Scalable Biocatalytic Production of Chiral (R)-3-Aminobutyric Acid for HIV Drug Intermediates
The pharmaceutical industry is currently witnessing a paradigm shift towards sustainable biocatalytic routes for the production of high-value chiral intermediates, particularly for antiretroviral therapies. Patent CN108866028B introduces a groundbreaking advancement in this domain by disclosing a novel amino lyase mutant protein capable of efficiently synthesizing (R)-3-aminobutyric acid, a critical building block for the HIV integrase inhibitor Dolutegravir. This technology addresses the longstanding challenges associated with traditional chemical synthesis, such as poor stereoselectivity and environmental toxicity, by leveraging precision enzyme engineering. The disclosed mutant proteins, derived from the Bacillus subtilis aspB gene, exhibit dramatically enhanced catalytic activity towards trans-2-butenoic acid, enabling a direct and highly selective ammoniation reaction. For procurement and R&D leaders, this represents a viable pathway to secure a reliable supply of optically pure intermediates while adhering to increasingly stringent green chemistry mandates. The ability to produce this compound with 100 percent stereoselectivity and high yield underscores the potential for this technology to redefine the manufacturing standards for complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of optically pure (R)-3-aminobutyric acid and its derivatives has been plagued by significant technical and economic hurdles inherent to both chemical and early-generation biological methods. Traditional chemical synthesis routes often involve multi-step processes that require harsh reaction conditions, including the use of severe temperatures and pressures, which inevitably lead to safety concerns and high energy consumption. Furthermore, these chemical pathways frequently rely on toxic organic solvents and heavy metal catalysts, generating substantial hazardous waste that complicates disposal and increases the overall environmental footprint of the manufacturing process. From a quality perspective, chemical methods often struggle to achieve high stereoselectivity, resulting in racemic mixtures that require costly and yield-reducing resolution steps to isolate the desired (R)-enantiomer. Even existing enzymatic approaches, such as those utilizing decarboxylases, have faced limitations due to the high cost of raw materials and relatively low utilization rates, making them less economically attractive for large-scale commercial production compared to the emerging mutant amino lyase technology.
The Novel Approach
The innovative approach detailed in the patent utilizes a specifically engineered amino lyase mutant that overcomes the activity barriers of wild-type enzymes, enabling the direct catalytic addition of ammonia to trans-2-butenoic acid. This biocatalytic strategy simplifies the synthetic route significantly by reducing the number of reaction steps and eliminating the need for protecting groups or complex chiral auxiliaries. The mutant enzyme demonstrates exceptional stability and activity under mild aqueous conditions, facilitating a cleaner reaction profile that minimizes the formation of by-products and simplifies downstream purification. By shifting the substrate base to inexpensive and readily available trans-2-butenoic acid, the process drastically reduces raw material costs while simultaneously improving the overall atom economy of the synthesis. This novel methodology not only ensures the production of the target molecule with 100 percent stereoselectivity but also aligns perfectly with modern principles of green chemistry, offering a scalable and environmentally benign alternative to legacy manufacturing processes for this vital pharmaceutical intermediate.
Mechanistic Insights into Mutant Amino Lyase Catalysis
The core of this technological breakthrough lies in the precise modification of the amino acid sequence of the aspB protein, where specific mutations at residues 187, 321, 324, and 326 fundamentally alter the enzyme's active site architecture to favor the asymmetric ammoniation of alpha,beta-unsaturated acids. Structural analysis suggests that substituting the native threonine at position 187 with residues like cysteine, valine, or alanine modifies the steric environment near the catalytic center, thereby enhancing substrate binding affinity for trans-2-butenoic acid. Similarly, mutations at positions 321, 324, and 326, involving replacements such as methionine to isoleucine or lysine to leucine, likely optimize the hydrogen bonding network and electrostatic interactions required to stabilize the transition state during the ammonia addition. These cumulative changes result in a mutant enzyme that possesses a significantly higher turnover number compared to the wild-type protein, which exhibits negligible activity towards this specific substrate. The engineering strategy effectively converts a non-natural activity into a highly efficient catalytic function, demonstrating the power of rational design and directed evolution in creating bespoke biocatalysts for industrial applications.
From an impurity control perspective, the high stereoselectivity of the mutant amino lyase is paramount for ensuring the quality of the final pharmaceutical intermediate. The enzyme's active site is configured to strictly recognize and process the substrate in a specific orientation that leads exclusively to the formation of the (R)-enantiomer, effectively preventing the generation of the unwanted (S)-isomer which could act as a difficult-to-remove impurity in downstream API synthesis. This intrinsic selectivity eliminates the need for chiral chromatography or diastereomeric crystallization steps that are typically required to upgrade optical purity in chemical synthesis. Furthermore, the use of a whole-cell biocatalyst system or purified enzyme in an aqueous buffer minimizes the risk of introducing organic solvent residues or heavy metal contaminants, which are common critical quality attributes (CQAs) monitored by regulatory agencies. The robustness of the biocatalytic process ensures consistent batch-to-batch reproducibility, providing R&D teams with a reliable platform for generating high-purity material that meets the stringent specifications required for clinical and commercial drug manufacturing.
