Advanced Biocatalytic Synthesis of Sitagliptin Intermediates Using Engineered Recombinant Transaminases
The pharmaceutical industry continuously seeks robust biocatalytic solutions to enhance the efficiency of chiral amine synthesis, particularly for high-volume drugs like sitagliptin. A significant breakthrough in this domain is documented in Chinese patent CN111518783B, which discloses a novel recombinant (R)-ω-transaminase and its specific mutants designed for the preparation of sitagliptin. This technology addresses critical bottlenecks in enzymatic asymmetric synthesis by offering a biocatalyst with remarkably improved specific activity and stability. The patent details the development of a genetically engineered strain capable of converting the precursor ketone substrate, 1-[3-(trifluoromethyl)-5,6,7,8-tetrahydro-1,2,4-triazolo[4,3-a]pyrazin-7-yl]-4-(2,4,5-trifluorophenyl)-1,3-butanedione, into the corresponding chiral amine with unprecedented efficiency. By leveraging advanced gene recombination and site-directed mutagenesis techniques, the inventors have created a biocatalytic system that not only surpasses the activity of wild-type enzymes but also outperforms previously known mutants in terms of conversion rates at high substrate loadings. This innovation represents a pivotal shift towards more sustainable and cost-effective manufacturing processes for dipeptidyl peptidase-4 (DPP-4) inhibitors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional chemical synthesis routes for sitagliptin often involve the use of heavy metal catalysts or require complex resolution steps to achieve the desired chirality, which can lead to significant environmental burdens and increased production costs. Even within the realm of biocatalysis, earlier generations of transaminases, such as the widely cited ATA-117, faced limitations regarding their catalytic efficiency at industrially relevant substrate concentrations. For instance, prior art indicates that while certain mutants could achieve reasonable conversion, they often required lower substrate loadings to maintain activity, resulting in larger reactor volumes and higher solvent consumption. Furthermore, the stability of these enzymes under process conditions was sometimes insufficient, leading to batch-to-batch variability and the need for excessive enzyme loading to drive reactions to completion. These factors collectively contribute to a higher cost of goods sold (COGS) and complicate the supply chain reliability for reliable sitagliptin intermediate supplier networks seeking to optimize their manufacturing footprint.
The Novel Approach
In contrast, the novel approach described in the patent utilizes a specifically engineered recombinant (R)-ω-transaminase mutant that has been optimized through rational design and directed evolution. This new biocatalyst demonstrates a substantial improvement in enzyme activity, reaching levels up to 785.2 U/g, which allows for effective catalysis even when the precursor ketone substrate concentration is pushed to 900 mM or higher. The process utilizes isopropylamine as an inexpensive amino donor and pyridoxal phosphate (PLP) as a cofactor, operating under mild aqueous conditions that are inherently safer and greener than traditional chemical methods. 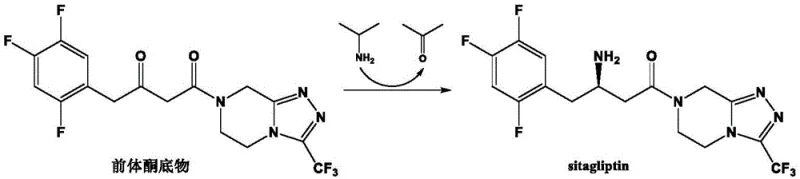 As illustrated in the reaction scheme, the enzyme facilitates the direct asymmetric amination of the ketone to yield sitagliptin with high fidelity. This method eliminates the need for precious metal catalysts and reduces the generation of hazardous waste, aligning perfectly with modern green chemistry principles while delivering superior cost reduction in API manufacturing through enhanced space-time yields.
As illustrated in the reaction scheme, the enzyme facilitates the direct asymmetric amination of the ketone to yield sitagliptin with high fidelity. This method eliminates the need for precious metal catalysts and reduces the generation of hazardous waste, aligning perfectly with modern green chemistry principles while delivering superior cost reduction in API manufacturing through enhanced space-time yields.
