Advanced Synthesis of Biphenyl Pyrogallol Intermediates for High-Performance Hydroformylation Ligands
Introduction to Next-Generation Ligand Precursors
The chemical industry's relentless pursuit of efficiency in catalytic processes has brought significant attention to the synthesis of advanced ligand precursors, specifically within the realm of hydroformylation technologies. Patent CN111747827A introduces a groundbreaking approach to producing novel pyrogallol compounds characterized by a biphenyl skeleton, which serve as indispensable intermediates for tridentate phosphite ligands. These ligands play a pivotal role in optimizing hydroformylation reactions, where olefins are converted into aldehydes with high selectivity and activity. The disclosed technology addresses critical bottlenecks in traditional synthetic routes by offering a direct oxidative coupling method that bypasses the need for complex pre-functionalization steps. This innovation not only streamlines the manufacturing workflow but also aligns with modern green chemistry principles by utilizing abundant metal catalysts and molecular oxygen. For R&D directors and procurement specialists, understanding the structural versatility and synthetic accessibility of these compounds is key to securing a reliable supply chain for high-performance catalytic systems.
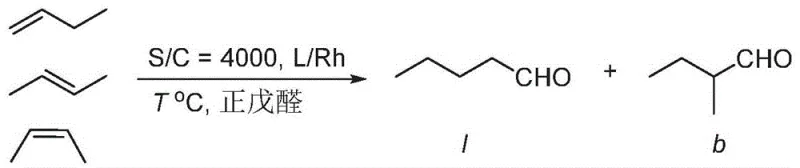
The strategic value of these intermediates lies in their ability to form robust coordination complexes with rhodium, enhancing the linear-to-branched ratio of aldehyde products in industrial applications. By leveraging the synthetic methodologies outlined in the patent, manufacturers can achieve substantial improvements in process economics while maintaining stringent purity specifications required for downstream catalytic performance. This report delves into the mechanistic nuances, comparative advantages, and commercial scalability of this technology, providing a comprehensive roadmap for integrating these advanced intermediates into existing production frameworks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of biphenyl scaffolds for ligand synthesis has relied heavily on transition-metal-catalyzed cross-coupling reactions such as Suzuki, Heck, or Kumada couplings. While effective, these classical methods impose significant logistical and economic burdens on the manufacturing process. They necessitate the prior preparation of halogenated aromatic precursors and organometallic reagents, which adds multiple synthetic steps and increases the overall material cost. Furthermore, the reliance on precious metal catalysts like palladium introduces challenges related to residual metal contamination, requiring extensive purification protocols to meet pharmaceutical and fine chemical standards. The generation of stoichiometric amounts of metal salts and halide waste also complicates environmental compliance and waste disposal strategies. Additionally, alternative routes involving lithiation require cryogenic conditions and strict anhydrous environments, posing safety risks and demanding specialized infrastructure that limits scalability. These cumulative inefficiencies result in longer lead times and higher operational expenditures, making traditional routes less attractive for large-scale commercial production.
The Novel Approach
In stark contrast, the oxidative coupling methodology presented in the patent offers a paradigm shift by enabling the direct formation of carbon-carbon bonds between phenolic units without pre-activation. This one-step process utilizes inexpensive copper salts or complexes combined with organic bases and molecular oxygen or other benign oxidants. By eliminating the need for halogenated starting materials and precious metal catalysts, the novel approach drastically reduces the raw material inventory and simplifies the reaction workflow. The method demonstrates excellent functional group tolerance and regioselectivity, particularly when bulky substituents like tert-butyl groups are employed to direct the coupling position. This directness translates to higher atom economy and a significantly reduced environmental footprint. Moreover, the reaction conditions are mild, often proceeding at temperatures between 30°C and 60°C, which lowers energy consumption. For supply chain managers, this translates to a more resilient production model with fewer dependency points on volatile precursor markets and a streamlined path from raw materials to finished intermediates.
