Advanced Synthesis of 3-Bromo-2-Chloro-4,6-Difluoroaniline for Commercial API Manufacturing
The pharmaceutical industry's relentless pursuit of novel kinase inhibitors and targeted therapies has placed a premium on highly functionalized aromatic building blocks, specifically poly-halogenated anilines. Among these, 3-bromo-2-chloro-4,6-difluoroaniline stands out as a critical scaffold for constructing complex heterocyclic systems found in modern oncology drugs. The recent disclosure in patent CN114539073A introduces a robust and economically viable synthetic pathway that addresses long-standing challenges in the manufacturing of this valuable intermediate. By leveraging a strategic sequence of nitration, reduction, and selective chlorination, this methodology offers a compelling alternative to traditional routes that often suffer from poor regioselectivity or hazardous reagent requirements. For R&D directors and procurement strategists alike, understanding the nuances of this patented process is essential for securing a stable supply chain of high-purity pharmaceutical intermediates. The technical breakthrough lies not just in the chemical transformation itself, but in the optimization of reaction conditions that facilitate safe scale-up while maintaining rigorous quality standards required for GMP production environments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of multi-halogenated anilines has been plagued by significant technical hurdles that impact both cost and yield. Conventional routes often rely on the direct chlorination of aniline derivatives using elemental chlorine gas, a process that is notoriously difficult to control due to the high reactivity of the amino group. This lack of control frequently leads to over-chlorination, resulting in a complex mixture of mono-, di-, and tri-chlorinated byproducts that are chemically similar and extremely difficult to separate via standard crystallization or distillation techniques. Furthermore, the use of gaseous chlorine poses severe safety risks and requires specialized corrosion-resistant equipment, driving up capital expenditure for manufacturing facilities. Another common bottleneck involves the order of functional group installation; introducing the nitro or amino group too early in the synthesis can lead to unwanted side reactions during subsequent halogenation steps, necessitating costly protecting group strategies that add unnecessary steps and reduce overall atom economy. These inefficiencies translate directly into higher production costs and longer lead times, creating vulnerabilities in the supply chain for downstream API manufacturers who depend on consistent quality and volume.
The Novel Approach
In stark contrast to these legacy methods, the process outlined in patent CN114539073A employs a sophisticated late-stage functionalization strategy that maximizes efficiency and safety. The core innovation involves the use of N-Chlorosuccinimide (NCS) as the chlorinating agent in the final step, which provides a much milder and more controllable source of electrophilic chlorine compared to molecular chlorine. This reagent choice allows for precise mono-chlorination at the ortho-position relative to the amino group, driven by the strong activating effect of the amine, while minimizing the risk of poly-substitution. Additionally, the route utilizes a cost-effective reduction step employing iron powder and ammonium chloride, replacing expensive catalytic hydrogenation or tin-based reductions that generate heavy metal waste. By establishing the fluorine and bromine substituents on the aromatic ring prior to the introduction of the nitrogen functionality, the synthesis avoids compatibility issues and ensures that the sensitive carbon-halogen bonds remain intact throughout the transformation. This logical disconnection of the molecule not only simplifies the operational workflow but also significantly enhances the purity profile of the crude product, reducing the burden on downstream purification processes.
Mechanistic Insights into Selective Electrophilic Chlorination
The success of this synthetic route hinges on a deep understanding of electronic effects and regioselectivity during the electrophilic aromatic substitution steps. The initial nitration of 1-bromo-3,5-difluorobenzene is directed by the interplay between the electron-withdrawing fluorine atoms and the bromine substituent. Although fluorine is deactivating, it is ortho/para directing, while bromine is weakly deactivating but also ortho/para directing. The reaction conditions specified, utilizing concentrated sulfuric acid and nitric acid at controlled low temperatures of 0-10°C, ensure that the nitro group enters the position ortho to the bromine atom, setting the stage for the correct substitution pattern in the final molecule. Following the reduction of the nitro group to the amine using iron powder, the electronic landscape of the ring changes dramatically. The amino group is a powerful activator and strongly ortho/para directing, overpowering the influence of the halogens. When N-Chlorosuccinimide is introduced in the final step, the electrophilic chlorine species attacks the position ortho to the amino group, which is sterically accessible and electronically favored. This mechanistic pathway ensures that the chlorine atom is installed exclusively at the C2 position, yielding the desired 3-bromo-2-chloro-4,6-difluoroaniline with high fidelity.
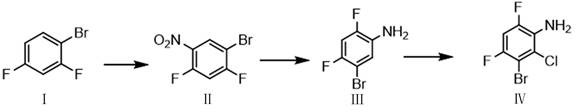
Beyond the primary transformation, the mechanism also accounts for impurity control, which is paramount for pharmaceutical applications. The use of iron powder for reduction, followed by a simple filtration workup, effectively removes metal residues that could otherwise catalyze decomposition or interfere with subsequent coupling reactions. The choice of solvents such as acetonitrile or dichloromethane in the chlorination step further aids in solubilizing the intermediate while keeping succinimide byproducts in solution or easily separable phases. The patent data indicates that careful temperature control during the exothermic addition of NCS prevents thermal degradation and minimizes the formation of oxidative byproducts. By isolating the intermediate aniline (Compound III) before chlorination, manufacturers have an additional checkpoint to verify identity and purity, ensuring that any unreacted nitro compounds or reduction byproducts do not carry through to the final expensive chlorination step. This modular approach to synthesis provides multiple opportunities for quality intervention, resulting in a final product that meets the stringent specifications required for clinical trial materials and commercial API production.
