Advanced Biocatalytic Synthesis of Edoxaban Intermediates via Engineered Esterase Mutants
Advanced Biocatalytic Synthesis of Edoxaban Intermediates via Engineered Esterase Mutants
The pharmaceutical industry is constantly seeking more efficient and sustainable pathways for synthesizing critical chiral intermediates, particularly for high-value anticoagulants like Edoxaban. A significant breakthrough in this domain is documented in patent CN116376872A, which discloses a novel recombinant esterase mutant, its encoding gene, and the corresponding engineering bacteria designed specifically for the preparation of (S)-3-cyclohexene-1-carboxylic acid. This technology represents a paradigm shift from traditional chemical resolution methods to highly selective biocatalysis. The patent highlights that the provided recombinant esterase mutants exhibit superior catalytic activity, allowing for mild reaction conditions that are both environmentally friendly and economically viable. Specifically, the data indicates that using 20g/L of esterase mutant wet cells, a substrate concentration of 100g/L of racemic methyl 3-cyclohexene-1-carboxylate can be processed with a conversion rate exceeding 49.9% within just 2 hours. This level of efficiency addresses the long-standing challenges of low yield and high cost associated with the manufacturing of this key pharmaceutical building block.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of (S)-3-cyclohexene-1-carboxylic acid has relied heavily on diastereomeric resolution technology, a process fraught with inefficiencies and economic burdens. This conventional approach utilizes chiral phenylethylamine as a resolving agent to form diastereomeric salts with the racemic acid, which are then separated based on solubility differences in acetone. The drawbacks of this method are substantial; it requires slow cooling and up to six recrystallization cycles to achieve optical purity greater than 99%, resulting in a dismal final yield of approximately 28.7% for the desired (S)-enantiomer. Furthermore, the chiral resolving agent itself is expensive and required in large stoichiometric quantities, driving up raw material costs significantly. The operational complexity of multiple crystallization steps also increases energy consumption and extends production lead times, making the supply chain vulnerable to bottlenecks. From a green chemistry perspective, the generation of substantial waste solvent and the inability to recycle the unwanted (R)-enantiomer efficiently render this process unsustainable for modern large-scale manufacturing demands.
The Novel Approach
In stark contrast, the novel approach detailed in the patent leverages the power of protein engineering to create esterase mutants with enhanced stereoselectivity and catalytic vigor. By introducing specific single-point mutations into the wild-type esterase EST10 derived from Geobacillus stearothermophilus, researchers have developed variants such as EST10-F25I, EST10-R141G, and EST10-L167F that dramatically outperform their predecessors. These mutants facilitate a kinetic resolution of the racemic ester substrate under remarkably mild conditions, typically at 30°C and neutral pH, eliminating the need for harsh chemicals or extreme temperatures. The process achieves a theoretical maximum conversion of nearly 50% for the desired enantiomer with exceptional enantiomeric excess (ee) values reaching 99%, effectively doubling the yield compared to traditional resolution methods. This biocatalytic route not only simplifies the workflow by using whole wet cells as catalysts but also aligns perfectly with the principles of green chemistry by reducing solvent usage and avoiding toxic heavy metals, thereby offering a reliable pharmaceutical intermediate supplier solution that is both cost-effective and environmentally responsible.