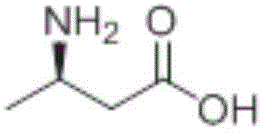
How to Synthesize (R)-3-Aminobutyric Acid Efficiently
The synthesis of (R)-3-aminobutyric acid using this patented technology involves a streamlined fermentation and biotransformation workflow that is amenable to scale-up. The process begins with the cultivation of recombinant Escherichia coli strains harboring the mutant aspB gene, where careful control of fermentation parameters such as dissolved oxygen and glucose feeding ensures high cell density and enzyme expression. Following harvest, the biomass is subjected to a transformation reaction with trans-2-butenoic acid in an aqueous ammonia solution, where the mutant enzyme catalyzes the stereoselective addition of the amino group. Detailed standardized synthesis steps see the guide below.
- Ferment recombinant E. coli BL21(DE3) harboring the mutant aspB gene in optimized medium with glucose feeding to achieve high cell density.
- Harvest cells and resuspend in substrate solution containing trans-2-butenoic acid and ammonia source at pH 9.0.
- Incubate at 50°C for 8 hours to complete the asymmetric ammoniation, followed by ultrafiltration and crystallization to isolate the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this mutant amino lyase technology offers compelling strategic advantages centered around cost optimization and supply security. The shift from complex chemical synthesis or expensive enzymatic decarboxylation to a direct ammoniation route using commodity chemicals like trans-2-butenoic acid fundamentally alters the cost structure of the intermediate. By removing the dependency on rare chiral starting materials and reducing the number of processing units required, manufacturers can achieve significant reductions in both variable and fixed production costs. Additionally, the aqueous nature of the reaction reduces the demand for volatile organic compounds (VOCs), leading to lower expenditures on solvent recovery systems and environmental compliance measures. This economic efficiency translates into a more competitive pricing model for the final API, allowing pharmaceutical companies to better manage their COGS (Cost of Goods Sold) in a price-sensitive market.
- Cost Reduction in Manufacturing: The elimination of expensive chiral reagents and toxic solvents directly lowers the raw material bill, while the high catalytic efficiency reduces the enzyme loading required per kilogram of product. This process intensification means that smaller reactors can produce the same output, or existing assets can yield significantly more product, driving down the capital depreciation cost per unit. Furthermore, the simplified downstream processing, which relies on crystallization rather than complex chromatographic separations, reduces utility consumption and labor hours associated with purification. These cumulative efficiencies create a leaner manufacturing operation that is more resilient to fluctuations in raw material pricing and energy costs.
- Enhanced Supply Chain Reliability: Utilizing trans-2-butenoic acid as a substrate leverages a mature and abundant global supply chain, mitigating the risks associated with sourcing specialized fine chemicals that may have limited vendors. The robustness of the fermentation process allows for flexible production scheduling, enabling manufacturers to ramp up output quickly in response to surges in demand for Dolutegravir or related antiviral medications. Moreover, the stability of the mutant enzyme ensures consistent performance over long storage periods, reducing the risk of production delays caused by biocatalyst degradation. This reliability is crucial for maintaining continuous API supply lines and avoiding stock-outs that could impact patient access to essential medicines.
- Scalability and Environmental Compliance: The biocatalytic process is inherently scalable, having been demonstrated effectively from laboratory shake flasks to multi-liter fermentors without loss of performance, which de-risks the technology transfer to commercial manufacturing sites. The absence of heavy metals and the reduction in hazardous waste generation simplify the permitting process and lower the liability associated with environmental regulations. This 'green' profile not only enhances the corporate sustainability metrics of the manufacturer but also future-proofs the supply chain against tightening environmental legislation. The ability to produce high-quality intermediates with a minimal environmental footprint is increasingly becoming a key differentiator in vendor selection processes for major pharmaceutical innovators.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this mutant amino lyase technology for the production of (R)-3-aminobutyric acid. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for evaluating the technology's feasibility. Understanding these details is essential for stakeholders assessing the potential integration of this biocatalytic route into their existing manufacturing portfolios.
Q: What is the stereoselectivity of the mutant amino lyase described in CN108866028B?
A: The patented mutant protein achieves 100% stereoselectivity, producing exclusively the (R)-enantiomer of 3-aminobutyric acid without detectable (S)-isomer contamination, which is critical for downstream API synthesis.
Q: How does this enzymatic method reduce costs compared to chemical synthesis?
A: By utilizing inexpensive trans-2-butenoic acid and ammonia instead of complex chiral pool starting materials, and eliminating the need for toxic organic solvents and heavy metal catalysts, the process significantly lowers raw material and waste disposal costs.
Q: Can this process be scaled for industrial manufacturing of Dolutegravir intermediates?
A: Yes, the patent demonstrates high-yield fermentation (OD600 > 20) and robust transformation conditions (50°C, pH 9.0) that are compatible with standard industrial bioreactors and downstream purification equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (R)-3-Aminobutyric Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of enzyme-engineered pathways like the one described in CN108866028B for the production of high-value chiral intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are successfully translated into robust industrial processes. Our facility is equipped with state-of-the-art fermentation suites and rigorous QC labs capable of meeting stringent purity specifications required for GMP manufacturing. We are committed to helping our clients navigate the complexities of biocatalytic synthesis, offering end-to-end support from strain optimization to commercial supply.
We invite you to engage with our technical procurement team to discuss how this advanced enzymatic route can be tailored to your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this biocatalytic method for your Dolutegravir intermediate requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with the highest level of technical confidence and commercial viability.