Mechanistic Insights into Recombinant (R)-ω-Transaminase Catalysis
The core of this technological advancement lies in the precise molecular engineering of the transaminase active site. The enzyme operates via a ping-pong bi-bi mechanism involving the cofactor pyridoxal 5'-phosphate (PLP). Initially, the amino group from the donor (isopropylamine) is transferred to the PLP bound to the enzyme, forming pyridoxamine 5'-phosphate (PMP) and releasing acetone. Subsequently, the PMP transfers the amino group to the carbonyl acceptor, the sitagliptin precursor ketone, regenerating the PLP form of the enzyme and releasing the chiral amine product. The specific mutations introduced, such as the substitution of serine at position 214 with proline (S214P) and methionine at position 146 with glutamine (M146Q), are strategically located to optimize the binding pocket geometry. These modifications enhance the accommodation of the bulky trifluoromethyl-substituted triazolopyrazine moiety of the substrate, thereby reducing steric hindrance and improving the turnover number (kcat).
Furthermore, the stereochemical outcome of the reaction is rigorously controlled by the chiral environment of the enzyme's active site, ensuring exclusive formation of the (R)-enantiomer. The patent data confirms that the recombinant mutants achieve an enantiomeric excess (e.e.) value of greater than 99.9%, which is critical for meeting the stringent purity requirements of pharmaceutical regulators. This high level of stereoselectivity minimizes the presence of the (S)-impurity, which could otherwise complicate downstream crystallization and purification processes. By understanding the structure-activity relationship (SAR) of these mutants, manufacturers can predictably scale the reaction knowing that the enzyme will maintain its specificity even under the stress of high substrate concentrations and extended reaction times, ensuring consistent quality of the high-purity sitagliptin precursor throughout the production campaign.
How to Synthesize Sitagliptin Efficiently
The implementation of this biocatalytic route requires a systematic approach to strain construction, fermentation, and biotransformation to fully realize its potential. The process begins with the cloning of the optimized gene sequence into a suitable expression vector, followed by transformation into a robust host strain like E. coli BL21(DE3). Once the recombinant strain is established, the focus shifts to optimizing the fermentation conditions to maximize cell density and enzyme expression levels. The subsequent biotransformation step involves balancing the substrate concentration, pH, temperature, and cofactor regeneration to drive the equilibrium towards the product. For a comprehensive understanding of the operational parameters, the detailed standardized synthesis steps are provided in the guide below.
- Construct recombinant expression plasmids containing the novel (R)-ω-transaminase gene or its specific mutants (e.g., S214P, M146Q) into E. coli BL21(DE3) host cells.
- Perform induced expression of the recombinant engineering bacteria in LB medium with IPTG, followed by ultrasonic disruption to collect the enzyme-containing supernatant or whole cells.
- Conduct the biocatalytic reaction using the precursor ketone substrate, isopropylamine as an amino donor, and PLP as a coenzyme in a triethanolamine-HCl buffer at optimized pH and temperature.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel recombinant transaminase technology offers tangible strategic benefits that extend beyond simple technical metrics. The primary advantage lies in the drastic simplification of the manufacturing process, which directly translates to improved supply chain resilience and reduced operational risks. By replacing multi-step chemical syntheses or less efficient enzymatic processes with a single, highly efficient biocatalytic step, companies can reduce their dependency on multiple raw material suppliers and minimize the number of unit operations required. This consolidation of the process flow enhances overall equipment effectiveness (OEE) and allows for faster turnaround times between batches, which is essential for meeting fluctuating market demands for diabetes medications without compromising on quality or compliance standards.
