of unwanted byproducts.
Furthermore, the engineered mutant displays a remarkably broad substrate scope, capable of accepting various ester derivatives beyond just the methyl or ethyl esters. As illustrated in the structural analysis of potential substrates, the enzyme maintains high activity and selectivity across different alkyl chain lengths and branching patterns, such as isopropyl and butyl esters. 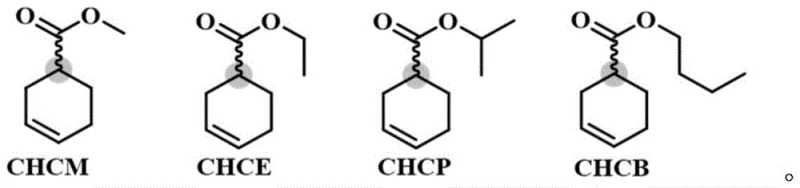 This versatility is crucial for process optimization, as it allows process chemists to select the most cost-effective or easily separable ester starting material without compromising on the enantiomeric purity of the final product. The stability of the enzyme under reaction conditions (20-40°C) further ensures that the catalytic cycle can be sustained over extended periods, facilitating high substrate loading concentrations up to 500g/L. This high tolerance for substrate concentration is a key metric for industrial viability, as it directly correlates to reactor productivity and the reduction of downstream processing volumes, thereby lowering capital expenditure requirements for large-scale manufacturing facilities.
This versatility is crucial for process optimization, as it allows process chemists to select the most cost-effective or easily separable ester starting material without compromising on the enantiomeric purity of the final product. The stability of the enzyme under reaction conditions (20-40°C) further ensures that the catalytic cycle can be sustained over extended periods, facilitating high substrate loading concentrations up to 500g/L. This high tolerance for substrate concentration is a key metric for industrial viability, as it directly correlates to reactor productivity and the reduction of downstream processing volumes, thereby lowering capital expenditure requirements for large-scale manufacturing facilities.
How to Synthesize (R)-3-Cyclohexene-1-Carboxylic Acid Efficiently
The implementation of this biocatalytic route involves a streamlined workflow designed for maximum recovery and purity. The process begins with the preparation of a reaction slurry containing the racemic ester substrate, a phosphate buffer system to maintain pH stability, and the lyophilized recombinant cells expressing the mutant enzyme. The reaction is monitored closely via chiral gas chromatography to track the enrichment of the (R)-enantiomer. Once the desired enantiomeric excess is achieved, the unreacted (R)-ester is separated from the aqueous phase containing the (S)-acid byproduct. The final step involves a standard saponification of the enriched ester to yield the free acid. For a comprehensive understanding of the operational parameters and specific stoichiometric ratios required for optimal performance, please refer to the standardized synthesis protocol outlined below.
- Prepare the reaction system containing 150-500g/L of racemic 3-cyclohexene-1-carboxylate substrate, PBS buffer (pH 8.0), and 1-20g/L of the freeze-dried AcEst1 mutant cells at 20-40°C.
- Maintain the reaction pH at 8.0 through automatic titration with base (e.g., Na2CO3) to neutralize the acid produced, ensuring optimal enzyme activity until the enantiomeric excess (ees) exceeds 99%.
- Extract the unreacted (R)-ester with ethyl acetate under alkaline conditions, followed by saponification with NaOH and acidification to isolate the final (R)-3-cyclohexene-1-carboxylic acid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this enzymatic technology translates into tangible strategic benefits that extend beyond simple yield metrics. The transition from chemical resolution to biocatalysis fundamentally alters the cost structure of producing chiral cyclohexene intermediates. By eliminating the need for hazardous reagents like gaseous butadiene and vast quantities of organic solvents such as acetone, the process significantly reduces raw material costs and waste disposal fees. The use of a recombinant enzyme produced in E. coli ensures a consistent and reliable supply of the biocatalyst, removing the volatility associated with sourcing animal organs for enzyme extraction. This stability is critical for long-term supply agreements with pharmaceutical clients who require rigorous validation and consistency in their raw material streams. Moreover, the mild reaction conditions reduce energy consumption for heating and cooling, contributing to a lower carbon footprint and aligning with corporate sustainability goals.