Mechanistic Insights into Cu-Catalyzed Oxidative Coupling
The core of this innovative synthesis lies in the copper-catalyzed oxidative coupling mechanism, which facilitates the dimerization of phenolic substrates through a radical-mediated pathway. The catalytic cycle typically initiates with the coordination of the copper species, such as CuCl or Cu(OTf)2, with bidentate nitrogen ligands like TMEDA to form an active complex. Upon exposure to oxygen, this complex undergoes oxidation to generate reactive copper-oxygen species capable of abstracting hydrogen atoms from the phenolic hydroxyl groups. This abstraction generates phenoxy radicals, which subsequently couple at the ortho-positions relative to the hydroxyl groups to form the biphenyl linkage. The presence of bulky substituents, such as tert-butyl groups at the 3 and 5 positions, plays a crucial steric role in directing the coupling to the desired 1,1'-position, thereby minimizing the formation of unwanted isomers or polymeric byproducts. Understanding this mechanistic nuance is vital for R&D teams aiming to optimize reaction parameters for specific substrate variations.
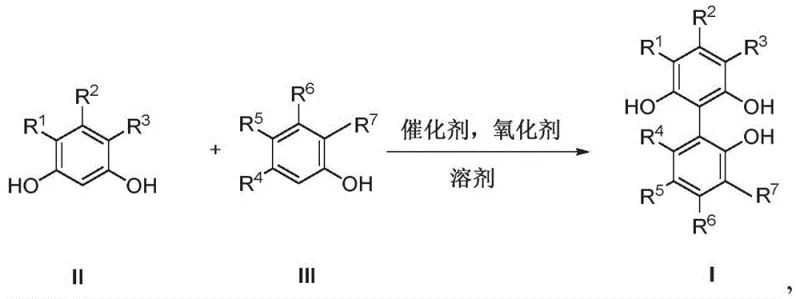
Furthermore, the choice of oxidant and solvent system significantly influences the reaction kinetics and product distribution. The patent highlights the efficacy of using molecular oxygen or air as the terminal oxidant, which is not only cost-effective but also produces water as the sole byproduct, enhancing the green profile of the process. In acidic variants, oxidants like potassium dichromate or iron chloride can be employed, though the copper-catalyzed aerobic system offers superior selectivity and milder conditions. The mechanism also accounts for the regeneration of the active copper catalyst, ensuring that low catalyst loadings (often 2-4 mol%) are sufficient to drive the reaction to completion. This catalytic efficiency is a key factor in reducing the overall cost of goods sold (COGS) and minimizing the burden on downstream purification units. By controlling the oxygen flow rate and maintaining optimal temperature profiles, manufacturers can suppress side reactions and maximize the yield of the target biphenyl pyrogallol intermediate.
How to Synthesize Biphenyl Pyrogallol Efficiently
The practical implementation of this oxidative coupling technology involves a straightforward protocol that is amenable to both laboratory and pilot-scale operations. The process begins with the preparation of a catalyst solution by dissolving the copper salt and organic base in a suitable solvent such as methanol or acetonitrile under an inert atmosphere. Oxygen is then introduced to activate the catalyst, signaled by a distinct color change, before the phenolic substrates are added dropwise. This controlled addition helps manage the exothermic nature of the oxidation and ensures consistent mixing. Following the reaction period, which typically ranges from 24 to 72 hours depending on the specific substrate and conditions, the product can be isolated via filtration or crystallization. The simplicity of the workup procedure, often requiring only solvent removal and recrystallization, underscores the operational ease of this method. For detailed standard operating procedures and specific parameter optimizations, please refer to the guide below.
- Prepare the catalyst system by dissolving copper salts and organic bases like TMEDA in solvent under inert atmosphere.
- Introduce oxygen or air to saturate the catalyst solution, indicated by a color change from light to dark.
- Add phenolic substrates dropwise while maintaining oxygen flow and controlled temperature to achieve coupling yields up to 60%.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this oxidative coupling technology offers transformative benefits for procurement and supply chain operations within the fine chemical sector. The most immediate impact is seen in cost reduction in fine chemical manufacturing, driven by the substitution of expensive palladium catalysts with abundant copper salts and the elimination of costly halogenated precursors. This shift not only lowers direct material costs but also reduces the complexity of the supply chain by decreasing the number of distinct raw materials that need to be sourced and qualified. The simplified synthetic route means fewer unit operations, which translates to reduced labor costs, lower energy consumption, and decreased equipment occupancy time. For procurement managers, this equates to a more predictable pricing structure and reduced exposure to the volatility of precious metal markets. Additionally, the high atom economy of the process minimizes waste generation, leading to significant savings in waste disposal fees and environmental compliance costs.
