Revolutionizing LCZ696 Intermediate Production: A Deep Dive into High-Efficiency Transaminase Biocatalysis
The global pharmaceutical landscape is witnessing a sustained surge in demand for angiotensin receptor-neprilysin inhibitors (ARNIs), particularly for the blockbuster heart failure medication LCZ696 (Entresto). At the heart of this supply chain lies the critical chiral building block, D-4,4'-biphenylalanine, which serves as the key intermediate for synthesizing Sacubitril. As procurement strategies evolve towards greener and more cost-effective methodologies, the industry is actively seeking alternatives to traditional chemical synthesis. Patent CN116004562A introduces a groundbreaking biocatalytic solution that addresses these exact pain points. By leveraging a specifically engineered quadruple transaminase mutant derived from Mycolicibacterium agri, this technology achieves a staggering 30-fold increase in catalytic efficiency compared to wild-type enzymes. This report analyzes the technical merits of this innovation, demonstrating how it offers a reliable D-4,4'-biphenylalanine supplier pathway that aligns with modern sustainability goals and rigorous quality standards required by top-tier pharmaceutical manufacturers.
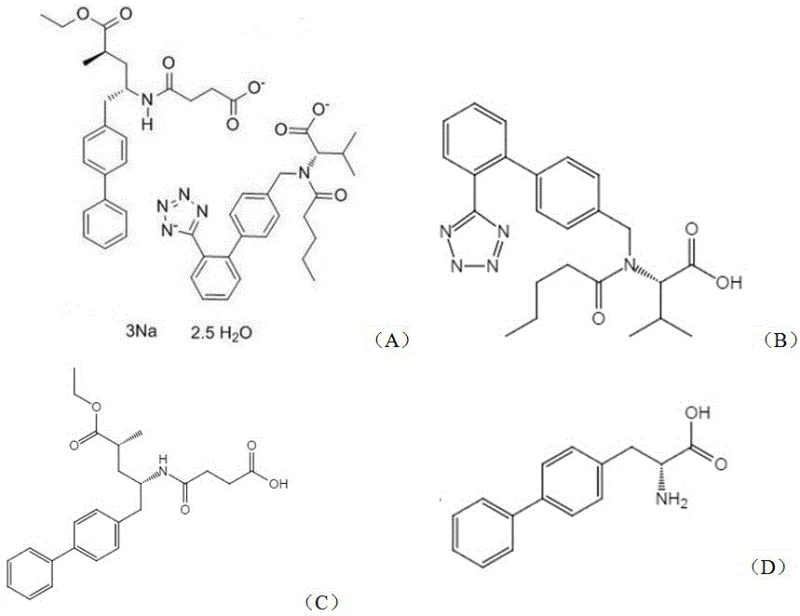
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of chiral amino acids like D-4,4'-biphenylalanine has relied heavily on classical organic synthesis routes, which are fraught with significant operational and environmental drawbacks. Traditional chemical methods often involve the use of hazardous reagents, such as heavy metal catalysts for asymmetric hydrogenation or toxic cyanide sources for Strecker synthesis, creating substantial waste disposal challenges and safety risks for plant operators. Furthermore, achieving high enantiomeric excess (e.e.) through chemical means frequently necessitates multi-step resolution processes, such as diastereomeric salt formation or chiral chromatography, which drastically reduce overall yield and inflate production costs. Even earlier generations of biocatalytic approaches, while greener, suffered from sluggish reaction kinetics; for instance, prior art transaminases required reaction times extending up to 18 hours to reach near-complete conversion, limiting reactor throughput and increasing energy consumption for prolonged agitation and temperature control. These inefficiencies create a bottleneck in the supply chain, making it difficult to meet the escalating global demand for Sacubitril without significant capital investment in larger reactor volumes.
