Revolutionizing Pseudouridine Production: A Chemo-Enzymatic Strategy for High-Yield mRNA Vaccine Intermediates
Introduction to Next-Generation Nucleoside Manufacturing
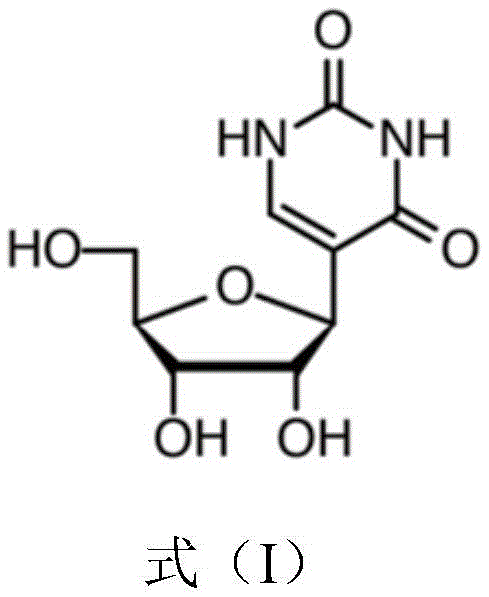
The rapid advancement of mRNA therapeutics and vaccines has placed an unprecedented demand on the supply chain for modified nucleosides, specifically pseudouridine. As a critical component in reducing the immunogenicity of synthetic mRNA while enhancing translation efficiency, the ability to produce this molecule at scale is a strategic priority for the global pharmaceutical industry. Patent CN114196715A introduces a groundbreaking chemo-enzymatic methodology that addresses the historical bottlenecks of pseudouridine synthesis. By leveraging a specifically engineered pseudouridine-5-phosphate glycosidase mutant, this technology enables the direct formation of the stable C-C glycosidic bond between the ribose moiety and the uracil base. This innovation represents a paradigm shift from labor-intensive chemical total synthesis to a streamlined, biocatalytic approach that aligns perfectly with modern green chemistry principles and industrial scalability requirements.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of pseudouridine has been plagued by significant technical and economic hurdles inherent to traditional chemical synthesis routes. Conventional methods typically require multi-step protection and deprotection strategies to manage the reactivity of the multiple hydroxyl groups on the ribose sugar, leading to extended reaction times and substantial material loss at each stage. Furthermore, these processes often rely on hazardous, flammable, and explosive reagents to facilitate the difficult C-glycosylation reaction, posing severe safety risks and environmental compliance challenges for large-scale manufacturing facilities. Alternatively, early biosynthetic attempts using engineered E. coli fermentation suffered from long culture periods and extremely complex downstream purification processes, where separating the target nucleoside from the cellular broth proved cost-prohibitive. These legacy limitations have resulted in high production costs and supply chain fragility, creating a critical need for a more robust and efficient manufacturing platform.
The Novel Approach
The chemo-enzymatic strategy disclosed in the patent data offers a decisive solution by decoupling the sugar preparation from the glycosylation step, optimizing each for maximum efficiency. In this novel workflow, ribose-5-phosphate is first generated through the controlled chemical hydrolysis of AMP, a readily available and cost-effective starting material. This activated sugar phosphate is then coupled with uracil using the highly active pseudouridine-5-phosphate glycosidase mutant, bypassing the need for harsh chemical catalysts. The process operates under mild physiological conditions (37°C, pH 7.5), which not only preserves the integrity of the sensitive nucleoside structure but also drastically simplifies the reactor engineering requirements. By achieving a conversion rate of 51% with the optimized triple mutant, this approach effectively doubles the substrate utilization compared to wild-type enzymes, demonstrating a clear path toward cost reduction in nucleoside manufacturing through superior catalytic performance.
Mechanistic Insights into Triple-Mutant Glycosidase Catalysis
The core intellectual property of this technology lies in the rational design of the pseudouridine-5-phosphate glycosidase enzyme, specifically the R146E/H156Q/K210A triple mutant. Through advanced molecular docking simulations with ribose-5-phosphate and uracil substrates, researchers identified key amino acid residues near the active site that limited catalytic turnover in the wild-type protein. The simultaneous mutation of arginine to glutamic acid at position 146, histidine to glutamine at position 156, and lysine to alanine at position 210 generates a profound synergistic effect that reshapes the enzyme's active pocket. This structural optimization facilitates a more favorable binding orientation for the substrates, lowering the activation energy required for the formation of the carbon-carbon bond between the C5 of uracil and the C1 of the ribose phosphate. The result is a biocatalyst that exhibits significantly enhanced stability and turnover frequency, allowing for high-density reaction setups that were previously unattainable with natural enzymes.
Beyond the catalytic mechanism, the process design incorporates sophisticated impurity control strategies essential for pharmaceutical grade output. A critical challenge in enzymatic synthesis is the removal of unreacted starting materials, particularly uracil, which can co-elute with the product during purification. The patent details a rigorous intermediate treatment protocol where the reaction mixture is heated to denature the protein catalyst, followed by centrifugation and nanofiltration. This nanofiltration step is pivotal, as it reduces the conductivity of the filtrate to 10 mS/cm and ensures that uracil content is undetectable before the final dephosphorylation step. By eliminating these impurities early in the downstream process, the final crystallization or chromatographic purification becomes far more efficient, ensuring the final pseudouridine product consistently achieves a purity of 98.3% without the need for excessive solvent consumption or repetitive recrystallization cycles.
