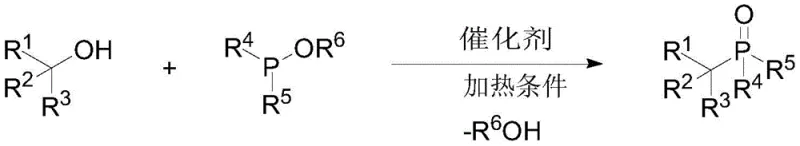
Advanced TMSOTf-Catalyzed Synthesis of Bulky Alkyl Substituted Phosphonite Diesters for Commercial Scale-up
The landscape of organophosphorus chemistry is undergoing a significant transformation driven by the urgent need for greener, more efficient synthetic routes capable of handling complex molecular architectures. A pivotal advancement in this domain is detailed in patent CN110922427A, which discloses a novel preparation method for large sterically hindered alkyl substituted phosphonite diesters. This technology represents a substantial leap forward from traditional methodologies by employing trimethylsilyl trifluoromethanesulfonate (TMSOTf) as a highly effective catalyst. The core innovation lies in the direct reaction between bulky sterically hindered alcohols and phosphite triesters, bypassing the need for toxic halogenated hydrocarbons that have long plagued the industry. For R&D directors and process chemists seeking reliable high-purity pharmaceutical intermediates, this patent offers a robust solution that combines operational simplicity with exceptional substrate tolerance, effectively filling a critical gap in the synthesis of complex C-P(O) bond-containing compounds.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of organophosphorus compounds containing C-P(O) bonds has relied heavily on the classical Arbuzov reaction, which involves the reaction of haloalkanes with triesters of phosphorous acid under high-temperature heating conditions. While this method has served as a cornerstone in laboratory and industrial settings, it suffers from profound disadvantages that limit its applicability in modern fine chemical manufacturing. Primarily, the requirement for highly toxic haloalkanes as raw materials poses significant safety and environmental hazards, necessitating rigorous containment and waste treatment protocols. Furthermore, the reaction inevitably generates equivalent amounts of volatile byproducts such as bromoethane, which are not only toxic but also prone to causing side reactions with the phosphite triester, leading to low atom economy and poor product selectivity. Most critically, the classical Arbuzov reaction is severely restricted to primary halogenated hydrocarbons; secondary and tertiary halides are largely unsuitable due to insurmountable steric hindrance effects, rendering the synthesis of bulky alkyl substituted phosphonites nearly impossible via this traditional route.
The Novel Approach
In stark contrast to these legacy constraints, the novel approach outlined in the patent data utilizes stable and low-toxicity bulky alcohols as the primary feedstock, catalyzed by TMSOTf to facilitate the direct formation of the C-P(O) bond. This methodology effectively circumvents the steric limitations of the classical Arbuzov reaction, allowing for the successful conversion of secondary and tertiary alcohols that were previously considered unreactive or low-yielding substrates. The reaction conditions are remarkably mild and operator-friendly, typically proceeding at temperatures around 100 °C without the absolute necessity for organic solvents, thereby aligning with the principles of green chemistry. By replacing hazardous halides with benign alcohols and eliminating the generation of toxic volatile halide byproducts, this new route offers a universally applicable platform for synthesizing a diverse array of sterically hindered phosphonite diesters with high efficiency and purity.
Mechanistic Insights into TMSOTf-Catalyzed Phosphonite Formation
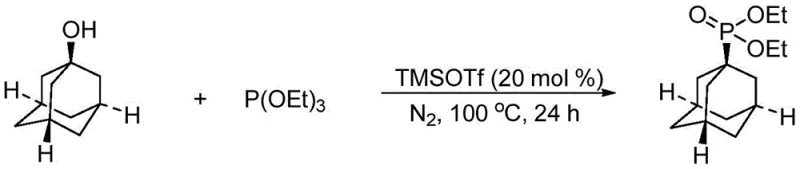
The mechanistic elegance of this transformation relies on the potent Lewis acidity of trimethylsilyl trifluoromethanesulfonate (TMSOTf), which activates the hydroxyl group of the bulky alcohol towards nucleophilic attack by the phosphite triester. Unlike transition metal catalysts that often require complex ligand systems and pose risks of metal leaching, TMSOTf operates through a clean silylation-mediated pathway that avoids the introduction of heavy metal contaminants entirely. This is of paramount importance for the production of high-purity OLED materials or API intermediates where trace metal specifications are exceptionally stringent. The catalyst loading is optimized between 2 to 30 mol%, with 20 mol% proving highly effective across a broad substrate scope, ensuring that the reaction proceeds to completion even with sterically demanding substrates like adamantanol or diphenylmethanol derivatives. The absence of transition metals not only simplifies the downstream purification process but also eliminates the need for expensive and time-consuming metal scavenging steps, directly contributing to process intensification.
