Advanced Biphenyl Tetradentate Phosphonite Ligands for High-Efficiency Hydroformylation Manufacturing
Advanced Biphenyl Tetradentate Phosphonite Ligands for High-Efficiency Hydroformylation Manufacturing
The global demand for high-performance plasticizers has driven a significant shift away from traditional phthalates like DEHP towards safer, higher molecular weight alternatives such as DPHP. Central to this transition is the efficiency of the hydroformylation process used to generate valeraldehyde precursors from mixed butenes. Patent CN111909209A introduces a groundbreaking preparation method for a novel biphenyl tetradentate phosphonite ligand, specifically 2,2',6,6'-tetra[(1,1'-biphenyl-2,2'-diyl)phosphonite]-3,3',5,5'-tetra-tert-butyl-1,1'-biphenyl. This innovation addresses critical limitations in current catalytic systems by offering superior stability and regioselectivity. As a leading entity in fine chemical manufacturing, we recognize that the structural integrity of such ligands is paramount for maintaining continuous industrial operations. The general structure of this advanced ligand class, designated as Formula I, allows for versatile substitution patterns that can be tuned for specific catalytic requirements.
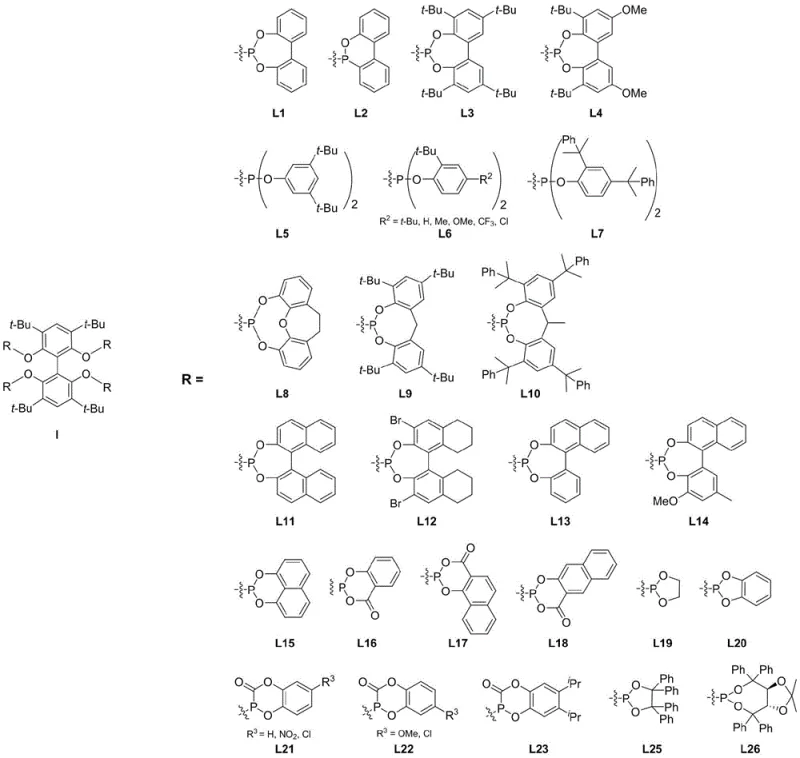
The strategic importance of this technology lies in its ability to facilitate the production of linear aldehydes with unprecedented precision. In the context of modern industrial chemistry, where feedstock costs fluctuate and regulatory pressures mount, the deployment of robust catalysts is not merely a technical preference but a commercial necessity. This patent outlines a synthesis pathway that is not only chemically elegant but also practically viable for scale-up, ensuring that downstream manufacturers can secure a reliable ligand supplier capable of meeting rigorous purity specifications. By leveraging this intellectual property, we can offer partners a distinct competitive edge in the production of next-generation plasticizers and specialty chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the hydroformylation industry has relied heavily on monodentate ligands like triphenylphosphine (PPh3) or bidentate systems such as Biphephos, famously utilized by major chemical corporations. While these systems established the foundation for industrial aldehyde production, they suffer from inherent instability issues that plague long-term operational efficiency. Specifically, conventional bidentate phosphonite ligands are notoriously susceptible to hydrolysis and acidolysis under reaction conditions, leading to the degradation of the catalyst complex. This degradation manifests physically as the formation of solid byproducts that tend to block pipelines and reactors, necessitating frequent shutdowns for maintenance and cleaning. Furthermore, the reliance on these older ligand technologies often requires the sporadic addition of fresh ligand to maintain catalytic activity, driving up operational expenditures and complicating process control. Additionally, traditional processes often favor pure 1-butene feedstocks, which are significantly more expensive than mixed butene streams derived from steam cracking or fluid catalytic cracking units.
