Scalable Production of 2-Nonene-Gamma-Lactone via Green Oxidation for Global Flavor Markets
Scalable Production of 2-Nonene-Gamma-Lactone via Green Oxidation for Global Flavor Markets
The global demand for high-purity flavor and fragrance intermediates continues to surge, driven by the expanding food and beverage industry's need for natural-identical aromatic compounds. Patent CN102702146A introduces a groundbreaking methodology for the preparation of 2-nonene-gamma-lactone, a critical molecule known for its intense coconut and sweet aroma profiles. This compound, recognized as FEMA No. 4188, is naturally occurring in various food sources such as beef soup, sunflower oil, and mushrooms, making it a highly sought-after ingredient for flavorists worldwide. The disclosed technology offers a distinct departure from legacy synthetic routes by leveraging a simple, two-step sequence that prioritizes operational safety and environmental compliance. By utilizing hydrogen peroxide as a green oxidant, the process eliminates the reliance on hazardous heavy metals, addressing a major pain point for modern chemical manufacturers who are under increasing regulatory pressure to reduce their ecological footprint. This report analyzes the technical merits and commercial viability of this novel approach for executive decision-makers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alpha,beta-unsaturated gamma-lactones has been plagued by significant technical and economic hurdles that hinder efficient commercial scale-up. Traditional pathways often rely on complex multi-step sequences involving expensive and sensitive reagents, such as butyllithium, which require stringent anhydrous conditions and cryogenic temperatures to prevent side reactions. Other established methods utilize transition metal catalysts like Ruthenium chloride for olefin metathesis, introducing the risk of heavy metal contamination that necessitates costly and time-consuming purification steps to meet pharmaceutical or food-grade standards. Furthermore, older protocols involving selenium-containing electrophiles pose severe toxicity risks, creating substantial liabilities regarding worker safety and hazardous waste disposal. These conventional approaches frequently suffer from low overall yields due to the accumulation of losses across multiple stages, rendering them economically unviable for high-volume production. The reliance on difficult-to-source starting materials further exacerbates supply chain fragility, leading to unpredictable lead times and volatile pricing structures that disrupt procurement planning.
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the method described in patent CN102702146A presents a streamlined and robust alternative that radically simplifies the manufacturing landscape. This innovative route begins with the direct oxidation of readily available (E)-3-nonenoic acid using hydrogen peroxide in a formic acid medium, effectively combining oxidation and lactonization into a single, high-yielding transformation. The subsequent step involves a straightforward mesylation followed by base-mediated elimination to install the crucial double bond, achieving the target unsaturated lactone structure with remarkable efficiency. By circumventing the need for precious metal catalysts or toxic organometallic reagents, this process not only lowers the barrier to entry for production but also significantly enhances the purity profile of the final product. The operational simplicity allows for execution in standard glass-lined or stainless steel reactors without the need for specialized equipment, facilitating easier technology transfer and rapid deployment in existing facilities. This strategic shift towards greener chemistry aligns perfectly with modern sustainability goals while delivering superior economic performance through improved material throughput.
Mechanistic Insights into Green Oxidation and Elimination
The core of this synthetic strategy lies in the elegant utilization of performic acid generated in situ, which acts as the active oxidizing species for the initial transformation. When hydrogen peroxide is introduced into the formic acid solution, it reacts to form performic acid, a potent epoxidizing agent that attacks the electron-rich double bond of the (E)-3-nonenoic acid substrate. This epoxidation is immediately followed by an intramolecular nucleophilic attack by the carboxylic acid group, resulting in the ring-closing formation of the 3-hydroxy-gamma-nonalactone intermediate. This tandem sequence is highly regioselective and stereospecific, ensuring that the hydroxyl group is installed at the correct position to facilitate the subsequent elimination reaction. The mild reaction conditions, typically maintained between 20°C and 40°C, prevent thermal degradation of the sensitive lactone ring, thereby preserving the integrity of the molecular scaffold. This mechanistic pathway exemplifies atom economy, as the oxygen atoms from the peroxide are efficiently incorporated into the product structure with minimal generation of stoichiometric byproducts.
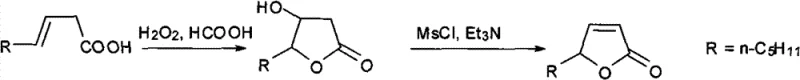
Following the formation of the saturated hydroxy-lactone intermediate, the synthesis proceeds through a classic activation-elimination sequence to generate the desired alpha,beta-unsaturation. The hydroxyl group at the 3-position is first activated by conversion into a methanesulfonate ester using methanesulfonyl chloride in the presence of a base like triethylamine. This activation transforms the poor leaving group (hydroxyl) into an excellent leaving group (mesylate), priming the molecule for elimination. Upon warming to ambient temperature, the base abstracts the acidic proton at the alpha-position relative to the carbonyl, triggering an E2 elimination mechanism that expels the mesylate group and forms the double bond. This step is critical for establishing the conjugated system responsible for the molecule's stability and aromatic properties. The use of dichloromethane as a solvent ensures good solubility of the organic intermediates while allowing for precise temperature control during the exothermic mesylation phase, ultimately leading to a high-purity final product after simple distillation.