Furthermore, the impurity profile of this reaction is significantly cleaner compared to halide-based methods. The primary byproduct is a small-molecule alcohol, such as ethanol or methanol, depending on the phosphite ester used, which is virtually non-toxic and easily removed during workup. This contrasts sharply with the complex mixtures of alkyl halides and phosphorus-containing side products often observed in thermal Arbuzov reactions. The high selectivity observed in examples such as the synthesis of diethyl 1-adamantyl phosphinate demonstrates the system's ability to tolerate extreme steric bulk without compromising yield or generating elimination byproducts. For supply chain managers, this predictable and clean reaction profile translates to higher batch consistency and reduced variability in raw material consumption, ensuring a stable supply of critical phosphonite building blocks for downstream coupling reactions.
How to Synthesize Bulky Alkyl Phosphonite Diesters Efficiently
Implementing this synthesis route in a production environment requires adherence to specific operational parameters to maximize yield and safety while maintaining the green advantages of the process. The procedure generally involves charging the bulky alcohol into a reactor, purging with an inert gas such as nitrogen to prevent oxidation, and then adding the phosphite triester and TMSOTf catalyst. The reaction is typically heated to 100 °C for approximately 24 hours, although optimization may allow for shorter times or lower temperatures depending on the specific substrate reactivity. Detailed standardized synthesis steps see the guide below.
- Charge the bulky sterically hindered alcohol compound into a reactor and purge with nitrogen three times to ensure an inert atmosphere.
- Add phosphite triester (2.0 to 3.0 equivalents) and TMSOTf catalyst (2 to 30 mol%, preferably 20 mol%) under nitrogen protection.
- Heat the reaction mixture to 100 °C and stir for 24 hours, then purify the product via column chromatography to obtain the diester.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this TMSOTf-catalyzed technology offers transformative benefits for procurement and supply chain operations, particularly in the context of cost reduction in pharmaceutical intermediate manufacturing. The elimination of toxic haloalkanes removes a major regulatory and disposal burden, significantly lowering the total cost of ownership associated with hazardous waste management. Moreover, the ability to operate under solvent-free conditions drastically reduces the volume of organic solvents required, leading to substantial savings in raw material procurement and solvent recovery infrastructure. The use of stable alcohol feedstocks, which are often more readily available and cheaper than their corresponding specialized halides, further enhances the economic viability of the process, making it an attractive option for large-scale commercial production.
- Cost Reduction in Manufacturing: The removal of transition metal catalysts from the process equation eliminates the need for costly metal scavengers and extensive purification protocols designed to meet strict residual metal limits. This streamlining of the downstream processing workflow results in significantly reduced operational expenditures and shorter cycle times per batch. Additionally, the high atom economy and the generation of non-toxic alcohol byproducts minimize waste treatment costs, allowing manufacturers to achieve a leaner and more cost-effective production model without compromising on product quality or yield.
- Enhanced Supply Chain Reliability: By utilizing widely available and stable alcohol starting materials instead of specialized and potentially supply-constrained halogenated hydrocarbons, the risk of raw material shortages is markedly decreased. The robustness of the catalytic system ensures consistent performance across different batches, reducing the likelihood of production delays caused by failed reactions or off-spec products. This reliability is crucial for maintaining continuous supply lines for critical intermediates, ensuring that downstream customers receive their orders on time and fostering long-term partnerships based on trust and dependability.
- Scalability and Environmental Compliance: The simplicity of the reaction setup, which does not require high-pressure equipment or extreme temperatures, facilitates easy scale-up from laboratory to pilot and eventually to full commercial production scales. The green nature of the process, characterized by low toxicity reagents and minimal waste generation, ensures compliance with increasingly stringent environmental regulations globally. This proactive approach to environmental stewardship not only mitigates regulatory risks but also enhances the corporate sustainability profile, appealing to eco-conscious stakeholders and customers in the global marketplace.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis technology. These answers are derived directly from the experimental data and technical disclosures within the patent documentation to provide accurate guidance for potential adopters.
Q: How does this method overcome the limitations of the classical Arbuzov reaction regarding steric hindrance?
A: Unlike the classical Arbuzov reaction which struggles with secondary and tertiary halides due to steric bulk, this TMSOTf-catalyzed method successfully utilizes bulky secondary and tertiary alcohols directly, enabling the synthesis of previously difficult-to-access sterically hindered phosphonite diesters.
Q: Are there any transition metal residues in the final phosphonite products?
A: No, the catalyst used is trimethylsilyl trifluoromethanesulfonate (TMSOTf), which is free of transition metals. This eliminates the risk of heavy metal contamination in the final product, a critical advantage for pharmaceutical applications.
Q: What are the environmental benefits of this solvent-free synthesis route?
A: The process can be conducted under solvent-free conditions, significantly reducing organic waste generation. Additionally, the byproduct is small-molecule alcohol like ethanol, which is low toxicity and easily removed, making the overall process greener and more environmentally compliant.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phosphonite Diester Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of adopting cutting-edge synthetic methodologies like the TMSOTf-catalyzed route to deliver superior value to our global clientele. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to industrial manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of phosphonite diester we produce adheres to the highest standards of quality required by the pharmaceutical and agrochemical industries.
We invite you to collaborate with us to leverage this innovative technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this green synthesis route can optimize your budget. Please contact us today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable and cost-effective supply chain for your critical organophosphorus intermediates.