How to Synthesize 3-Bromo-2-Chloro-4,6-Difluoroaniline Efficiently
Implementing this synthesis in a pilot or commercial plant requires strict adherence to the optimized parameters defined in the patent to ensure reproducibility and safety. The process begins with the preparation of the nitro-intermediate, where temperature control is critical to prevent dinitration or oxidation side reactions. Following isolation, the reduction step utilizes a heterogeneous mixture of iron and ammonium chloride, requiring efficient stirring to maintain mass transfer between the solid metal and the organic phase. The final chlorination step demands careful monitoring of heat evolution, as the reaction between the amine and NCS can be exothermic. For a detailed breakdown of the specific molar ratios, solvent volumes, and workup procedures necessary to achieve the reported yields, please refer to the standardized protocol below.
- Nitration of 1-bromo-3,5-difluorobenzene using sulfuric and nitric acid at low temperatures (0-10°C) to form the nitro-intermediate.
- Reduction of the nitro group to an amine using iron powder and ammonium chloride in an alcoholic solvent at 20-60°C.
- Selective chlorination of the aniline derivative using N-Chlorosuccinimide (NCS) in organic solvents like acetonitrile to yield the final product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers substantial benefits that extend far beyond the laboratory bench, directly impacting the bottom line and supply chain resilience for pharmaceutical manufacturers. The shift away from hazardous gaseous reagents to solid, manageable alternatives like NCS and iron powder drastically reduces the regulatory burden and safety infrastructure costs associated with production facilities. This simplification of the hazard profile allows for manufacturing in a broader range of facilities, increasing the available capacity and reducing the risk of supply disruptions caused by specialized equipment maintenance or safety audits. Furthermore, the use of commodity chemicals such as sulfuric acid, nitric acid, and ethanol ensures that raw material sourcing is not dependent on niche suppliers, thereby stabilizing costs against market volatility. The streamlined nature of the synthesis, with fewer purification steps and higher crude purity, translates into reduced solvent consumption and waste generation, aligning with modern sustainability goals and lowering disposal costs.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the replacement of expensive catalysts and hazardous gases with inexpensive, bulk-available reagents. By utilizing iron powder for reduction instead of precious metal catalysts like palladium or platinum, the process eliminates the need for costly catalyst recovery systems and reduces the risk of heavy metal contamination in the final product. Additionally, the high selectivity of the NCS chlorination step minimizes the loss of valuable fluorinated intermediates to byproduct formation, effectively increasing the overall yield of the process without requiring complex chromatographic separations. These factors combine to lower the cost of goods sold (COGS) significantly, providing a competitive pricing advantage for the final API intermediate while maintaining healthy profit margins for the manufacturer.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the robustness of the reaction conditions and the availability of starting materials. The starting material, 1-bromo-3,5-difluorobenzene, is a widely produced commodity chemical with a stable global supply base, reducing the risk of raw material shortages. The tolerance of the reaction to minor variations in temperature and stoichiometry, as evidenced by the broad ranges provided in the patent examples, ensures consistent output even when scaling up from pilot to commercial tonnage. This reliability allows procurement managers to forecast demand more accurately and negotiate long-term supply agreements with greater confidence, knowing that the manufacturing process is not prone to frequent batch failures or unpredictable delays caused by sensitive reaction parameters.
- Scalability and Environmental Compliance: The environmental footprint of this synthesis is markedly lower than traditional methods, facilitating easier regulatory approval and permitting for new production lines. The aqueous workup procedures and the ability to recycle solvents like acetonitrile and dichloromethane contribute to a greener manufacturing profile. The absence of heavy metal waste streams from the reduction step simplifies wastewater treatment requirements and reduces the liability associated with hazardous waste disposal. As environmental regulations become increasingly stringent globally, adopting a synthesis route that inherently minimizes waste and toxicity provides a strategic advantage, future-proofing the supply chain against evolving compliance standards and ensuring uninterrupted production capabilities.
Frequently Asked Questions (FAQ)
To further clarify the technical and commercial implications of this synthesis method, we have compiled answers to common questions regarding its implementation and performance. These insights are derived directly from the experimental data and technical disclosures within the patent documentation, providing a transparent view of the process capabilities. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this intermediate into their own drug development pipelines.
Q: What are the key advantages of using NCS for chlorination in this synthesis?
A: Using N-Chlorosuccinimide (NCS) instead of chlorine gas allows for milder reaction conditions, improved safety profiles, and better regioselectivity, minimizing the formation of poly-chlorinated byproducts.
Q: How does this method address impurity control in fluorinated anilines?
A: The stepwise approach isolates the nitro-intermediate before reduction, allowing for purification prior to the sensitive chlorination step, thereby ensuring high purity specifications for the final API intermediate.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the patent explicitly highlights the use of inexpensive raw materials like iron powder and common solvents, along with simple workup procedures like filtration and extraction, making it highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Bromo-2-Chloro-4,6-Difluoroaniline Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of novel therapeutics depends on the availability of high-quality, consistently supplied intermediates. Our team of expert chemists has thoroughly analyzed the patented route for 3-bromo-2-chloro-4,6-difluoroaniline and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are committed to delivering this critical building block with stringent purity specifications, utilizing our rigorous QC labs to ensure every batch meets the exacting standards required for pharmaceutical synthesis. Our state-of-the-art facilities are equipped to handle the specific safety and processing requirements of fluorinated chemistry, ensuring a secure and reliable supply for your most demanding projects.
We invite you to collaborate with us to optimize your supply chain and accelerate your development timelines. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can support your long-term strategic goals.
