Scalable Synthesis of 7-Iodo-4-Aminopyrrolo Triazine for Advanced Antiviral Manufacturing
The pharmaceutical industry's relentless pursuit of efficient antiviral therapies has placed significant emphasis on the robust manufacturing of key heterocyclic intermediates. Patent CN112457318A discloses a groundbreaking synthesis process for 7-iodo-4-aminopyrrolo[2,1-F][1,2,4]triazine, a critical building block for Reidesciclovir. This technical disclosure outlines a streamlined four-step pathway originating from readily available pyrrole, leveraging a unique ammonium sulfate-mediated transformation to install the cyano group. For R&D directors and procurement strategists, this methodology represents a paradigm shift away from hazardous cyanation reagents toward safer, commodity-grade nitrogen sources. The process is characterized by mild thermal conditions, typically maintaining reaction temperatures below 30°C, which drastically reduces energy consumption and thermal runaway risks. Furthermore, the reported purity levels reaching 99% in the final isolation step suggest a highly selective reaction profile that minimizes downstream purification burdens. As a reliable pharmaceutical intermediates supplier, understanding such process intensifications is vital for securing long-term supply chain resilience.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional routes to functionalized pyrrolo-triazines often rely on aggressive cyanation protocols involving toxic reagents such as sodium cyanide or trimethylsilyl cyanide, which pose severe environmental and safety liabilities during commercial scale-up. These conventional methods frequently require stringent anhydrous conditions and expensive catalysts to drive the nucleophilic substitution, leading to complex workup procedures and significant heavy metal waste streams. Moreover, the thermal instability of certain intermediates in older pathways often necessitates cryogenic conditions, inflating operational expenditures through excessive cooling requirements. The accumulation of impurities from side reactions, particularly polymerization of the pyrrole ring under harsh acidic or basic conditions, often results in lower overall yields and necessitates resource-intensive chromatographic purification. For supply chain heads, these factors translate into volatile lead times and unpredictable batch-to-batch consistency, creating bottlenecks in the production of high-value antiviral APIs.
The Novel Approach
The innovative strategy detailed in the patent circumvents these historical bottlenecks by employing ammonium sulfate as a benign nitrogen donor for the conversion of the aldehyde to the nitrile functionality. This approach not only simplifies the reagent profile by utilizing a stable, non-toxic solid but also enables the reaction to proceed in a mixed ethanol-water solvent system, enhancing green chemistry metrics. The subsequent cyclization with formamidine acetate is executed under reflux conditions that are easily manageable in standard glass-lined steel reactors, eliminating the need for specialized high-pressure equipment. By consolidating what was previously a multi-step sequence into a more direct transformation, the novel approach significantly reduces the number of unit operations and solvent exchanges required. This streamlining directly correlates to cost reduction in pharmaceutical intermediates manufacturing, offering a competitive edge through reduced cycle times and simplified logistics.
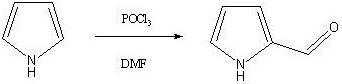
Mechanistic Insights into the Four-Step Cascade Synthesis
The synthetic journey begins with the Vilsmeier-Haack formylation, where phosphorus oxychloride reacts with DMF to generate a highly electrophilic chloroiminium ion in situ. This species attacks the electron-rich pyrrole ring at the 2-position, followed by hydrolysis to yield 2-formylpyrrole with high regioselectivity. The control of temperature below 20°C during the addition of pyrrole is critical to prevent over-chlorination or ring degradation, ensuring the integrity of the heterocyclic core. Following isolation, the aldehyde undergoes a remarkable transformation where ammonium sulfate acts as the ammonia source in a condensation-dehydration sequence to form the nitrile group. This step is mechanistically distinct from traditional oxime dehydration, offering a safer alternative that avoids the generation of hazardous gaseous byproducts.
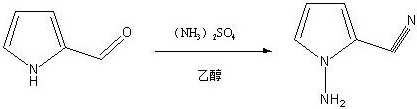
Subsequent cyclization involves the reaction of the 2-cyano intermediate with formamidine acetate in the presence of potassium phosphate, which serves as a base to facilitate the ring closure into the triazine system. The use of activated carbon during the workup of this step is a crucial purification tactic, effectively adsorbing colored polymeric impurities and trace organics to yield a pale, high-purity solid. The final stage employs N-iodosuccinimide (NIS) for electrophilic aromatic substitution, selectively installing the iodine atom at the 7-position of the fused ring system. This iodination is performed at mild temperatures (20-30°C), preserving the amino functionality and preventing dehalogenation or over-iodination side reactions. The precise stoichiometry and temperature control described ensure that the final product meets the rigorous specifications required for downstream coupling reactions in API synthesis.