The Novel Approach
The methodology disclosed in CN111909209A represents a paradigm shift by introducing a tetradentate architecture that fundamentally alters the stability profile of the catalyst system. Unlike their bidentate counterparts, these novel ligands possess a rigid, sterically hindered backbone that protects the phosphorus centers from nucleophilic attack by water or acidic species. This structural robustness translates directly into enhanced catalyst longevity and consistent performance over extended run times. The synthetic route utilizes a modular approach, starting from simple dimethoxybenzene precursors and building complexity through controlled oxidative coupling and functionalization. This allows for the precise installation of tert-butyl groups, which provide the necessary steric bulk to enforce high normal-to-iso regioselectivity during the hydroformylation of mixed or etherified C4 streams. By enabling the use of cheaper mixed butene feedstocks while achieving superior conversion rates, this novel approach offers substantial cost reduction in hydroformylation manufacturing without compromising on product quality.
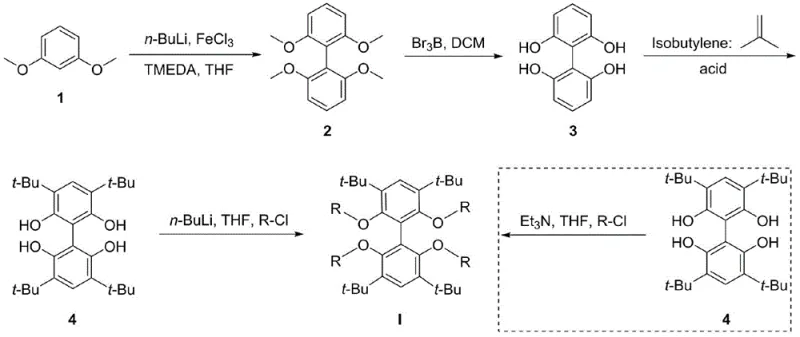
Mechanistic Insights into FeCl3-Catalyzed Oxidative Coupling and Phosphitylation
The core of this synthesis strategy relies on a sophisticated sequence of organometallic transformations, beginning with the construction of the biphenyl scaffold. The initial step involves an iron-mediated oxidative coupling of 1,3-dimethoxybenzene, utilizing n-butyllithium and ferric chloride in the presence of TMEDA. This reaction is critical as it establishes the central carbon-carbon bond connecting the two aromatic rings, setting the stage for the subsequent tetradentate geometry. The mechanism likely proceeds through the generation of aryl lithium species which are then oxidatively coupled by the iron salt, a process that must be carefully controlled at cryogenic temperatures (-78°C) to prevent over-lithiation or polymerization side reactions. The efficiency of this step determines the overall yield of the backbone, and the patent reports an impressive 80% yield, indicating a highly optimized protocol that minimizes waste and maximizes atom economy.
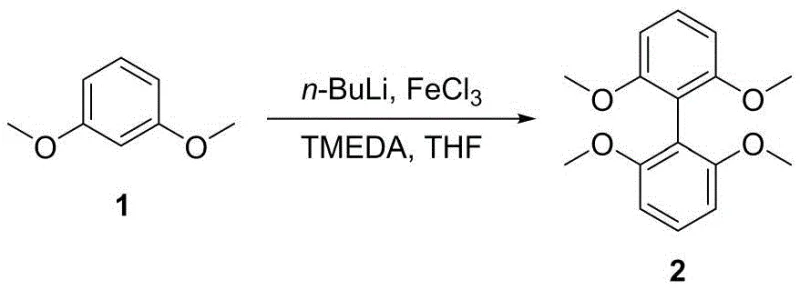
Following the backbone construction, the synthesis proceeds through demethylation and bulky group installation before the final phosphitylation. The conversion of methoxy groups to hydroxyls using boron tribromide is a standard yet crucial deprotection step that activates the phenols for subsequent alkylation. The introduction of tert-butyl groups via acid-catalyzed reaction with isobutylene is particularly noteworthy, as these groups lock the conformation of the ligand, preventing unfavorable rotations that could diminish catalytic selectivity. The final assembly involves the reaction of the tetrahydroxy intermediate with 1,1'-dioxyphosphorochloridite. This phosphitylation step forms the P-O-C linkages that define the ligand's coordination sphere. The use of a chlorophosphite intermediate ensures high reactivity, allowing the formation of the P-O bonds under relatively mild conditions. The resulting tetradentate structure creates a well-defined pocket for the rhodium or other transition metal center, enforcing the electronic and steric environment required for high linear aldehyde selectivity.
