Scalable Synthesis of Etodolac Methyl Ester: A High-Yield Acid-Catalyzed Route for API Manufacturing
The pharmaceutical industry continuously seeks robust synthetic routes for non-steroidal anti-inflammatory drugs (NSAIDs), particularly for key intermediates that dictate the final quality and cost of the active pharmaceutical ingredient (API). Patent CN101077872B, published in early 2012, introduces a transformative methodology for the preparation of Etodolac Methyl Ester, a critical precursor in the synthesis of Etodolac. This specific intermediate, chemically defined as 1,8-diethyl-1,3,4,9-tetrahydropyrano[3,4-b]indole-1-acetic acid methyl ester, serves as the structural backbone for a widely prescribed analgesic used in treating rheumatoid arthritis and osteoarthritis. The disclosed innovation addresses long-standing inefficiencies in traditional manufacturing by leveraging a specialized acid-catalyzed cyclization strategy that utilizes a binary solvent system. By shifting away from cumbersome gas-handling procedures to a liquid-phase acid catalysis model, this technology offers a pathway to significantly higher purity profiles and operational stability. For global supply chain stakeholders, understanding the nuances of this patent is essential for securing a reliable pharmaceutical intermediate supplier capable of delivering consistent quality at scale.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Etodolac Methyl Ester has relied on methodologies referenced in earlier patents such as US6,006,741, which involve the reaction of 7-ethyl tryptophanol with methyl 3-oxopentanoate. However, these legacy processes are fraught with significant operational drawbacks that hinder efficient commercial production. The conventional protocol typically requires the preparation of a hydrogen chloride isobutanol solution, a step that introduces complexity and safety hazards associated with handling corrosive gases and volatile solvents. Furthermore, the post-reaction workup in these older methods involves pouring the reaction mixture directly into aqueous sodium bicarbonate, a procedure that often results in emulsions and difficult phase separations. Consequently, the final product frequently exhibits poor physical appearance and suboptimal purity, with reported yields capping at a mere 78%. This low efficiency translates directly into higher raw material consumption and increased waste generation, creating a bottleneck for cost reduction in API manufacturing.
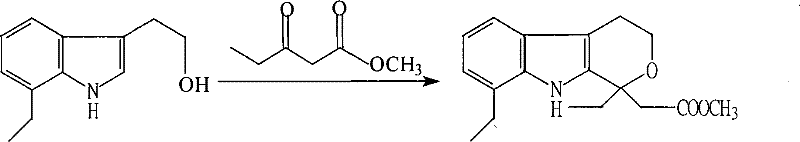
The Novel Approach
In stark contrast to the limitations of prior art, the novel approach detailed in CN101077872B employs a streamlined acid-catalyzed mechanism that fundamentally simplifies the reaction engineering. The core innovation lies in the utilization of a mixed solvent system composed of C1 to C2 lower alcohols, preferably methanol, and benzene compounds, most effectively toluene. Within this optimized medium, the reaction proceeds under the catalytic influence of concentrated sulfuric acid or oleum, eliminating the need for pre-prepared hydrogen chloride solutions. The process is characterized by its mild operating conditions, specifically maintained between -5°C and 0°C, which ensures precise control over the cyclization kinetics. Upon completion, the workup is remarkably straightforward: the acid layer is simply separated, and the organic phase is neutralized and concentrated. This methodological shift not only boosts the conversion rate to nearly 100% but also elevates the isolated yield to above 98%, representing a quantum leap in process efficiency.
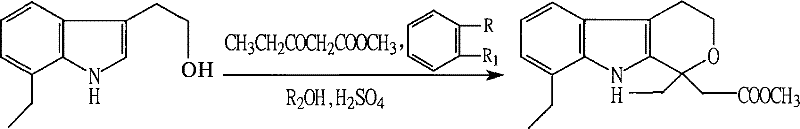
Mechanistic Insights into Sulfuric Acid-Catalyzed Cyclization
The success of this synthetic route is deeply rooted in the specific mechanistic interactions facilitated by the strong acid catalyst within the biphasic solvent environment. The reaction essentially functions as a variation of the Pictet-Spengler cyclization, where the nucleophilic indole ring attacks the electrophilic carbonyl carbon of the keto-ester. The presence of concentrated sulfuric acid serves a dual purpose: it protonates the carbonyl oxygen of the methyl 3-oxopentanoate, thereby enhancing its electrophilicity, and it simultaneously activates the indole nucleus for nucleophilic attack. The choice of a methanol-toluene mixture is critical; methanol acts as a polar protic solvent that stabilizes the charged intermediates and transition states, while toluene provides the necessary hydrophobic environment to facilitate the eventual phase separation of the spent acid. This delicate balance prevents the formation of polymeric by-products and ensures that the cyclization proceeds exclusively to form the desired tetrahydropyrano-indole core structure with high regioselectivity.
Furthermore, the strict temperature control between -5°C and 0°C is a vital parameter for impurity management and stereochemical integrity. At higher temperatures, the risk of over-alkylation or degradation of the sensitive indole moiety increases significantly, leading to colored impurities that are difficult to remove. By maintaining the reaction at sub-zero temperatures, the kinetic energy of the molecules is regulated, favoring the formation of the thermodynamically stable cyclic product while suppressing side reactions. The molar ratio of reactants is also optimized, with a preferred stoichiometry of 7-ethyl tryptophanol to methyl 3-oxopentanoate to acid at 1:1.2-1.5:3-4. This excess of acid ensures complete protonation and drives the equilibrium forward, while the slight excess of the keto-ester guarantees full consumption of the more valuable tryptophanol derivative. The result is a crude product of such high purity that simple recrystallization from methanol yields a final specification suitable for downstream pharmaceutical processing without the need for chromatographic purification.
