Revolutionizing Sitagliptin Intermediate Production with Engineered Transaminase Mutants for Commercial Scale
Revolutionizing Sitagliptin Intermediate Production with Engineered Transaminase Mutants for Commercial Scale
The pharmaceutical industry's relentless pursuit of efficient, sustainable, and cost-effective manufacturing processes has brought biocatalysis to the forefront of API intermediate production. A pivotal development in this domain is documented in Chinese patent CN108866021B, which discloses a highly specialized omega-transaminase mutant derived from Arthrobacter nitroguajacolicus. This technology addresses the critical bottlenecks associated with the synthesis of sitagliptin, a blockbuster DPP-IV inhibitor for type II diabetes. By leveraging directed evolution and protein engineering, this patent presents a robust biocatalytic route that converts prochiral ketone precursors into optically pure chiral amines with exceptional stereoselectivity. For R&D directors and procurement strategists, this represents a paradigm shift from traditional chemocatalysis to a greener, more precise enzymatic methodology that promises significant operational advantages.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of sitagliptin intermediates has relied heavily on complex chemical methodologies that pose substantial challenges for large-scale manufacturing. Prior art, such as US6699871 and various international patents, describes routes involving chiral pool synthesis or asymmetric hydrogenation using precious metal catalysts like Ruthenium or Platinum. These conventional chemical pathways are fraught with inefficiencies; they often require harsh reaction conditions, including high-pressure hydrogenation, which necessitates specialized and costly reactor infrastructure. Furthermore, the stereoselectivity achieved through these chemical means is frequently insufficient, typically yielding e.e. values around 94-96%, thereby mandating additional recrystallization steps to meet pharmaceutical purity standards. The reliance on heavy metals also introduces severe environmental and regulatory burdens regarding metal residue limits in the final API, complicating the purification workflow and inflating production costs.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes an engineered omega-transaminase (specifically Mutant 1, SEQ ID NO: 4) to catalyze the asymmetric transamination of sitagliptin precursor ketones. This biocatalytic strategy operates under mild physiological conditions, typically between 30-45°C and at atmospheric pressure, drastically reducing energy consumption and safety risks. The process employs isopropylamine as a cheap and readily available amino donor, driving the equilibrium towards the desired chiral amine product. Crucially, this enzymatic route achieves an outstanding enantiomeric excess (e.e.) of greater than 99%, effectively eliminating the need for downstream chiral resolution. The versatility of this mutant enzyme is further demonstrated by its ability to accept a wide range of ester substrates, as illustrated in the reaction scheme below, enabling the flexible production of various sitagliptin ester intermediates.
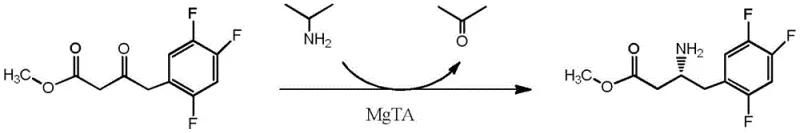
Mechanistic Insights into Omega-Transaminase Catalyzed Asymmetric Transamination
The core of this technological breakthrough lies in the sophisticated mechanism of the omega-transaminase, which facilitates the transfer of an amino group from a donor molecule to a prochiral ketone acceptor. The enzyme utilizes pyridoxal phosphate (PLP) as an essential cofactor, forming a Schiff base intermediate with the substrate to lower the activation energy for the stereo-specific amination. The mutant enzyme described in the patent features six specific amino acid substitutions (I45V, G68Y, I103L, S128A, Y157S, D327E) compared to the wild-type sequence. These mutations are strategically located to reshape the active site pocket, enhancing substrate binding affinity and accommodating the bulky 2,4,5-trifluorophenyl moiety of the sitagliptin precursor. This structural optimization allows the enzyme to distinguish effectively between the pro-R and pro-S faces of the ketone, ensuring the exclusive formation of the (R)-enantiomer required for biological activity.
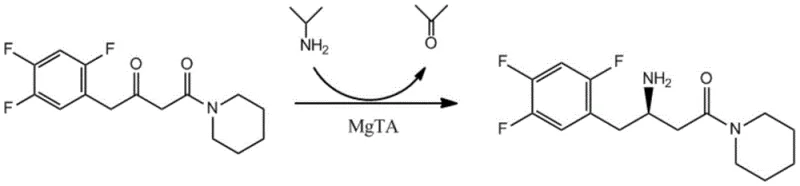
From an impurity control perspective, the high specificity of the biocatalyst inherently minimizes the formation of by-products that are common in chemical synthesis, such as over-reduction products or racemic mixtures. The reaction system is carefully buffered using triethanolamine at pH 8-9, which maintains the optimal ionization state of the enzyme and the substrate throughout the conversion. The use of dimethyl sulfoxide (DMSO) as a co-solvent further enhances the solubility of the hydrophobic fluorinated ketone substrate, ensuring homogeneous reaction kinetics and preventing substrate inhibition. This precise control over the reaction environment results in a clean product profile, simplifying the subsequent isolation and purification steps and ensuring that the final intermediate meets stringent pharmaceutical quality specifications without extensive chromatographic purification.
