Scalable Copper-Catalyzed Synthesis of Phenyl Thiodiphenyl Phosphate for High-Purity Agrochemical Intermediates
Introduction to Advanced Phosphorothioate Synthesis
The chemical industry is currently witnessing a paradigm shift towards greener, more sustainable synthetic methodologies, particularly in the realm of organophosphorus compounds which serve as critical backbones for agrochemicals and pharmaceuticals. Patent CN110563760B introduces a groundbreaking preparation method for phenyl thiodiphenyl phosphate, a valuable intermediate, by leveraging a copper-catalyzed oxidative coupling strategy. This innovation addresses long-standing challenges associated with traditional phosphorylation techniques, specifically the reliance on hazardous phosphorus halides and harsh reaction conditions. By utilizing stable thiosulfonates and diphenylphosphine oxide in the presence of molecular oxygen from air, this technology offers a robust pathway that aligns with modern principles of atom economy and environmental stewardship. For R&D directors and process chemists, this represents a significant opportunity to streamline synthesis routes while enhancing the safety profile of manufacturing operations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of thiophosphate compounds has predominantly relied on the nucleophilic substitution reaction between phosphorus oxychloride derivatives (P(O)-Cl) and various nucleophiles such as alcohols or thiols. As illustrated in the general reaction scheme below, this traditional approach necessitates the use of highly toxic, corrosive, and moisture-sensitive phosphorus halide reagents.
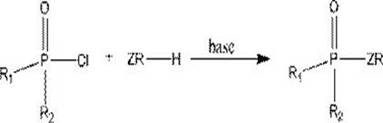
The inherent instability of P-Cl bonds requires stringent anhydrous conditions and specialized equipment to prevent hydrolysis, which generates corrosive hydrogen chloride gas as a byproduct. Furthermore, the low selectivity of these reactions often leads to complex impurity profiles that are difficult and costly to remove during downstream purification. From a supply chain perspective, the handling of such hazardous materials imposes severe regulatory burdens and increases the risk of production delays due to safety incidents. Consequently, there is an urgent industrial demand for alternative synthetic routes that can bypass these intrinsic limitations while maintaining high efficiency and product quality.
The Novel Approach
In stark contrast to the legacy methods, the technology disclosed in CN110563760B employs a transition metal-catalyzed cross-coupling strategy that fundamentally alters the reaction landscape. The core innovation lies in the direct coupling of a thiosulfonate (specifically phenyl thiobenzenesulfonate) with diphenylphosphine oxide, mediated by a廉价 copper salt catalyst under aerobic conditions. The reaction equation depicted below highlights the elegance of this transformation, where molecular oxygen serves as the terminal oxidant, eliminating the need for stoichiometric hazardous oxidants.
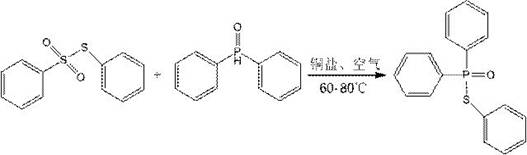
This novel approach operates under remarkably mild thermal conditions, typically between 60°C and 80°C, which significantly reduces energy consumption compared to high-temperature processes. The use of air as the oxidant not only lowers reagent costs but also simplifies the workup procedure, as the only byproduct is benign. For procurement managers, this translates to a drastic reduction in raw material volatility and a more predictable cost structure, as the starting materials are stable, commercially available solids rather than sensitive liquids requiring special storage. The method effectively decouples the synthesis of high-value phosphorothioates from the supply chain risks associated with hazardous chlorinating agents.
Mechanistic Insights into Copper-Catalyzed Oxidative Coupling
The mechanistic pathway of this transformation involves a sophisticated interplay between the copper catalyst and the phosphorus substrate, facilitating the formation of the P-S bond through an oxidative process. Initially, the copper species activates the P-H bond of the diphenylphosphine oxide, generating a reactive phosphorus-copper intermediate. Simultaneously, the thiosulfonate undergoes activation, likely involving the cleavage of the S-S or S-O bond to generate a sulfur-centered radical or electrophilic species. The presence of molecular oxygen is critical, as it regenerates the active copper catalyst from its reduced state, thereby sustaining the catalytic cycle without the accumulation of inactive metal species. This redox-neutral cycle ensures that the reaction proceeds with high turnover numbers, minimizing the required catalyst loading to as low as 10 mol%.