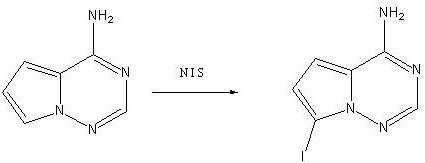
How to Synthesize 7-Iodo-4-Aminopyrrolo Triazine Efficiently
The synthesis protocol outlined in the patent provides a robust framework for laboratory and pilot-scale production, emphasizing precise temperature control and specific mass ratios to maximize yield. Operators must adhere to the sequential addition of reagents, particularly during the exothermic Vilsmeier activation and the final iodination steps, to maintain safety and product quality. The detailed standardized synthesis steps below provide a comprehensive guide for replicating this high-efficiency route in a GMP-compliant environment.
- Perform Vilsmeier-Haack formylation on pyrrole using POCl3 and DMF at low temperature to obtain 2-formylpyrrole.
- React 2-formylpyrrole with ammonium sulfate in ethanol/water mixture under reflux to generate the 2-cyano intermediate.
- Cyclize the 2-cyano intermediate with formamidine acetate and potassium phosphate to form the 4-amino pyrrolo triazine core.
- Conduct electrophilic iodination using N-iodosuccinimide (NIS) on the 4-amino intermediate to yield the final 7-iodo product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthesis route offers substantial strategic benefits for organizations aiming to optimize their supply chain for antiviral ingredients. The substitution of hazardous cyanide reagents with ammonium sulfate not only lowers regulatory compliance costs but also simplifies the procurement of raw materials, as ammonium sulfate is a globally available commodity chemical. The ability to run reactions in ethanol and water mixtures reduces reliance on expensive, anhydrous aprotic solvents, leading to significant savings in solvent recovery and disposal costs. Furthermore, the mild operating temperatures reduce the energy load on manufacturing facilities, contributing to a lower carbon footprint and aligning with modern sustainability goals. These factors collectively enhance the economic viability of producing this complex intermediate at a commercial scale.
- Cost Reduction in Manufacturing: The elimination of toxic cyanide sources and the use of inexpensive reagents like ammonium sulfate and formamidine acetate drastically lower the bill of materials. Additionally, the simplified workup procedures, which avoid complex extractions or chromatography in favor of crystallization and filtration, reduce labor hours and solvent consumption. This streamlined process flow translates to a lower cost of goods sold (COGS), allowing for more competitive pricing in the global market for pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: By utilizing stable, non-hazardous starting materials, the risk of supply disruptions due to strict transportation regulations for dangerous goods is minimized. The robustness of the reaction conditions, which tolerate slight variations in temperature and mixing without compromising purity, ensures consistent batch quality and reliable delivery schedules. This stability is crucial for maintaining continuous API production lines and meeting the demanding timelines of drug development projects.
- Scalability and Environmental Compliance: The process has been demonstrated to scale effectively from gram to kilogram quantities using standard reactor configurations, indicating high readiness for metric-ton production. The aqueous workups and reduced solvent usage simplify wastewater treatment, ensuring compliance with increasingly stringent environmental regulations. This scalability ensures that the supply can grow in tandem with market demand for Reidesciclovir without requiring massive capital investment in new infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of 7-iodo-4-aminopyrrolo[2,1-F][1,2,4]triazine, based on the specific embodiments and advantages disclosed in the patent literature. These insights are designed to clarify the operational feasibility and quality attributes of this manufacturing route for potential partners.
Q: What are the safety advantages of using ammonium sulfate in this synthesis?
A: The use of ammonium sulfate eliminates the need for highly toxic cyanide sources typically required for nitrile introduction, significantly reducing hazardous waste handling and improving operator safety profiles during commercial scale-up.
Q: How does this process improve purity compared to conventional methods?
A: By utilizing mild reaction temperatures (below 30°C) and specific workup procedures like activated carbon treatment, the process minimizes side reactions and polymerization, consistently achieving purities of 98-99% without complex chromatography.
Q: Is this synthesis route suitable for large-scale production?
A: Yes, the patent demonstrates successful scaling from gram to kilogram levels (e.g., 250L reactors) using common solvents like ethanol and dichloromethane, indicating high feasibility for metric-ton annual production capacities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 7-Iodo-4-Aminopyrrolo Triazine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of next-generation antiviral therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that employ advanced analytical techniques to verify every batch against the highest industry standards. Our capability to adapt and optimize processes like the one described in CN112457318A allows us to offer a secure and consistent supply of this vital building block.
We invite global pharmaceutical partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this safer, more efficient manufacturing method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your volume needs, ensuring a partnership built on transparency, quality, and mutual success.