Mechanistic Insights into Esterase-Catalyzed Kinetic Resolution
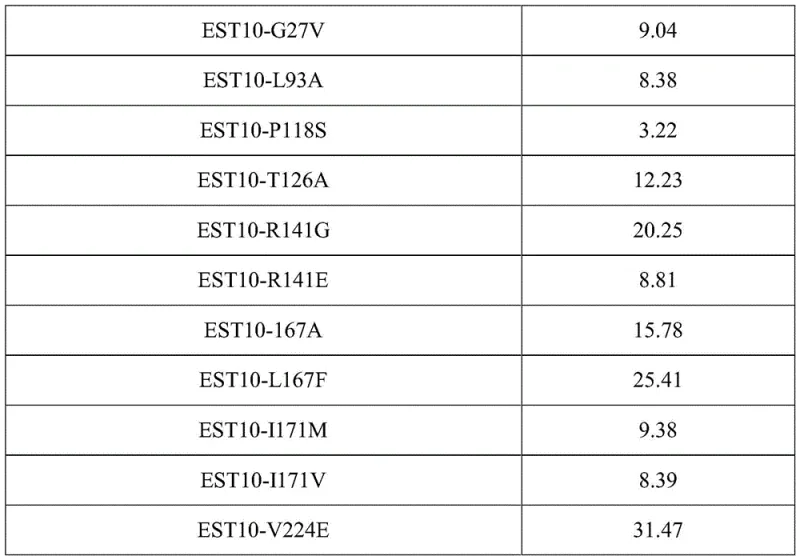
The core of this technological advancement lies in the precise modification of the enzyme's active site to optimize substrate binding and transition state stabilization. Esterases, belonging to the α/β-hydrolase fold superfamily, typically utilize a catalytic triad composed of Serine, Aspartate/Glutamate, and Histidine residues to hydrolyze ester bonds. In the wild-type EST10, the spatial arrangement of the active pocket may not be perfectly suited for the bulky cyclohexene ring of the substrate, leading to lower activity or selectivity. The patent describes a rational design strategy where key amino acid residues surrounding the active pocket—specifically at positions 25, 141, 167, 171, and 224—were targeted for saturation mutagenesis. Computational modeling using tools like Swiss-Model and DeepDDG allowed for the prediction of energy changes associated with these mutations, guiding the selection of variants that lower the activation energy for the hydrolysis of the specific (S)-enantiomer precursor. For instance, mutating Phenylalanine at position 25 to Isoleucine (F25I) likely alters the hydrophobic interactions within the binding cleft, allowing for a tighter and more specific fit for the substrate, thereby enhancing the turnover number.
Furthermore, the mechanism ensures rigorous impurity control through high stereoselectivity, which is critical for regulatory compliance in API manufacturing. The enzymatic hydrolysis proceeds via a classic acyl-enzyme intermediate mechanism where the serine residue attacks the carbonyl carbon of the ester, forming a tetrahedral intermediate stabilized by the oxyanion hole. The specificity of the mutant enzymes ensures that only the (R)-ester is hydrolyzed rapidly to the acid (or vice versa depending on the specific enantio-preference defined, here producing the S-acid), leaving the other enantiomer largely untouched or converting it at a negligible rate. This biological discrimination results in product streams with ee values consistently above 99%, minimizing the formation of chiral impurities that are difficult to remove downstream. The stability of these mutants in aqueous buffer systems at moderate temperatures further prevents non-enzymatic background hydrolysis, which could otherwise erode optical purity. This high fidelity in chiral recognition translates directly to reduced downstream purification costs and higher overall process robustness.
How to Synthesize (S)-3-cyclohexene-1-carboxylic acid Efficiently
Implementing this biocatalytic route requires a structured approach to fermentation and biotransformation to maximize the potential of the engineered strains. The process begins with the cultivation of the recombinant E.coli BL21(DE3) host strains carrying the specific esterase mutant plasmids. Precise control of induction parameters, such as IPTG concentration and temperature, is essential to ensure high expression levels of soluble, active enzyme within the cells. Once the biomass is harvested, it can be used directly as wet cells, bypassing the need for extensive purification, which streamlines the operation significantly. The subsequent biotransformation step involves suspending these cells in a buffered medium and feeding the racemic ester substrate. Maintaining optimal pH and temperature throughout the reaction is crucial to sustain enzyme activity and prevent denaturation. For a detailed breakdown of the standardized operating procedures, including specific media compositions and workup protocols, please refer to the technical guide below.
- Ferment recombinant E.coli BL21(DE3) strains containing specific esterase mutant genes (e.g., EST10-F25I) in LB medium with kanamycin resistance, inducing expression with IPTG at 28°C to obtain wet cell biomass.
- Prepare the reaction system by suspending wet cells (20g/L) in potassium phosphate buffer (pH 7.0) and adding racemic methyl 3-cyclohexene-1-carboxylate substrate to a final concentration of 100g/L.
