Advanced Biphenyl Tridentate Phosphite Ligands for High-Efficiency Hydroformylation Manufacturing
The chemical industry is currently witnessing a significant paradigm shift in hydroformylation catalysis, driven by the urgent need for more robust and selective ligands in the production of high-value aldehydes. Patent CN111848683A introduces a novel class of biphenyl tridentate phosphite ligands that address critical stability and activity limitations found in earlier generations of catalysts. Unlike traditional bidentate systems that often suffer from hydrolytic instability and lower regioselectivity, these new tridentate structures feature a rigid biphenyl backbone heavily substituted with tert-butyl groups, providing exceptional steric protection to the phosphorus centers. This structural innovation ensures that the ligand remains intact even under the demanding conditions of industrial hydroformylation, where trace moisture or oxygen can deactivate less robust catalysts. The general structure, as depicted in Formula (I), allows for tunable electronic properties through the variation of the R0 groups, enabling precise optimization for specific substrate profiles.
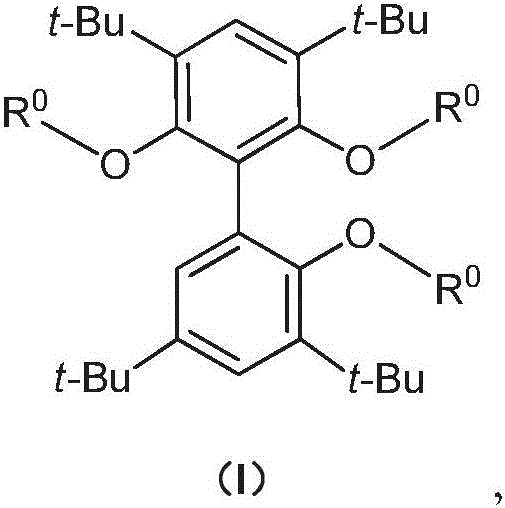
For procurement managers and supply chain directors, the implications of this technology extend far beyond mere catalytic performance; it represents a strategic opportunity for cost reduction in plasticizer intermediate manufacturing. The legacy technologies relying on propylene-derived butyraldehyde are facing regulatory headwinds due to the toxicity of downstream phthalates like DEHP, pushing the market towards higher molecular weight alternatives like DPHP derived from valeraldehyde. However, producing valeraldehyde efficiently from cheaper mixed C4 feedstocks has been historically challenging due to poor selectivity. The novel approach detailed in this patent overcomes these barriers by utilizing a tridentate coordination sphere that stabilizes the active rhodium species more effectively than bidentate analogues. This results in a catalytic system that not only tolerates the complex mixture of isomers in ether-after C4 streams but also drives the reaction towards the desired linear aldehyde with remarkable precision, thereby reducing downstream separation costs and waste generation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the hydroformylation of olefins has relied heavily on bidentate phosphine or phosphite ligands, such as the widely used Biphephos or triphenylphosphine (PPh3) systems. While these catalysts established the foundation of the oxo-process, they exhibit significant drawbacks when applied to modern feedstock challenges. Conventional bidentate ligands often lack the necessary thermal and hydrolytic stability required for continuous industrial operation, leading to ligand degradation and the formation of inactive rhodium black precipitates. Furthermore, their selectivity profile is frequently insufficient for complex mixed C4 streams, resulting in a lower ratio of linear to branched aldehydes (n/iso ratio). This poor selectivity necessitates energy-intensive distillation columns to separate the desired linear product from its branched isomers, drastically increasing the operational expenditure (OPEX). Additionally, many existing high-performance ligands are protected by expired or expiring patents from major chemical conglomerates, forcing manufacturers to pay substantial licensing fees or face legal risks, which erodes profit margins in a highly competitive commodity chemical market.