The Novel Approach
The technological breakthrough detailed in CN116004562A circumvents these legacy issues through the application of protein engineering and directed evolution. By identifying specific mutation sites—R60G, D72A, K196N, and Q199L—within the transaminase structure, researchers have created a biocatalyst with fundamentally altered kinetic properties. This novel approach utilizes an asymmetric transamination reaction where 4,4'-biphenylpyruvic acid is converted directly into the desired D-enantiomer using isopropylamine as the amine donor. The beauty of this system lies in its thermodynamic driving force; the byproduct, acetone, can be easily removed or managed to push the equilibrium towards product formation. Unlike the wild-type enzyme which struggles with substrate load and speed, this mutant maintains robust activity even at lower enzyme loadings (as low as 0.01 wt%), enabling rapid conversion within 4 to 8 hours. This shift from a slow, resource-intensive process to a rapid, high-turnover biocatalytic cycle represents a paradigm shift in cost reduction in pharmaceutical intermediate manufacturing, allowing producers to achieve higher output with existing infrastructure.
Mechanistic Insights into Quadruple Mutant Transaminase Catalysis
To fully appreciate the value proposition for R&D directors, one must examine the mechanistic underpinnings of this enhanced catalysis. The reaction proceeds via a Ping-Pong Bi-Bi mechanism typical of pyridoxal 5'-phosphate (PLP) dependent enzymes. The engineered transaminase first binds the PLP cofactor to form an internal aldimine. Upon binding the ketone substrate (4,4'-biphenylpyruvic acid), the enzyme facilitates the transfer of the amino group from the external amine donor (isopropylamine) to the substrate. The specific mutations introduced (R60G, D72A, K196N, Q199L) are hypothesized to optimize the active site geometry, reducing steric hindrance for the bulky biphenyl substrate and stabilizing the transition state. This structural optimization results in a dramatic acceleration of the rate-limiting steps in the catalytic cycle. Consequently, the reaction achieves substrate conversion rates approaching 100% in a fraction of the time required by previous benchmarks, while strictly preserving the stereochemical integrity of the product.
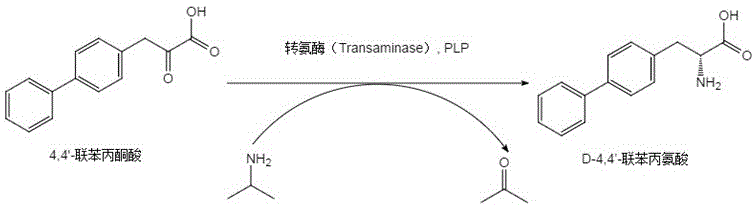
Impurity control is another critical aspect where this mutant excels. In conventional chemical synthesis, side reactions such as over-reduction or non-specific amination can lead to complex impurity profiles that are difficult to purge. In contrast, the biological specificity of the transaminase mutant ensures that only the 4-position of the pyruvate backbone is aminated, and exclusively in the D-configuration. The patent data indicates that the resulting product consistently achieves an enantiomeric excess (e.e.) of greater than 99.8%, with HPLC purity exceeding 99.5% after simple workup. This high fidelity eliminates the need for downstream chiral purification steps, which are often the most expensive part of API intermediate production. The stability of the enzyme under reaction conditions (pH 8.5-9.0, 30-40°C) further ensures consistent batch-to-batch reproducibility, a key metric for regulatory compliance and quality assurance in GMP environments.
How to Synthesize D-4,4'-Biphenylalanine Efficiently
Implementing this technology requires a precise understanding of the bioprocess parameters to maximize yield and enzyme longevity. The patent outlines a robust protocol starting with the expression of the mutant gene in an E. coli host, followed by cell lysis or whole-cell application. The reaction is conducted in a phosphate buffer system which serves to maintain the optimal pH range essential for enzyme activity. Detailed standard operating procedures regarding induction temperatures, cofactor supplementation, and substrate feeding strategies are critical for scaling this from gram to kilogram quantities. For a comprehensive guide on the specific laboratory protocols and scale-up considerations, please refer to the standardized synthesis steps outlined below.
- Preparation of the biocatalyst: Express the quadruple mutant transaminase gene (SEQ ID NO: 4) in E. coli BL21(DE3), induce with IPTG, and harvest the cells for whole-cell catalysis or enzyme extraction.
- Reaction Setup: Combine 4,4'-biphenylpyruvic acid substrate with isopropylamine (amine donor) and PLP cofactor in a phosphate buffer system maintained at pH 8.5-9.0.