How to Synthesize Sitagliptin Intermediate Efficiently
The implementation of this biocatalytic process involves a streamlined workflow that integrates microbial fermentation with enzymatic conversion. The protocol begins with the cultivation of recombinant Escherichia coli harboring the transaminase mutant gene, followed by induction to express the biocatalyst. The resulting wet cells are then employed directly in the transamination reaction, avoiding the need for enzyme purification. This "whole-cell" approach not only preserves enzyme stability but also significantly reduces processing time and material costs. The reaction proceeds efficiently with high substrate loading, and the workup involves a straightforward acid-base extraction protocol to isolate the pure chiral amine. For detailed operational parameters and step-by-step instructions, please refer to the standardized guide below.
- Ferment recombinant Escherichia coli BL21/pET28b-MgTAmut1 in LB medium with kanamycin, induce expression with IPTG at OD600 0.6-0.8, and harvest wet cells via centrifugation.
- Conduct the biocatalytic reaction in a triethanolamine buffer (pH 8-9) containing the sitagliptin precursor ketone, isopropylamine as amino donor, PLP cofactor, and DMSO cosolvent at 30-45°C.
- Separate the product by adjusting pH to acidic conditions to remove cells, followed by liquid-liquid extraction with dichloromethane and basification to isolate the high-purity chiral amine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this transaminase-mediated synthesis offers compelling economic and logistical benefits that extend beyond mere technical feasibility. The transition from metal-catalyzed chemical synthesis to biocatalysis fundamentally alters the cost structure of API intermediate manufacturing. By eliminating the dependency on scarce and expensive precious metals like Ruthenium and Platinum, companies can insulate their supply chains from volatile commodity price fluctuations. Furthermore, the removal of heavy metal catalysts obviates the need for complex and costly metal scavenging steps, which are mandatory to comply with strict ICH Q3D guidelines for elemental impurities in pharmaceuticals. This simplification of the downstream process translates directly into reduced operational expenditures and shorter batch cycle times.
- Cost Reduction in Manufacturing: The biocatalytic route leverages inexpensive raw materials, such as isopropylamine, and utilizes whole-cell biocatalysts that can be produced via scalable fermentation processes. This eliminates the capital expenditure associated with high-pressure hydrogenation reactors and the recurring costs of noble metal catalysts. Additionally, the high stereoselectivity (>99% e.e.) removes the financial burden of chiral resolution steps, such as recrystallization or chiral chromatography, which typically result in significant yield losses. The overall process efficiency leads to a substantial reduction in the cost of goods sold (COGS) for the sitagliptin intermediate.
- Enhanced Supply Chain Reliability: Relying on biological systems for synthesis enhances supply chain resilience by diversifying the source of chirality away from finite mineral resources. The recombinant strains can be stored and propagated indefinitely, ensuring a consistent and renewable supply of the biocatalyst. The mild reaction conditions reduce the risk of safety incidents associated with high-pressure hydrogen handling, thereby minimizing unplanned downtime. Moreover, the robustness of the mutant enzyme across a range of substrate concentrations allows for flexible manufacturing schedules that can adapt to fluctuating market demands without compromising product quality.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, having been validated from laboratory scale to potential industrial tonnage. The aqueous-based reaction system generates significantly less hazardous waste compared to traditional organic synthesis, aligning with green chemistry principles and reducing waste disposal costs. The absence of toxic reagents like cyanide (used in some alternative chemical routes) simplifies environmental permitting and regulatory compliance. This eco-friendly profile not only mitigates regulatory risk but also enhances the corporate sustainability image, which is increasingly valued by global pharmaceutical partners.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating this technology for integration into their manufacturing pipelines, we have compiled answers to common inquiries regarding the transaminase mutant's performance and application. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation. Understanding these nuances is critical for assessing the feasibility of adopting this biocatalytic route for your specific production needs.
Q: What are the advantages of this transaminase mutant over traditional chemical synthesis for sitagliptin?
A: Unlike traditional chemical routes that require expensive heavy metal catalysts (such as Ruthenium or Platinum) and high-pressure hydrogenation, this biocatalytic method operates under mild atmospheric conditions (30-45°C) and achieves superior stereoselectivity with an e.e. value exceeding 99%, eliminating the need for complex chiral resolution steps.
Q: Can this enzyme mutant handle different ester substrates beyond the piperidine ketone?
A: Yes, the engineered transaminase mutant (SEQ ID NO: 4) demonstrates broad substrate specificity. Patent data confirms high conversion rates (83-91%) for various ester derivatives including methyl, ethyl, propyl, isopropyl, isobutyl, and benzyl esters of 3-carbonyl-4-(2,4,5-trifluorophenyl)butyric acid.
Q: How does the use of wet cells impact the production cost compared to pure enzyme?
A: Using wet cells directly as the biocatalyst significantly reduces downstream processing costs by bypassing the expensive protein purification steps (such as affinity chromatography and dialysis) required for pure enzyme isolation, while maintaining high catalytic efficiency and conversion rates suitable for industrial application.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sitagliptin Intermediate Supplier
The technological advancements presented in patent CN108866021B underscore the immense potential of enzyme engineering in modern pharmaceutical manufacturing. At NINGBO INNO PHARMCHEM, we recognize the strategic value of such innovations and possess the technical expertise to translate these laboratory-scale breakthroughs into commercial reality. As a premier CDMO partner, we have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex biocatalytic processes are optimized for efficiency and yield. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of sitagliptin intermediate meets the highest global regulatory standards.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this cutting-edge transaminase technology for their API supply chains. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our biocatalytic capabilities can drive down your manufacturing costs while securing a reliable supply of high-purity pharmaceutical intermediates.