How to Synthesize Pseudouridine Efficiently
The synthesis protocol outlined in the patent provides a reproducible framework for transitioning from laboratory benchtop to pilot plant operations. The process begins with the chemical preparation of the sugar donor, followed by the enzymatic coupling reaction in a controlled bioreactor environment, and concludes with a streamlined workup procedure. This sequence is designed to minimize unit operations while maximizing yield, making it an ideal candidate for tech transfer. For detailed operational parameters, stoichiometry, and specific equipment configurations required to replicate this high-efficiency synthesis, please refer to the standardized technical guide below.
- Hydrolyze AMP chemically to obtain ribose-5-phosphate, purifying via ion exchange resin to remove impurities.
- React ribose-5-phosphate with uracil in the presence of the pseudouridine-5-phosphate glycosidase mutant R146E/H156Q/K210A at 37°C and pH 7.5.
- Denature proteins, perform nanofiltration to remove salts and unreacted uracil, and finally dephosphorylate using NaOH to yield crude pseudouridine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this chemo-enzymatic platform translates into tangible strategic advantages regarding cost structure and supply security. The shift from multi-step organic synthesis to a concise enzymatic coupling fundamentally alters the cost of goods sold (COGS) profile by reducing the number of processing units and the volume of solvents required. The use of AMP as a precursor leverages an existing, mature supply chain for nucleotide raw materials, mitigating the risk of sourcing bottlenecks associated with exotic chemical intermediates. Furthermore, the high conversion efficiency of the R146E/H156Q/K210A mutant means that less raw material is wasted, directly contributing to substantial cost savings in raw material procurement without compromising on output quality.
- Cost Reduction in Manufacturing: The elimination of hazardous reagents and complex protection group chemistry significantly lowers the operational expenditure associated with waste disposal and safety compliance. By operating at ambient pressure and moderate temperatures, the process reduces energy consumption compared to high-temperature chemical reflux methods. Additionally, the simplified downstream processing, driven by the high specificity of the enzyme, reduces the load on purification columns and filtration systems, leading to lower maintenance costs and longer equipment lifecycles. These factors combine to create a leaner manufacturing model that is highly resilient to fluctuations in utility and consumable pricing.
- Enhanced Supply Chain Reliability: The robustness of the enzymatic reaction allows for flexible batch scheduling, as the biocatalyst can be produced and stored independently of the chemical synthesis steps. This modularity ensures that production can be ramped up quickly to meet surging demand for mRNA vaccine ingredients without the long lead times typical of fermentative processes. The reliance on stable, commercially available substrates like AMP and uracil further insulates the supply chain from the volatility often seen in the market for specialized fine chemical building blocks, ensuring consistent delivery timelines for downstream drug manufacturers.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively in 20L reactor systems with linear scalability potential to industrial volumes. The aqueous nature of the enzymatic step and the minimization of organic solvents align perfectly with increasingly strict environmental regulations regarding volatile organic compound (VOC) emissions. This green chemistry profile not only simplifies permitting for new manufacturing facilities but also enhances the corporate sustainability metrics of the final drug product, a factor of growing importance to end-users and regulatory bodies alike.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chemo-enzymatic pseudouridine synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the process capabilities and limitations for potential licensees and manufacturing partners.
Q: Why is the chemo-enzymatic method superior to traditional chemical synthesis for pseudouridine?
A: Traditional chemical synthesis involves long reaction sequences, hazardous reagents, and low overall yields due to complex protection and deprotection steps. The chemo-enzymatic route described in CN114196715A utilizes a highly specific enzyme mutant to form the C-C glycosidic bond directly, significantly reducing step count and eliminating the need for toxic solvents.
Q: How does the R146E/H156Q/K210A mutant improve production efficiency?
A: This triple-site mutation creates a synergistic effect that drastically enhances the catalytic activity of pseudouridine-5-phosphate glycosidase. Compared to the wild-type enzyme which shows only 25% conversion, this mutant achieves a conversion rate of 51%, effectively doubling the substrate utilization and reducing raw material waste.
Q: What are the purity specifications achievable with this new process?
A: The process incorporates a rigorous purification strategy including protein denaturation, centrifugation, and nanofiltration to remove conductivity and residual uracil before the final dephosphorylation step. This ensures the final pseudouridine product reaches a purity of 98.3%, meeting the stringent requirements for pharmaceutical grade nucleoside intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pseudouridine Supplier
As the global demand for modified nucleosides continues to surge, partnering with an experienced CDMO capable of navigating complex biocatalytic pathways is essential for maintaining competitive advantage. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of pseudouridine meets the exacting standards required for clinical and commercial mRNA applications. We understand the critical nature of these intermediates in the drug development timeline and are committed to delivering reliability at every stage of the partnership.
We invite you to engage with our technical procurement team to discuss how this innovative chemo-enzymatic route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a detailed projection of the economic benefits specific to your volume requirements. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions based on verified performance metrics and our proven track record in nucleoside manufacturing excellence.