- Cost Reduction in Manufacturing: The significantly higher specific activity of the mutant enzyme (785.2 U/g) means that substantially less biocatalyst is required to achieve the same conversion compared to older generations of transaminases. This reduction in enzyme loading directly lowers the variable cost per kilogram of the product. Additionally, the ability to operate at high substrate concentrations (up to 900 mM) reduces the volume of solvent and water needed per unit of product, leading to smaller reactor footprints and lower energy consumption for heating, cooling, and downstream solvent recovery. The elimination of expensive transition metal catalysts further contributes to cost reduction in API manufacturing by removing the need for costly metal scavenging steps and associated waste disposal fees.
- Enhanced Supply Chain Reliability: The robustness of the recombinant E. coli strain ensures consistent enzyme production, mitigating the risk of supply interruptions caused by biological variability. The high conversion rate of 97.1% at elevated substrate loads implies that the process is less sensitive to minor fluctuations in reaction conditions, providing a wider operating window for manufacturing teams. This reliability is crucial for maintaining a steady flow of high-purity sitagliptin precursor to downstream formulation sites. Furthermore, the use of readily available and inexpensive reagents like isopropylamine ensures that the supply chain is not vulnerable to the price volatility often associated with specialized chemical reagents or rare earth metals.
- Scalability and Environmental Compliance: The process is inherently scalable, having been validated from laboratory screening to conditions suitable for commercial scale-up of complex pharmaceutical intermediates. The aqueous nature of the reaction and the use of biodegradable enzymes align with increasingly strict environmental regulations regarding volatile organic compound (VOC) emissions and heavy metal discharge. By minimizing the generation of hazardous waste and reducing the overall E-factor (mass of waste per mass of product), manufacturers can achieve better sustainability scores and avoid potential regulatory penalties. This environmental compatibility not only future-proofs the manufacturing asset but also enhances the brand reputation of the pharmaceutical company among eco-conscious stakeholders and investors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this recombinant transaminase technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a factual basis for decision-making. Understanding these nuances is essential for R&D teams evaluating the feasibility of technology transfer and for procurement specialists assessing the long-term viability of the supply source. The high stereoselectivity and activity data provide strong evidence for the maturity of this biocatalytic platform.
Q: What represents the key advantage of the novel recombinant (R)-ω-transaminase mutant over wild-type enzymes?
A: The novel mutant exhibits significantly higher enzyme activity (up to 785.2 U/g) and exceptional stereoselectivity (>99.9% e.e. value), allowing for efficient catalysis at high substrate concentrations compared to prior art enzymes.
Q: Can this biocatalytic process support high-concentration substrate loading for industrial production?
A: Yes, the optimized mutant demonstrates robust performance at substrate concentrations ranging from 800 mM to 1000 mM, achieving conversion rates as high as 97.1%, which is critical for reducing solvent usage and improving process economics.
Q: How does the stereoselectivity of this enzyme impact the purification of the final API?
A: With an e.e. value exceeding 99.9% for the R-configuration, the enzyme minimizes the formation of unwanted S-enantiomer impurities, thereby simplifying downstream purification steps and ensuring compliance with stringent pharmaceutical purity specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sitagliptin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced biocatalysis in the production of next-generation pharmaceutical intermediates. As a dedicated CDMO partner, we possess the technical expertise and infrastructure to translate innovative patent technologies like the recombinant (R)-ω-transaminase mutant into commercial reality. Our facilities are equipped to handle the commercial scale-up of complex pharmaceutical intermediates, with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We adhere to stringent purity specifications and operate rigorous QC labs to ensure that every batch of sitagliptin intermediate meets the highest global regulatory standards, providing our partners with the confidence needed to accelerate their drug development timelines.
We invite pharmaceutical companies and biotech firms to collaborate with us to leverage this high-efficiency enzymatic route for their sitagliptin supply needs. By partnering with our technical team, you can access a Customized Cost-Saving Analysis tailored to your specific production volumes and quality requirements. We encourage you to contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments. Let us help you optimize your supply chain and achieve superior economic outcomes through the power of precision biocatalysis.