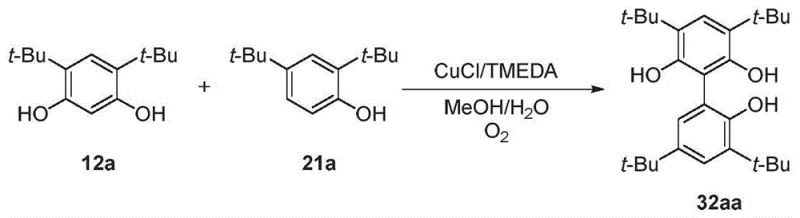
Enhanced supply chain reliability is another critical advantage, as the process relies on commercially available and stable starting materials like substituted phenols and resorcinols. Unlike sensitive organometallic reagents that require special handling and storage, these substrates are robust and easy to transport, reducing the risk of supply disruptions. The scalability of the process has been validated at the kilogram level using standard glass-lined or stainless steel reactors, demonstrating that the technology can be seamlessly transferred from R&D to commercial production without significant re-engineering. This scalability ensures that suppliers can respond quickly to fluctuating market demands, reducing lead time for high-purity intermediates. Furthermore, the environmental benefits of using oxygen as an oxidant and generating minimal hazardous waste align with increasingly stringent global sustainability regulations, future-proofing the supply chain against regulatory changes. These factors collectively contribute to a more resilient, cost-effective, and sustainable sourcing strategy for critical catalytic intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these biphenyl pyrogallol compounds. The answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for industry professionals. These insights cover aspects ranging from reaction optimization to downstream processing, helping stakeholders make informed decisions about integrating this technology into their operations. Understanding these details is essential for maximizing the value proposition of this novel synthetic route.
Q: What are the primary advantages of oxidative coupling over traditional Suzuki coupling for these intermediates?
A: Oxidative coupling eliminates the need for pre-functionalized halogenated precursors and expensive palladium catalysts. It reduces the step count significantly, improves atom economy, and utilizes inexpensive copper catalysts with oxygen as the terminal oxidant, leading to lower production costs and simplified waste management.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the patent explicitly demonstrates kilogram-scale preparation in a 20L reactor. The process uses standard equipment like glass-jacketed reactors with mechanical stirring and temperature control, confirming its viability for commercial scale-up without requiring specialized cryogenic or high-pressure infrastructure.
Q: What is the specific application of the resulting pyrogallol compounds?
A: These biphenyl pyrogallol compounds serve as critical precursors for synthesizing tridentate phosphite ligands. These ligands are essential in rhodium-catalyzed hydroformylation reactions, particularly for converting mixed C4 olefins into valuable aldehydes with high linear-to-branched ratios.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrogallol Compound Supplier
As the demand for high-performance ligands in hydroformylation and other catalytic applications continues to grow, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM becomes a strategic imperative. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our state-of-the-art facilities are equipped to handle the specific requirements of oxidative coupling chemistries, including rigorous safety protocols for oxygen usage and advanced purification capabilities to meet stringent purity specifications. Our rigorous QC labs employ sophisticated analytical techniques to verify the identity and purity of every batch, guaranteeing that the intermediates you receive are perfectly suited for subsequent ligand synthesis and catalytic testing. We understand the critical nature of these materials in your value chain and are committed to delivering quality that supports your innovation goals.
We invite you to engage with our technical procurement team to discuss how we can tailor our manufacturing capabilities to your specific project requirements. Whether you need a Customized Cost-Saving Analysis for transitioning from traditional coupling methods or require specific COA data and route feasibility assessments for new derivatives, we are ready to provide the support you need. By leveraging our expertise in process development and scale-up, we can help you accelerate your time-to-market while optimizing your production costs. Contact us today to explore how our advanced synthesis platforms can enhance your supply chain resilience and drive your catalytic applications forward.