The Novel Approach
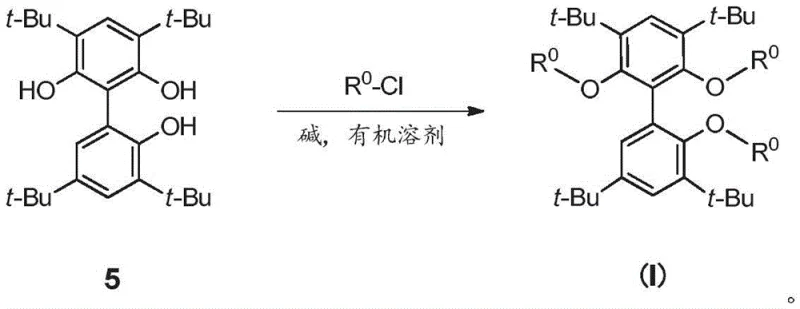
The innovative strategy presented in CN111848683A fundamentally reengineers the ligand architecture by introducing a third coordinating arm to the biphenyl scaffold. This tridentate design creates a more defined and rigid pocket around the metal center, which kinetically inhibits the formation of inactive species and thermodynamically favors the transition state leading to the linear product. The synthesis of the core scaffold, Compound 5, can be achieved through versatile routes such as lithium-halogen exchange followed by coupling, or oxidative coupling using copper catalysts, offering flexibility in raw material sourcing. As illustrated in the reaction scheme below, the final ligand assembly involves the reaction of the tri-hydroxy biphenyl precursor with specific chlorophosphine reagents (R0-Cl) in the presence of a strong base. This modular synthesis allows for the rapid generation of a library of ligands (L1-L33) to fine-tune performance for specific reactor configurations and feedstock compositions without altering the core manufacturing infrastructure.
Mechanistic Insights into Tridentate Phosphite-Rhodium Catalysis
The superior performance of these biphenyl tridentate ligands can be attributed to the unique electronic and steric environment they create around the rhodium atom. In the catalytic cycle, the tridentate ligand coordinates to the rhodium center, forming a highly stable complex that resists dissociation even at elevated temperatures ranging from 40°C to 80°C. The bulky tert-butyl groups positioned ortho to the oxygen linkages provide a 'shielding effect' that prevents nucleophilic attack by water or oxygen, which are common impurities in industrial gas streams. This stability is crucial for maintaining a consistent concentration of the active hydrido-rhodium-carbonyl species throughout the reaction duration. Moreover, the bite angle and flexibility of the tridentate system favor the insertion of the olefin into the Rh-H bond in a manner that places the metal at the terminal carbon, thus promoting the formation of the linear aldehyde over the branched isomer. This mechanistic advantage translates directly into higher process efficiency, as evidenced by conversion rates exceeding 90% and linear selectivity significantly outperforming benchmark bidentate ligands in comparative studies.
Impurity control is another critical aspect where this technology excels, particularly for R&D directors focused on product purity specifications. The robust nature of the ligand minimizes the leaching of rhodium metal into the product stream, a common issue that requires expensive scavenging steps in downstream processing. The specific substitution pattern on the biphenyl ring also suppresses side reactions such as olefin isomerization or hydrogenation, which can lead to the formation of saturated alkanes and reduce overall atom economy. By maintaining a tight coordination sphere, the catalyst ensures that the reaction pathway remains focused on hydroformylation, resulting in a cleaner crude product profile. This reduction in byproduct formation simplifies the purification train, allowing for the production of high-purity valeraldehyde suitable for sensitive downstream applications like the synthesis of specialty plasticizers or pharmaceutical intermediates without the need for extensive refining.
How to Synthesize Biphenyl Tridentate Phosphite Ligand Efficiently
The preparation of these advanced ligands is designed with industrial scalability in mind, utilizing readily available starting materials and standard unit operations. The process begins with the synthesis of the key intermediate, 2,2',6-trihydroxy-3,3',5,5'-tetra-tert-butyl-1,1'-biphenyl (Compound 5), which serves as the central scaffold. This precursor can be accessed via multiple pathways, including the lithiation of dimethoxybenzenes followed by coupling with bromo-derivatives, or through direct oxidative coupling of phenols using copper catalysts. Each route offers distinct advantages depending on the available infrastructure; for instance, the palladium-catalyzed Suzuki coupling route provides excellent control over regiochemistry, while the oxidative coupling method avoids the use of cryogenic conditions. Once Compound 5 is secured, the final ligation step involves deprotonation with n-butyllithium at low temperatures followed by the addition of the chlorophosphine electrophile. The detailed standardized synthesis steps for optimizing yield and purity are outlined in the guide below.
