Revolutionizing HMB Production: Advanced Nitrilase Catalysis for Commercial Scale-Up
The landscape of fine chemical manufacturing is undergoing a significant transformation driven by the urgent need for sustainable and efficient synthetic routes, particularly for high-value nutritional and pharmaceutical intermediates. A pivotal development in this sector is documented in patent CN115851685A, which discloses a novel nitrilase enzyme and its specific mutant derived from Erwinia billingiae. This technology addresses long-standing challenges in the synthesis of 3-hydroxy-3-methylbutyric acid (HMB), a critical compound widely utilized as a nutritional supplement for muscle health and as a potential therapeutic agent. The patent highlights a breakthrough where the engineered mutant, designated as EbNLE-R126A, demonstrates exceptional catalytic performance, achieving a substrate conversion rate of 99% and a production efficiency of 29.5g/L/d. For R&D directors and procurement strategists in the global fine chemicals market, this represents a paradigm shift from traditional, hazardous chemical oxidation methods to a precise, green biocatalytic platform that promises enhanced purity profiles and operational safety.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of 3-hydroxy-3-methylbutyric acid has relied heavily on chemical oxidation strategies that are increasingly viewed as unsustainable due to their environmental footprint and safety hazards. As illustrated in the prior art referenced by the patent, such as the methodology disclosed in EP2744489A1, the conventional route involves the oxidation of diacetone alcohol using sodium hypochlorite. This chemical pathway is fraught with significant drawbacks, primarily the generation of stoichiometric amounts of inorganic salts and the production of hazardous by-products like chloroform. The presence of such toxic halogenated solvents necessitates complex and costly downstream purification processes to ensure the final product meets the stringent safety standards required for human consumption or pharmaceutical applications. Furthermore, the overall yield of these oxidation reactions is often suboptimal, leading to material inefficiencies that drive up the cost of goods sold and complicate waste management protocols for manufacturers aiming to adhere to modern environmental regulations.
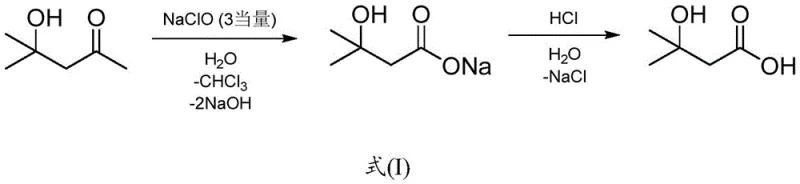
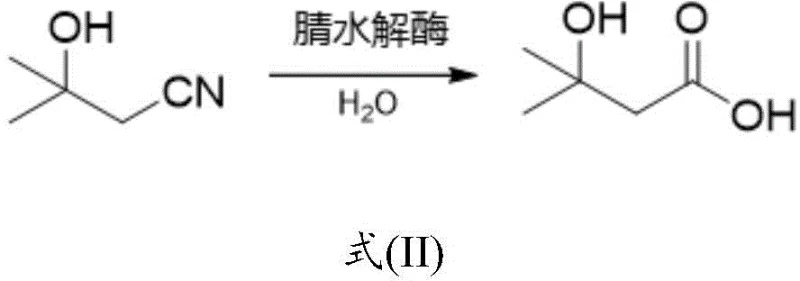
The Novel Approach
In stark contrast to these legacy chemical processes, the novel biocatalytic approach detailed in CN115851685A offers a streamlined and environmentally benign alternative that leverages the power of protein engineering. By utilizing the nitrilase mutant EbNLE-R126A, manufacturers can directly hydrolyze 3-hydroxy-3-methylbutyronitrile into the target acid under mild aqueous conditions. This biological route eliminates the need for harsh oxidizing agents and avoids the formation of toxic chlorinated organic compounds entirely. The specificity of the enzyme ensures that the reaction proceeds with high selectivity, minimizing the formation of side products that typically plague chemical synthesis. This shift not only aligns with the principles of green chemistry and atom economy but also drastically simplifies the isolation of the final product, thereby reducing the operational burden on production facilities and enabling a more reliable supply chain for high-purity nutritional ingredients.