- Maintain the reaction at 30°C with stirring at 600rpm for 2 hours, controlling pH at 7.0 via NaOH addition, followed by extraction and purification to isolate the high-purity (S)-enantiomer.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this recombinant esterase technology offers transformative benefits that extend far beyond simple yield improvements. The shift from chemical resolution to enzymatic kinetic resolution fundamentally alters the cost structure of manufacturing (S)-3-cyclohexene-1-carboxylic acid. By eliminating the requirement for expensive chiral resolving agents like phenylethylamine and removing the need for multiple energy-intensive recrystallization steps, the direct material and utility costs are significantly reduced. Additionally, the ability to use crude wet cell biomass as the catalyst avoids the capital expenditure and operational complexity associated with enzyme purification facilities. This simplification of the manufacturing process enhances supply chain reliability by reducing the number of unit operations and potential failure points, ensuring a more consistent and continuous supply of this critical intermediate for anticoagulant drug production.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the drastic simplification of the downstream processing train. Traditional methods suffer from a theoretical yield cap of 50% which is practically reduced to ~28% due to mechanical losses during repeated crystallizations, meaning over 70% of the starting material is wasted or requires recycling. The enzymatic process pushes conversion efficiency close to the theoretical limit for kinetic resolution (>49.9%) in a single pass, effectively doubling the output per kilogram of raw material input. Furthermore, the mild reaction conditions (30°C) reduce energy consumption for heating and cooling compared to processes requiring reflux or cryogenic conditions. The removal of stoichiometric chiral auxiliaries also eliminates the cost of purchasing, recovering, or disposing of these expensive reagents, leading to substantial overall cost savings in API manufacturing.
- Enhanced Supply Chain Reliability: Reliability in the supply of key intermediates is paramount for pharmaceutical companies managing global drug portfolios. This biocatalytic process utilizes robust E.coli expression systems that are well-established in the industry, ensuring that the catalyst itself can be produced reliably at scale without dependency on rare natural sources or complex extraction from animal tissues (like porcine liver esterase). The high substrate tolerance (up to 100g/L) means that reactor volumes can be optimized, allowing for larger batch sizes and fewer production runs to meet demand. This scalability reduces the risk of supply shortages and provides flexibility to ramp up production quickly in response to market fluctuations for Edoxaban and related anticoagulants, securing the supply chain against volatility.
- Scalability and Environmental Compliance: As regulatory pressures regarding environmental sustainability intensify, the green credentials of this process offer a distinct competitive advantage. The enzymatic reaction occurs in aqueous buffers, significantly reducing the volume of organic solvents required compared to the acetone-heavy crystallization processes of the past. This reduction in solvent usage lowers the burden on waste treatment facilities and minimizes the facility's environmental footprint. Moreover, the biodegradability of the biocatalyst and the absence of heavy metal catalysts simplify waste disposal and compliance reporting. The process is inherently safer, operating at ambient pressure and moderate temperatures, which reduces industrial safety risks and insurance costs, making it an ideal candidate for commercial scale-up of complex pharmaceutical intermediates in regulated markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this esterase mutant technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on how this innovation resolves historical pain points in chiral synthesis. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this biocatalytic route into existing production frameworks.
Q: How does the new esterase mutant technology improve upon traditional chiral resolution methods?
A: Traditional methods rely on diastereomeric salt formation using expensive chiral phenylethylamine, requiring multiple recrystallizations and yielding only about 28% of the desired product. The new recombinant esterase mutants enable kinetic resolution under mild conditions, achieving conversion rates exceeding 49.9% with enantiomeric excess (ee) values greater than 99% in just 2 hours, significantly reducing material loss and processing time.
Q: What are the specific advantages of using wet cell biomass over purified enzymes for this process?
A: Using wet cell biomass directly as the biocatalyst eliminates the complex and costly downstream processing steps associated with enzyme purification, such as cell lysis, centrifugation, and chromatography. The patent demonstrates that 20g/L of wet cells are sufficient to catalyze high substrate loads (100g/L), simplifying the supply chain and drastically lowering the overall production cost while maintaining high catalytic efficiency and stereoselectivity.
Q: Is this biocatalytic process suitable for large-scale industrial manufacturing of anticoagulant intermediates?
A: Yes, the process is highly scalable due to its mild reaction conditions (30°C, pH 7.0) and high substrate tolerance. The use of robust E.coli expression systems allows for the production of large quantities of catalyst biomass, and the high conversion efficiency minimizes reactor volume requirements, making it an ideal candidate for commercial scale-up of complex pharmaceutical intermediates like those needed for Edoxaban.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-3-cyclohexene-1-carboxylic acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the synthesis of life-saving medications like Edoxaban. Our team of experts has thoroughly analyzed the potential of the recombinant esterase mutants described in patent CN116376872A and is well-positioned to leverage this technology for your projects. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs and stringent purity specifications, guaranteeing that every batch of (S)-3-cyclohexene-1-carboxylic acid meets the highest international standards for chirality and chemical purity required by top-tier pharmaceutical companies.
We invite you to collaborate with us to unlock the full commercial potential of this advanced biocatalytic process. By partnering with our technical procurement team, you can gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and supply chain constraints. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise in enzymatic synthesis can drive down your costs and secure your supply of this vital anticoagulant intermediate.