From an impurity control perspective, this mechanism offers distinct advantages over traditional nucleophilic substitutions. Since the reaction does not generate acidic byproducts like HCl, the risk of acid-catalyzed decomposition of the sensitive thiophosphate product is virtually eliminated. Furthermore, the high chemoselectivity of the copper catalyst ensures that side reactions, such as the oxidation of the sulfide to sulfone or the hydrolysis of the phosphate ester, are suppressed. This results in a cleaner crude reaction mixture, which simplifies the purification process—often requiring only standard silica gel chromatography to achieve high-purity specifications. For quality control teams, this means a more consistent impurity profile and reduced batch-to-batch variability, which is crucial for regulatory compliance in agrochemical and pharmaceutical applications.
How to Synthesize Phenyl Thiodiphenyl Phosphate Efficiently
The practical implementation of this synthesis route is designed for ease of operation, making it accessible for both laboratory-scale optimization and pilot-plant production. The process begins with the simple mixing of the solid starting materials in a common organic solvent, followed by the addition of the catalyst and heating under an open air atmosphere. Detailed standardized operating procedures regarding specific molar ratios, solvent volumes, and purification parameters are essential for reproducibility.
- Combine phenyl thiobenzenesulfonate and diphenylphosphine oxide in an organic solvent such as acetonitrile under an air atmosphere.
- Add a copper salt catalyst (e.g., CuI or CuCl2) and heat the reaction mixture to 60-80°C for approximately 12 hours to facilitate oxidative coupling.
- Remove the solvent via rotary evaporation, dissolve the residue in dichloromethane, and purify the final product using silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
The adoption of this copper-catalyzed methodology presents compelling economic and logistical benefits for organizations managing the supply of fine chemical intermediates. By shifting away from hazardous P-Cl reagents, companies can significantly reduce the overhead costs associated with safety compliance, waste disposal, and specialized storage infrastructure. The stability of the raw materials allows for bulk purchasing and long-term inventory management without the degradation risks typical of sensitive halides, thereby enhancing supply chain resilience against market fluctuations.
- Cost Reduction in Manufacturing: The utilization of inexpensive copper salts as catalysts, combined with the use of air as a free oxidant, drastically lowers the direct material costs of the synthesis. Furthermore, the elimination of expensive transition metal catalysts (like palladium or rhodium) and the avoidance of complex quenching steps for acidic byproducts lead to substantial operational savings. The simplified workup procedure reduces solvent consumption and labor hours, contributing to a leaner and more cost-effective manufacturing process overall.
- Enhanced Supply Chain Reliability: The reliance on stable, non-hazardous starting materials such as diphenylphosphine oxide and thiosulfonates mitigates the risk of supply disruptions caused by regulatory restrictions on toxic reagents. These precursors are widely available from multiple global suppliers, ensuring a robust and diversified sourcing strategy. Additionally, the mild reaction conditions reduce the wear and tear on reactor equipment, minimizing unplanned maintenance downtime and ensuring consistent production throughput to meet customer demand.
- Scalability and Environmental Compliance: The green nature of this synthesis, characterized by high atom economy and the absence of toxic emissions, aligns perfectly with increasingly stringent environmental regulations. Scaling this process from grams to tons does not require exponential increases in safety measures, as the thermal profile is manageable and the reagents are benign. This facilitates a smoother technology transfer from R&D to commercial production, allowing manufacturers to rapidly ramp up capacity for high-purity agrochemical intermediates without facing significant environmental permitting hurdles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on process feasibility and product quality.
Q: What are the primary advantages of this copper-catalyzed method over traditional P-Cl coupling?
A: This method eliminates the need for highly toxic and moisture-sensitive phosphorus halide reagents (P-Cl), replacing them with stable diphenylphosphine oxide. It utilizes cheap copper salts and air as a green oxidant, significantly reducing environmental hazards and operational complexity.
Q: What represents the optimal reaction conditions for maximum yield?
A: According to the patent data, the optimal conditions involve using acetonitrile as the solvent, CuI as the catalyst, and maintaining a temperature of 80°C under an air atmosphere, which achieves yields up to 86%.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is highly scalable due to the use of stable raw materials, mild reaction temperatures (60-80°C), and the avoidance of hazardous reagents, making it ideal for commercial manufacturing of agrochemical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phenyl Thiodiphenyl Phosphate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and sustainable synthesis routes in the modern chemical landscape. Our team of expert process chemists has extensively evaluated the copper-catalyzed oxidative coupling technology described in CN110563760B and confirmed its potential for delivering high-quality intermediates. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of materials that meet stringent purity specifications. Our rigorous QC labs are equipped to verify the absence of heavy metal residues and monitor impurity profiles, guaranteeing that every batch adheres to the highest industry standards.
We invite procurement leaders and R&D directors to collaborate with us to leverage this advanced manufacturing capability for their specific projects. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to contact us directly to obtain specific COA data and comprehensive route feasibility assessments, ensuring that your supply chain is optimized for both performance and profitability in the competitive agrochemical and pharmaceutical markets.