Mechanistic Insights into Nitrilase-Catalyzed Hydrolysis
The core of this technological advancement lies in the precise molecular engineering of the nitrilase enzyme, which facilitates the direct hydrolysis of the nitrile group to a carboxylic acid without forming an amide intermediate. The wild-type enzyme EbNLE, sourced from Erwinia billingiae, already possesses the inherent capability to catalyze this transformation, but the introduction of the R126A mutation—where the arginine at position 126 is substituted with alanine—results in a dramatic enhancement of catalytic efficiency. Structural analysis suggests that this mutation likely optimizes the geometry of the active site or improves substrate accessibility, allowing for a much tighter binding affinity and faster turnover rate for the bulky 3-hydroxy-3-methylbutyronitrile substrate. This level of enzymatic optimization is critical for industrial applications where reaction time and catalyst loading directly impact the economic viability of the process.
From an impurity control perspective, the mechanism of nitrilase-catalyzed hydrolysis offers distinct advantages over non-enzymatic methods. Because the reaction occurs in a buffered aqueous system at a controlled pH of 6.5 to 7.5 and a moderate temperature of 30-42°C, there is minimal risk of thermal degradation or unwanted side reactions that often occur under the extreme conditions of chemical synthesis. The enzyme's high specificity means that it ignores other functional groups that might be present in crude feedstocks, leading to a cleaner reaction profile. For quality assurance teams, this translates to a significantly reduced impurity spectrum in the crude reaction mixture, which eases the burden on analytical laboratories and reduces the number of purification steps required to achieve pharmaceutical-grade purity. The ability to operate at near-neutral pH also preserves the integrity of the product, preventing acid-catalyzed dehydration or other decomposition pathways that could compromise yield.
How to Synthesize 3-Hydroxy-3-methylbutyric Acid Efficiently
Implementing this biocatalytic route requires a systematic approach to strain construction and process optimization to fully realize the productivity gains reported in the patent literature. The process begins with the genetic engineering of a robust host organism, typically E. coli BL21(DE3), transformed with a recombinant plasmid carrying the gene for the EbNLE-R126A mutant. Following fermentation and induction, the resulting biomass serves as the source of the biocatalyst, which can be used directly as resting cells or processed into a stable lyophilized powder. This flexibility allows manufacturers to decouple the enzyme production from the synthesis step, facilitating better inventory management and process control. The actual conversion step is remarkably straightforward, involving the suspension of the biocatalyst in a standard buffer system with the nitrile substrate, followed by agitation at physiological temperatures. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating these results.
- Construct recombinant E. coli BL21(DE3) strains harboring the pET-26b(+)-EbNLE-R126A plasmid and cultivate under induced conditions to express the mutant nitrilase.
- Prepare the biocatalyst by harvesting resting cells via centrifugation, followed by lyophilization to obtain freeze-dried cells or powder for stable storage.
- Perform the hydrolysis reaction in a Tris-HCl buffer system at 37°C and pH 7.0, converting 3-hydroxy-3-methylbutyronitrile to the target acid with high efficiency.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel nitrilase-based technology presents a compelling value proposition centered on cost efficiency, risk mitigation, and sustainability. Transitioning from a hazardous chemical oxidation process to a mild enzymatic hydrolysis fundamentally alters the cost structure of manufacturing 3-hydroxy-3-methylbutyric acid. By eliminating the need for expensive and dangerous reagents like sodium hypochlorite, and more importantly, by removing the generation of toxic chloroform, companies can achieve substantial cost savings in waste disposal and environmental compliance. The simplified downstream processing required to isolate the product from a cleaner reaction mixture further contributes to a reduction in overall manufacturing expenses, making the final ingredient more competitive in the global market.
