Advanced Synthesis of 2-Imine-1,10-Phenanthroline Ligands for High-Efficiency Ethylene Oligomerization
The chemical industry is currently witnessing a paradigm shift in the synthesis of critical catalyst ligands, driven by the dual imperatives of environmental compliance and process efficiency. Patent CN102040608A introduces a groundbreaking methodology for the preparation of 2-imine-1,10-phenanthroline ligands, which serve as the cornerstone for high-performance ethylene oligomerization catalysts. This innovation fundamentally alters the synthetic landscape by replacing hazardous potassium cyanide with environmentally benign methyllithium reagents, thereby addressing long-standing safety concerns in fine chemical manufacturing. The strategic implementation of low-polarity solvents during the oxidation phase further mitigates the formation of undesirable ring-opened byproducts, ensuring a higher fidelity of the molecular architecture. For R&D Directors and Procurement Managers alike, this represents a significant opportunity to optimize the supply chain for Linear Alpha Olefins (LAO), which are essential precursors for polyethylene comonomers and synthetic lubricants. By adopting this novel route, manufacturers can achieve a more sustainable production profile while maintaining the rigorous purity standards required for downstream polymerization applications.
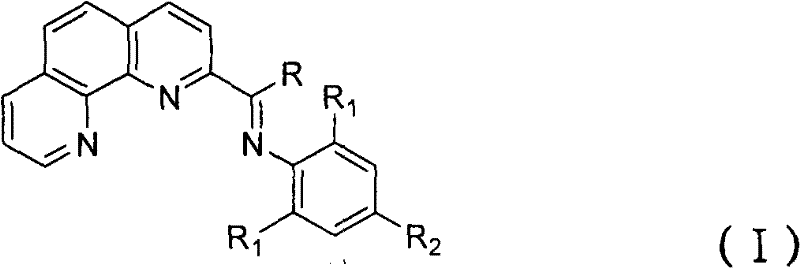
Furthermore, the versatility of this synthetic approach allows for the precise tuning of steric and electronic properties through the variation of substituents on the imine nitrogen. This adaptability is crucial for tailoring catalyst performance to specific oligomerization targets, whether the goal is maximizing selectivity for C4-C8 olefins or producing higher molecular weight fluids. The patent data indicates that the resulting iron, cobalt, or nickel complexes exhibit exceptional catalytic activity, with selectivity for linear alpha-olefins exceeding 96% under optimized conditions. Such high selectivity translates directly into reduced separation costs and improved energy efficiency in the final processing stages. As a reliable specialty chemical supplier, understanding the nuances of this ligand synthesis is paramount for securing a competitive advantage in the global market for advanced polymer additives and industrial catalysts.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-acyl-1,10-phenanthroline intermediates, which are prerequisites for the target ligands, has relied heavily on the use of potassium cyanide. This traditional pathway, while chemically viable, presents severe logistical and safety challenges that are increasingly untenable in modern regulatory environments. The handling of cyanide salts requires specialized infrastructure, rigorous personnel training, and expensive waste treatment protocols to neutralize toxic effluents before discharge. Moreover, the Reissert-Henze type reactions often involve harsh conditions that can lead to inconsistent yields and the generation of complex impurity profiles difficult to remove. From a supply chain perspective, reliance on cyanide-based chemistry introduces significant risk factors, including potential regulatory bans and fluctuating costs associated with hazardous material transport. These limitations not only inflate the operational expenditure but also constrain the ability of manufacturers to scale production rapidly in response to market demand for ethylene oligomerization catalysts.
The Novel Approach
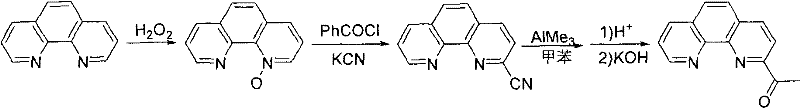
In stark contrast, the methodology disclosed in CN102040608A leverages organolithium chemistry to effect direct functionalization of the phenanthroline ring system. By utilizing methyllithium as a nucleophile, the process bypasses the need for cyanide entirely, substituting a high-risk reagent with one that, while reactive, is manageable within standard inert atmosphere protocols. This shift not only enhances the safety profile of the manufacturing facility but also simplifies the waste stream, eliminating the need for cyanide destruction units. Additionally, the patent highlights the strategic use of low-polarity solvents during the subsequent oxidation step with potassium permanganate. This specific solvent choice is critical as it suppresses the oxidative cleavage of the phenanthroline ring, a common side reaction that plagues conventional methods. The result is a cleaner reaction profile with fewer byproducts, reducing the burden on downstream purification processes and enhancing the overall economic viability of the synthesis.
Mechanistic Insights into Organolithium-Mediated Functionalization
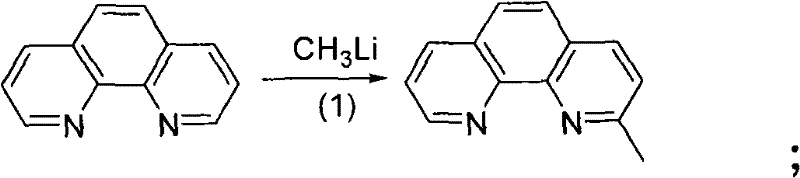
The core of this technological advancement lies in the precise control of the lithiation step, where 1,10-phenanthroline is treated with methyllithium at cryogenic temperatures ranging from -80°C to -20°C. This low-temperature regime is essential for directing the nucleophilic attack to the C2 position of the heterocyclic ring, preventing non-specific addition or decomposition of the sensitive organometallic intermediate. The resulting 2-methyl-1,10-phenanthroline is then subjected to oxidation, where the choice of solvent plays a pivotal mechanistic role. In high-polarity media, permanganate ions can aggressively attack the electron-rich aromatic system, leading to ring opening; however, in solvents like tetrahydrofuran or dioxane, the oxidation is selectively confined to the methyl group. This selectivity ensures the preservation of the tridentate coordination geometry required for effective metal binding in the final catalyst complex.