How to Synthesize Etodolac Methyl Ester Efficiently
Implementing this high-yield synthesis requires adherence to precise operational parameters to replicate the success demonstrated in the patent examples. The process begins with the careful preparation of the reaction vessel, ensuring it is equipped with efficient stirring and cooling capabilities to manage the exothermic addition of the acid catalyst. The selection of reagents, particularly the use of anhydrous methanol and dry toluene, is paramount to prevent hydrolysis of the ester functionality during the acidic conditions. Operators must monitor the temperature rigorously during the dropwise addition of sulfuric acid, as any deviation above 0°C could compromise the yield and purity profile. Following the reaction period, the phase separation step offers a unique opportunity for process intensification, as the dense acid layer can be removed and potentially regenerated or disposed of with minimal environmental impact compared to aqueous quenching methods.
- Prepare the reaction mixture by dissolving 7-ethyl tryptophanol and methyl 3-oxopentanoate in a mixed solvent of anhydrous methanol and toluene, then cool the solution to between -5°C and 0°C.
- Slowly add concentrated sulfuric acid dropwise to the cooled mixture while maintaining the temperature between -5°C and 0°C, and stir for approximately 1 hour to ensure complete cyclization.
- Separate the acid layer after the reaction completes, extract the acid phase with toluene, combine organic phases, neutralize with aqueous sodium carbonate, and recrystallize from methanol to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented process offers tangible strategic benefits that extend beyond mere chemical yield. The elimination of complex gas-handling infrastructure required for hydrogen chloride generation represents a significant capital expenditure saving and reduces the operational risk profile of the manufacturing facility. Moreover, the ability to continuously recycle the mother liquor means that solvent consumption is drastically reduced, aligning with modern green chemistry principles and lowering the variable costs associated with raw material procurement. The robustness of the process also implies a more predictable production schedule, as the high conversion rate minimizes the need for reprocessing batches that fail to meet quality specifications, thereby enhancing overall supply chain reliability.
- Cost Reduction in Manufacturing: The economic advantages of this route are driven primarily by the simplification of the workflow and the maximization of atom economy. By replacing the multi-step preparation of hydrogen chloride solutions with the direct use of commercially available concentrated sulfuric acid, the process removes several unit operations, reducing labor hours and energy consumption. Additionally, the near-quantitative yield ensures that expensive starting materials like 7-ethyl tryptophanol are utilized with maximum efficiency, minimizing the cost per kilogram of the final intermediate. The reduction in waste generation further lowers disposal costs, contributing to a leaner and more cost-effective manufacturing model that enhances competitiveness in the global market.
- Enhanced Supply Chain Reliability: From a logistics perspective, the reliance on stable, liquid reagents rather than gaseous ones simplifies inventory management and transportation. Concentrated sulfuric acid and toluene are commodity chemicals with established supply chains, reducing the risk of raw material shortages that can plague specialized reagent markets. The stability of the reaction conditions also means that the process is less susceptible to variations in ambient conditions or minor equipment fluctuations, ensuring consistent output quality. This reliability is crucial for maintaining uninterrupted supply to downstream API manufacturers, preventing costly production stoppages and fostering long-term partnerships based on trust and dependability.
- Scalability and Environmental Compliance: The design of this synthesis is inherently scalable, moving seamlessly from laboratory benchtop to multi-ton commercial reactors without the need for fundamental process re-engineering. The simple workup involving phase separation is easily adapted to large-scale separation equipment, avoiding the bottlenecks often associated with filtration or complex extractions. Furthermore, the reduction in three-waste emissions aligns with increasingly stringent environmental regulations, mitigating the risk of regulatory fines and facilitating smoother permitting processes for expansion. This environmental stewardship not only protects the corporate reputation but also future-proofs the manufacturing asset against evolving sustainability mandates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this method outperforms traditional techniques. Understanding these details is vital for technical teams evaluating the feasibility of technology transfer and for commercial teams assessing the value proposition of sourcing intermediates produced via this method.
Q: What is the primary advantage of this new synthesis method over conventional processes?
A: The primary advantage is a drastic improvement in yield and purity. While conventional methods using hydrogen chloride isobutanol solutions typically achieve a maximum yield of only 78% with poor product appearance, this novel acid-catalyzed route consistently achieves yields exceeding 98% with conversion rates approaching 100%, significantly reducing raw material waste.
Q: How does the solvent system contribute to process efficiency?
A: The use of a mixed solvent system comprising C1-C2 lower alcohols (specifically methanol) and benzene compounds (specifically toluene) optimizes solubility and reaction kinetics. This specific combination allows for a simplified workup procedure where the acid layer can be directly separated, eliminating complex extraction steps and enabling the continuous recycling of mother liquor, which lowers overall production costs.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is highly suitable for industrial scale-up. It operates under mild reaction conditions (-5°C to 0°C), utilizes readily available reagents like concentrated sulfuric acid instead of complex gas solutions, and generates minimal three-waste emissions. The simple separation and recrystallization steps ensure robust quality control and operational safety at commercial volumes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Etodolac Methyl Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the CN101077872B process are fully realized in practice. We maintain stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify that every batch of Etodolac Methyl Ester meets the exacting standards required for pharmaceutical applications. Our commitment to quality assurance means that clients can rely on us not just for volume, but for the consistency and integrity of the chemical supply.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic impact of switching to this high-efficiency process. We encourage you to contact us today to索取 specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and efficiency in your NSAID manufacturing operations.