- Cost Reduction in Manufacturing: The implementation of the EbNLE-R126A mutant drives down production costs primarily through the elimination of costly waste treatment protocols associated with halogenated by-products. In traditional chemical synthesis, a significant portion of the operational budget is allocated to the safe handling, neutralization, and disposal of toxic solvents and salt wastes; the biocatalytic route effectively removes this financial burden. Additionally, the high conversion rate of 99% ensures that raw material utilization is maximized, minimizing the loss of valuable substrate and reducing the cost per kilogram of the final active ingredient. This efficiency gain allows manufacturers to offer more competitive pricing to downstream clients in the nutraceutical and pharmaceutical sectors while maintaining healthy profit margins.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the regulatory scrutiny surrounding the transport and storage of hazardous chemicals; switching to a biological process mitigates these risks significantly. The enzymes and substrates used in this pathway are generally safer to handle and store, reducing the likelihood of logistical delays caused by safety inspections or regulatory hold-ups. Furthermore, the robustness of the recombinant E. coli expression system ensures a consistent and scalable supply of the biocatalyst itself, preventing bottlenecks that can occur when relying on scarce natural sources or complex chemical catalysts. This reliability is crucial for meeting the demanding delivery schedules of multinational corporations that require just-in-time inventory management.
- Scalability and Environmental Compliance: Scaling up biocatalytic processes is inherently safer and more straightforward than scaling exothermic chemical oxidations, which often require specialized reactors to manage heat and gas evolution. The mild reaction conditions of this nitrilase process mean that existing fermentation and conversion infrastructure can often be utilized with minimal modification, accelerating the time to market for commercial scale-up. From an environmental standpoint, the process aligns perfectly with increasingly strict global regulations on volatile organic compounds (VOCs) and hazardous waste, future-proofing the manufacturing site against potential regulatory crackdowns. This commitment to green manufacturing not only satisfies compliance requirements but also enhances the brand reputation of suppliers as responsible partners in the sustainable development of the fine chemicals industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this nitrilase technology, based on the specific data and embodiments disclosed in the patent documentation. These answers are designed to provide clarity on the operational parameters and the comparative benefits of this new synthetic route for stakeholders evaluating its adoption. Understanding these details is essential for making informed decisions about integrating this biocatalytic method into existing production portfolios.
Q: What are the primary advantages of the EbNLE-R126A mutant over conventional chemical synthesis?
A: Unlike conventional chemical oxidation which generates toxic chloroform and stoichiometric salt waste, the EbNLE-R126A mutant enables a green, aqueous-phase hydrolysis with a substrate conversion rate reaching 99%, significantly simplifying downstream purification and waste treatment.
Q: How does the production efficiency of this new biocatalytic method compare to previous enzymatic routes?
A: The new mutant achieves a production efficiency of 29.5g/L/d, which represents a substantial improvement over previously reported enzymatic cascades that struggled with low conversion rates around 26% and efficiencies near 3.28g/L/d.
Q: Is this process suitable for large-scale industrial manufacturing of nutritional intermediates?
A: Yes, the process utilizes robust E. coli expression systems and operates under mild conditions (37°C, neutral pH), making it highly scalable for commercial production of pharmaceutical and nutritional intermediates without requiring extreme pressure or temperature equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Hydroxy-3-methylbutyric acid Supplier
As the demand for high-quality nutritional and pharmaceutical intermediates continues to rise, partnering with a technically proficient manufacturer is essential for securing a stable supply of critical materials. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging advanced biocatalytic technologies like the nitrilase mutants described in CN115851685A to deliver superior products. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volume requirements of even the largest global contracts. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 3-hydroxy-3-methylbutyric acid meets the highest international standards for safety and efficacy.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to our biocatalytic supply chain. We encourage you to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to validate the quality and reliability of our manufacturing capabilities before making any commitments. Let us collaborate to drive efficiency and sustainability in your supply chain together.