Following oxidation, the resulting carboxylic acid is converted to the ketone via reaction with alkyllithium reagents, a transformation that proceeds through a stable tetrahedral intermediate before hydrolysis. This step is particularly noteworthy for its potential for reagent recycling; when the target R group is methyl, the methyllithium used in the initial step can theoretically be recovered or reused, minimizing reagent consumption. The final condensation with substituted anilines to form the imine bond is catalyzed by acid and proceeds under reflux, driving the equilibrium towards the product through water removal. This comprehensive mechanistic understanding allows for tight control over impurity profiles, ensuring that the final ligand meets the stringent specifications required for high-performance ethylene oligomerization. For technical teams, this level of control translates to consistent catalyst batch-to-batch performance and reliable polymer product quality.
How to Synthesize 2-Ethanoyl-1,10-Phenanthroline Efficiently
The practical execution of this synthesis requires strict adherence to anhydrous and oxygen-free conditions to maintain the integrity of the organolithium species throughout the reaction sequence. Operators must utilize inert gas blankets, typically nitrogen or argon, and ensure all solvents are rigorously dried prior to use to prevent premature quenching of the reactive intermediates. The detailed standardized synthesis steps involve precise temperature ramping and stoichiometric control to maximize yield while minimizing side reactions. For a complete breakdown of the operational parameters and safety protocols required for laboratory or pilot-scale execution, please refer to the technical guide below.
- Lithiation of 1,10-phenanthroline using methyllithium at low temperatures to form 2-methyl-1,10-phenanthroline.
- Oxidation of the methyl group to carboxylic acid using potassium permanganate in low-polarity solvents.
- Conversion to 2-acyl-1,10-phenanthroline via reaction with alkyllithium reagents.
- Condensation with substituted anilines to form the final imine ligand structure.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial standpoint, the adoption of this cyanide-free synthesis route offers profound advantages for procurement strategies and supply chain resilience. The elimination of potassium cyanide removes a major bottleneck in raw material sourcing, as cyanide is subject to strict transportation regulations and limited supplier availability in many jurisdictions. By switching to organolithium reagents, which are more widely available and easier to handle with standard chemical infrastructure, manufacturers can significantly reduce lead times for high-purity chemical intermediates. Furthermore, the simplified waste profile means lower disposal costs and reduced liability exposure, contributing to substantial cost savings in the overall manufacturing budget. This process optimization aligns perfectly with the goals of cost reduction in polymer additive manufacturing, allowing companies to remain competitive without compromising on safety or quality standards.
- Cost Reduction in Manufacturing: The ability to recycle methyllithium reagents in specific iterations of the process directly lowers the raw material cost per kilogram of the final ligand. Additionally, the reduction in byproduct formation means less material is lost during purification, improving the effective mass balance of the process. These efficiencies compound over large production volumes, leading to significant margin improvements for the final catalyst product. By avoiding the expensive remediation steps associated with cyanide waste, the overall operational expenditure is drastically simplified, providing a clear economic incentive for technology adoption.
- Enhanced Supply Chain Reliability: Diversifying the reagent base away from highly regulated toxic substances mitigates the risk of supply disruptions caused by regulatory changes or transportation incidents. The use of common organic solvents and commercially available organolithium compounds ensures a stable and continuous supply of inputs. This reliability is critical for maintaining production schedules and meeting delivery commitments to downstream polymer producers. A robust supply chain for these ligands ensures that ethylene oligomerization units can operate continuously without interruption due to catalyst shortages.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up of complex organometallic ligands, with reaction conditions that are translatable from laboratory glassware to industrial reactors. The reduced toxicity profile facilitates easier permitting and compliance with increasingly stringent environmental regulations globally. This forward-looking approach future-proofs the manufacturing asset against potential bans on hazardous chemicals, ensuring long-term operational viability. Companies investing in this technology position themselves as leaders in sustainable chemical manufacturing, appealing to eco-conscious partners and clients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel ligand synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this route into their existing production frameworks.
Q: Why is the lithiation method preferred over traditional cyanide routes?
A: The lithiation method eliminates the use of highly toxic potassium cyanide, significantly improving workplace safety and reducing hazardous waste disposal costs associated with cyanide detoxification.
Q: How does solvent polarity affect the oxidation step yield?
A: Using low-polarity organic solvents during potassium permanganate oxidation minimizes the formation of ring-opened byproducts, thereby preserving the integrity of the phenanthroline core and improving overall purity.
Q: Can this ligand be scaled for industrial ethylene oligomerization?
A: Yes, the process demonstrates robust scalability with total yields exceeding 40% and high selectivity for linear alpha-olefins, making it suitable for commercial-scale polymer additive manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Imine-1,10-Phenanthroline Ligand Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced ligand technology plays in the evolution of the polyolefin industry. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from patent to practice is seamless and efficient. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that validate every batch against the highest industry standards. Our capability to manage complex organometallic syntheses under inert conditions makes us an ideal partner for companies seeking to secure their supply of high-performance ethylene oligomerization catalysts.
We invite you to collaborate with us to explore how this innovative synthesis route can enhance your product portfolio and operational efficiency. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to support your decision-making process. Let us help you drive value through chemistry, ensuring a sustainable and profitable future for your manufacturing operations.