- Preparation of the core biphenyl scaffold (Compound 5) via lithiation-coupling or oxidative coupling of substituted phenols.
- Synthesis of chlorophosphine reagents (R0-Cl) from corresponding diols and PCl3 under reflux conditions.
- Coupling of Compound 5 with R0-Cl using a strong base like n-BuLi in THF at low temperatures to form the final tridentate ligand.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders, the adoption of this ligand technology offers a pathway to significantly reduced operational complexity and enhanced supply security. The synthesis relies on commodity chemicals such as phenols, alkyl halides, and standard solvents like tetrahydrofuran and dichloromethane, which are globally sourced and less susceptible to geopolitical supply disruptions compared to exotic ligand precursors. Furthermore, the high yields reported in the patent examples, such as the 95% yield in the demethylation step to form Compound 5, indicate a material-efficient process that minimizes waste disposal costs. The robustness of the catalyst also implies longer catalyst life and lower makeup rates, directly reducing the consumption of expensive rhodium metal, which is a major cost driver in hydroformylation plants. By switching to this domestic or non-restricted technology, manufacturers can avoid the hefty patent licensing fees associated with foreign-owned bidentate ligand technologies, resulting in substantial long-term cost savings.
- Cost Reduction in Manufacturing: The implementation of this tridentate ligand system eliminates the need for costly patent royalties often associated with proprietary bidentate ligands from major multinational corporations. The synthetic route is characterized by high-yielding steps and the use of inexpensive reagents, which collectively lower the cost of goods sold (COGS) for the catalyst itself. Additionally, the improved selectivity reduces the energy load on downstream distillation units, further driving down utility costs per ton of product.
- Enhanced Supply Chain Reliability: The raw materials required for the ligand synthesis, including substituted phenols and phosphorus chlorides, are produced by a wide network of chemical suppliers, ensuring a resilient supply chain. The process does not rely on single-source specialty monomers, mitigating the risk of production stoppages due to raw material shortages. This diversification allows procurement teams to negotiate better terms and maintain consistent inventory levels.
- Scalability and Environmental Compliance: The preparation methods described utilize standard reaction conditions and solvents that are easily managed in existing chemical manufacturing facilities, facilitating seamless scale-up from pilot to commercial production. The high stability of the ligand reduces the generation of phosphorus-containing waste streams, aligning with stricter environmental regulations and reducing the burden on wastewater treatment systems.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of biphenyl tridentate phosphite ligands in industrial hydroformylation processes. These insights are derived directly from the experimental data and embodiments disclosed in the patent literature, providing a factual basis for decision-making. Understanding these details is essential for evaluating the feasibility of integrating this technology into existing production lines.
Q: What are the stability advantages of this tridentate ligand compared to traditional bidentate ligands?
A: The biphenyl tridentate phosphite ligand described in CN111848683A exhibits exceptional stability against water and oxygen, preventing decomposition during storage and reaction, which is a common failure point for traditional bidentate phosphites like Biphephos.
Q: Can this ligand be used for mixed C4 hydroformylation?
A: Yes, the ligand is specifically optimized for mixed C4 streams containing 1-butene, cis-2-butene, and trans-2-butene, achieving high conversion rates above 90% and superior linear-to-branched aldehyde ratios.
Q: Is the synthesis scalable for commercial production?
A: The preparation method utilizes standard organic solvents like THF and dichloromethane with high-yield steps (up to 95% in demethylation), making it highly suitable for scale-up from kilogram to multi-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Biphenyl Tridentate Phosphite Ligand Supplier
As the global demand for high-performance plasticizers and fine chemicals continues to grow, the need for efficient and sustainable catalytic solutions has never been more critical. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging deep expertise in ligand design and homogeneous catalysis to deliver superior products. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent quality regardless of order volume. Our state-of-the-art rigorous QC labs and stringent purity specifications guarantee that every batch of ligand meets the exacting standards required for sensitive rhodium-catalyzed processes, minimizing the risk of catalyst deactivation in your plant.
We invite you to collaborate with us to optimize your hydroformylation operations and achieve significant competitive advantages. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific feedstock and reactor configuration. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you transition to a more profitable and sustainable manufacturing future.