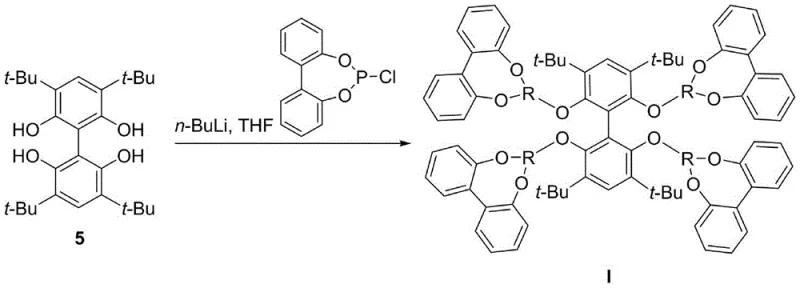
How to Synthesize Biphenyl Tetradentate Phosphonite Ligand Efficiently
The preparation of these high-value ligands requires strict adherence to anhydrous and anaerobic conditions to preserve the integrity of the reactive intermediates. The process begins with the lithiation of the aromatic precursor, followed by the sequential installation of functional groups that dictate the final catalyst performance. Each step, from the oxidative coupling to the final purification via column chromatography, is designed to maximize yield while ensuring the removal of impurities that could poison the downstream hydroformylation catalyst. For detailed operational parameters, including exact stoichiometry, temperature ramps, and workup procedures, please refer to the standardized synthesis guide below which encapsulates the critical process controls identified in the patent literature.
- Perform oxidative coupling of dimethoxybenzene using n-BuLi and FeCl3 to form the biphenyl backbone.
- Execute demethylation using BBr3 followed by tert-butylation with isobutylene to introduce steric bulk.
- React the tetrahydroxy intermediate with 1,1'-dioxyphosphorochloridite under inert atmosphere to yield the final ligand.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel ligand technology presents a compelling value proposition centered on operational stability and raw material flexibility. Traditional ligand systems often introduce hidden costs associated with catalyst degradation, pipeline fouling, and the need for premium feedstocks. By transitioning to this tetradentate system, manufacturers can mitigate these risks, leading to a more predictable and efficient production schedule. The robustness of the ligand means fewer interruptions for maintenance and a longer lifecycle for the catalytic system, which directly correlates to improved asset utilization. Furthermore, the ability to effectively utilize mixed butene streams, which are typically less expensive and more abundant than purified 1-butene, provides a significant buffer against feedstock price volatility. This flexibility is crucial for maintaining margin stability in a fluctuating petrochemical market.
- Cost Reduction in Manufacturing: The implementation of this ligand eliminates the need for frequent catalyst replenishment and reduces downtime associated with pipeline blockages caused by ligand hydrolysis. By enabling the use of lower-cost mixed butene feedstocks while maintaining high selectivity for linear aldehydes, the overall cost of goods sold for downstream plasticizers like DPHP is significantly optimized. The high yield of the synthesis itself also contributes to a more favorable cost structure for the ligand procurement, avoiding the premiums often charged for specialized, low-volume catalytic additives.
- Enhanced Supply Chain Reliability: The synthetic route described relies on commodity chemicals such as isobutylene, dimethoxybenzene, and phosphorus trichloride, all of which are widely available in the global chemical market. This reduces the risk of supply bottlenecks that often plague proprietary ligand supply chains dependent on exotic or single-source precursors. The scalability of the process ensures that production can be ramped up to meet increasing demand without requiring complex re-engineering of the manufacturing facility, thereby securing a continuous supply for long-term contracts.
- Scalability and Environmental Compliance: The process is designed with industrial scale-up in mind, utilizing standard unit operations like extraction, distillation, and crystallization. The high atom economy and reduced waste generation associated with the stable catalyst system align with modern green chemistry principles, simplifying environmental compliance and waste treatment protocols. The elimination of toxic byproducts and the reduction in solvent usage due to higher catalyst efficiency further enhance the sustainability profile of the manufacturing process, appealing to environmentally conscious stakeholders.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this ligand system is essential for making informed procurement and R&D decisions. The following questions address common inquiries regarding the stability, performance, and scalability of the biphenyl tetradentate phosphonite ligands. These answers are derived directly from the experimental data and technical disclosures within the patent, providing a factual basis for evaluating the technology's fit within your existing process infrastructure.
Q: What are the stability advantages of this tetradentate ligand compared to Biphephos?
A: Unlike Biphephos, which is prone to hydrolysis and acidolysis leading to pipeline blockage, this novel tetradentate ligand exhibits exceptional stability against water and oxygen, ensuring consistent catalytic activity without sporadic replenishment.
Q: How does this ligand impact the n/iso ratio in hydroformylation?
A: The specific steric and electronic configuration of the tetradentate structure significantly enhances the normal-to-iso aldehyde ratio in mixed butene hydroformylation, superior to traditional monodentate or bidentate systems.
Q: Is the synthesis scalable for industrial production?
A: Yes, the patent describes a robust synthetic route using readily available reagents like isobutylene and standard halogenation agents, designed specifically for easy amplification and large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Biphenyl Tetradentate Phosphonite Ligand Supplier
At NINGBO INNO PHARMCHEM, we understand that the transition to advanced catalytic systems requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising laboratory results of CN111909209A can be seamlessly translated into industrial reality. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of ligand meets the exacting standards required for high-performance hydroformylation. Our commitment to quality ensures that your catalytic processes run smoothly, maximizing yield and minimizing operational disruptions.
We invite you to engage with our technical procurement team to discuss how this innovative ligand technology can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits specific to your operation. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make data-driven decisions that enhance your competitive position in the global market for specialty chemicals and plasticizers.