- Biocatalytic Conversion: Incubate the mixture at 30-40°C with the engineered enzyme. Monitor conversion via HPLC until substrate depletion (typically 4-8 hours), then isolate the product via filtration and recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this transaminase mutant technology translates into tangible strategic advantages beyond mere technical specifications. The shift from chemical to enzymatic synthesis fundamentally alters the cost structure and risk profile of the supply chain. By eliminating the need for precious metal catalysts and harsh solvents, the process reduces both raw material volatility exposure and hazardous waste disposal costs. Furthermore, the drastic reduction in reaction time—from nearly a full day to a single shift—significantly enhances asset utilization rates, allowing manufacturers to produce more material with the same reactor footprint. This efficiency gain is crucial for meeting the tight delivery schedules often demanded by global pharmaceutical clients.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the remarkable increase in specific activity. Because the mutant enzyme is approximately 30 times more active than the wild type, the required enzyme loading is drastically reduced. This means less biomass needs to be cultivated, harvested, and processed per kilogram of final product. Additionally, the high conversion rate minimizes the loss of expensive starting materials, ensuring that the theoretical yield is closely approached in practice. The elimination of chiral resolution steps further compounds these savings by removing entire unit operations from the production line, leading to substantial cost savings in utilities, labor, and consumables without compromising on quality.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the complexity of synthetic routes. This biocatalytic route simplifies the supply chain by relying on fermentation-derived catalysts and readily available bulk chemicals like isopropylamine. The robustness of the enzyme under mild conditions reduces the risk of batch failures due to thermal runaways or pressure excursions, which are common risks in high-pressure chemical hydrogenation. Moreover, the ability to produce the intermediate with consistent high purity reduces the likelihood of downstream rejection by API manufacturers, thereby securing long-term supply contracts and fostering stronger partnerships with key stakeholders in the cardiovascular drug market.
- Scalability and Environmental Compliance: As regulatory scrutiny on pharmaceutical manufacturing intensifies, the environmental footprint of production becomes a key differentiator. This enzymatic process operates in aqueous media at near-neutral pH and moderate temperatures, generating minimal organic waste compared to solvent-heavy chemical processes. The absence of heavy metals simplifies the purification of the final product and ensures compliance with strict ICH guidelines on elemental impurities. From a scalability perspective, the linear relationship between enzyme loading and reaction rate allows for predictable scale-up from pilot plants to commercial multi-ton reactors, ensuring that supply can be ramped up quickly to meet market surges without requiring extensive process re-validation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this transaminase technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear picture of what partners can expect when integrating this route into their manufacturing portfolio. Understanding these nuances is essential for making informed decisions about technology licensing or contract manufacturing agreements.
Q: What represents the primary advantage of the R60G-D72A-K196N-Q199L mutant over wild-type transaminases?
A: The quadruple mutant exhibits approximately 30 times higher unit enzyme activity compared to the wild-type parent while maintaining exceptional stereoselectivity (>99.8% e.e.), significantly reducing reaction time from nearly 18 hours to under 8 hours.
Q: How does this biocatalytic route address the purity requirements for LCZ696 intermediates?
A: The engineered enzyme demonstrates exquisite chemo- and enantioselectivity, yielding D-4,4'-biphenylalanine with HPLC purity exceeding 99.5% and optical purity (e.e.) greater than 99.8%, effectively eliminating the need for complex chiral resolution steps common in chemical synthesis.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process operates under mild conditions (30-40°C, atmospheric pressure) using aqueous buffers and avoids hazardous heavy metal catalysts. The high turnover rate and reduced enzyme loading (0.01 wt%) make it highly economically viable for metric-ton scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable D-4,4'-Biphenylalanine Supplier
The transition to high-efficiency biocatalysis represents a significant opportunity for pharmaceutical companies to secure a competitive edge in the production of ARNI therapies. NINGBO INNO PHARMCHEM stands at the forefront of this technological shift, possessing the specialized expertise required to translate such complex enzymatic pathways into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising lab-scale results of patents like CN116004562A can be reliably reproduced on an industrial scale. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced chiral HPLC capabilities to guarantee that every batch of D-4,4'-biphenylalanine meets the exacting standards required for FDA-approved drug substances.
We invite forward-thinking procurement leaders to engage with us to explore how this technology can optimize your supply chain. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data from our recent pilot runs and to discuss route feasibility assessments that could redefine the economics of your Sacubitril production strategy.