- Cost Reduction in Manufacturing: The enzymatic process offers a distinct economic advantage by simplifying the downstream processing train. Traditional chemical methods often require multiple recrystallization steps to achieve pharmaceutical-grade purity, each step incurring losses in yield and increases in labor and utility costs. In contrast, the high stereoselectivity of the AcEst1 mutant allows for the direct isolation of high-purity intermediates with minimal purification. The elimination of expensive transition metal catalysts and the reduction in solvent usage further drive down the variable costs per kilogram. Additionally, the ability to operate at high substrate concentrations means that smaller reactors can produce the same output as larger conventional vessels, optimizing capital utilization and reducing the fixed cost burden on the manufacturing unit.
- Enhanced Supply Chain Reliability: Dependence on animal-derived enzymes introduces significant supply chain risks, including seasonal availability, geographic limitations, and regulatory hurdles regarding animal byproducts. The recombinant production of the carboxylesterase mutant in a microbial host decouples the catalyst supply from these biological constraints. This ensures a continuous, scalable production capacity that can be rapidly ramped up to meet surges in demand for key APIs like tacrolimus or edoxaban precursors. The robustness of the enzyme also minimizes the risk of batch failures due to catalyst degradation, ensuring that production schedules are met consistently and that lead times for high-purity pharmaceutical intermediates are significantly shortened.
- Scalability and Environmental Compliance: Regulatory pressure to reduce volatile organic compound (VOC) emissions is intensifying globally. This biocatalytic route operates primarily in aqueous buffers, drastically cutting VOC emissions compared to acetone-based chemical resolutions. The process generates less hazardous waste, simplifying compliance with environmental regulations and reducing the liability associated with waste treatment. From a scale-up perspective, the reaction kinetics are well-suited for large stirred-tank reactors, and the lack of exothermic hazards associated with chemical reagents makes the process inherently safer. This safety profile facilitates easier regulatory approval for new manufacturing sites and supports the seamless transfer of technology from pilot scale to multi-ton commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this carboxylesterase mutant technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear picture of the technology's capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this biocatalytic step into existing manufacturing workflows.
Q: How does the AcEst1 mutant differ from wild-type enzymes in terms of stereoselectivity?
A: Unlike wild-type AcEst1 or animal-derived esterases which typically favor the (S)-enantiomer, the engineered mutant (T248W/L249A/V254K/T258A/V257F/L297F/N329F) exhibits a complete inversion of stereoselectivity, specifically hydrolyzing the (S)-ester to leave behind the high-purity (R)-ester with an ee value exceeding 99%.
Q: What represents the primary advantage of this biocatalytic route over chemical resolution?
A: Traditional chemical resolution requires multiple recrystallizations in large volumes of acetone with a theoretical maximum yield of only 50% (often practically 20-30%). This enzymatic process operates under mild aqueous conditions, eliminates hazardous organic solvents, and achieves superior optical purity without the need for extensive downstream purification.
Q: Is this process suitable for large-scale industrial manufacturing of API intermediates?
A: Yes, the patent demonstrates high substrate tolerance up to 500g/L and uses a recombinant E. coli expression system, which ensures consistent enzyme supply and eliminates the batch-to-batch variability and viral risks associated with animal-derived enzymes, making it highly scalable for commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (R)-3-Cyclohexene-1-Carboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality chiral intermediates in the development and manufacture of life-saving medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which utilize state-of-the-art analytical instrumentation to verify enantiomeric excess and chemical purity at every stage of production. Our facility is equipped to handle complex biocatalytic processes, including the fermentation and downstream processing required for enzymes like the AcEst1 mutant, guaranteeing a secure and consistent supply for your global operations.
We invite you to collaborate with us to leverage this advanced biocatalytic technology for your specific project needs. Whether you are looking to optimize an existing route or develop a new synthetic pathway for (R)-3-cyclohexene-1-carboxylic acid derivatives, our experts are ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our commitment to innovation and quality can drive value for your organization.