How to Synthesize 2-Nonene-Gamma-Lactone Efficiently
Implementing this synthesis protocol requires careful attention to reaction parameters to maximize yield and ensure operator safety during the handling of oxidizing agents. The process is designed to be scalable, moving seamlessly from laboratory benchtop experiments to industrial pilot plants with minimal modification to the core chemistry. Operators must strictly control the addition rate of hydrogen peroxide to manage the exotherm during the formation of performic acid, preventing runaway reactions that could compromise safety or product quality. Following the oxidation, the workup involves neutralization and extraction, which are standard unit operations familiar to most chemical production teams. For the second step, maintaining low temperatures during the addition of methanesulfonyl chloride is essential to suppress potential side reactions, while the subsequent elimination benefits from gentle heating to drive the reaction to completion. Adherence to these standardized procedures guarantees consistent batch-to-batch reproducibility, a key requirement for supplying the rigorous flavor and fragrance markets.
- Oxidize (E)-3-nonenoic acid with 30% hydrogen peroxide in formic acid solution at 20-40°C to obtain 3-hydroxy-gamma-nonalactone.
- React the intermediate 3-hydroxy-gamma-nonalactone with methanesulfonyl chloride in dichloromethane at 0-5°C.
- Perform elimination using triethylamine at 25°C and purify via reduced pressure distillation to yield 2-nonene-gamma-lactone.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this patented methodology offers compelling advantages that directly address the cost and reliability concerns of procurement managers and supply chain directors. The substitution of expensive and supply-constrained catalysts with commodity chemicals like hydrogen peroxide and formic acid creates a more resilient supply chain that is less susceptible to market volatility. By eliminating the need for transition metals, manufacturers can avoid the significant costs associated with metal scavenging and validation testing, which are mandatory for food-grade ingredients. This reduction in downstream processing complexity translates into shorter production cycles and lower utility consumption, contributing to substantial overall cost savings without compromising on quality. Furthermore, the high yields reported in each step minimize raw material waste, improving the overall mass balance and reducing the volume of waste streams that require treatment. These factors combine to create a highly competitive cost structure that allows suppliers to offer aggressive pricing while maintaining healthy margins.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts and toxic reagents removes the need for expensive purification technologies such as chromatography or specialized filtration, drastically lowering capital and operational expenditures. The use of inexpensive, bulk-available oxidants ensures that raw material costs remain stable and predictable, shielding the project from price spikes associated with specialty chemicals. Additionally, the simplified workflow reduces labor hours and energy consumption per kilogram of product, further enhancing the economic efficiency of the manufacturing process. This lean approach to synthesis allows for better resource allocation and improved return on investment for production facilities.
- Enhanced Supply Chain Reliability: Sourcing strategies are significantly de-risked by relying on foundational chemicals that are produced globally in massive quantities, ensuring continuous availability even during regional disruptions. The robustness of the reaction conditions means that production is less likely to be halted by minor deviations in raw material specifications, providing greater flexibility in vendor selection. Shorter synthesis routes inherently reduce the cumulative lead time required to produce finished goods, enabling faster response to customer demand fluctuations. This agility is crucial for maintaining service levels in the fast-moving consumer goods sector where flavor trends can shift rapidly.
- Scalability and Environmental Compliance: The green nature of this process aligns with increasingly stringent environmental regulations, reducing the regulatory burden and permitting timelines for new production lines. The absence of heavy metals simplifies wastewater treatment protocols, lowering the cost of environmental compliance and minimizing the ecological impact of the facility. The process is inherently scalable, as the exotherms are manageable and the reaction kinetics are favorable for large-volume reactors, facilitating seamless expansion from pilot to commercial scale. This future-proofs the supply chain against growing sustainability mandates from downstream customers and regulatory bodies.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating this technology for potential adoption, we have compiled answers to common inquiries regarding the process specifics and quality attributes. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for feasibility assessments. Understanding these details is essential for R&D directors and quality assurance managers who are responsible for validating new supply sources. Clear communication regarding the technical capabilities of this route helps bridge the gap between laboratory innovation and commercial reality, fostering confidence among stakeholders.
Q: What are the primary advantages of this synthesis route over traditional methods?
A: This method avoids expensive transition metal catalysts like Ruthenium and toxic selenium reagents, utilizing environmentally friendly hydrogen peroxide instead, which significantly simplifies waste treatment and reduces raw material costs.
Q: What is the overall yield efficiency of the patented process?
A: The process demonstrates high efficiency with an 85.6% yield in the oxidation step and an 88.0% yield in the elimination step, ensuring robust material throughput for commercial production.
Q: Is the starting material readily available for large-scale manufacturing?
A: Yes, the process utilizes (E)-3-nonenoic acid, which is a commercially accessible feedstock, thereby enhancing supply chain reliability and reducing lead times compared to routes requiring complex custom synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Nonene-Gamma-Lactone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of delivering high-quality flavor intermediates that meet the exacting standards of the global food and fragrance industries. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of 2-nonene-gamma-lactone performs optimally in your final formulations. Our expertise in green chemistry allows us to implement efficient processes like the one described in CN102702146A, delivering value through both superior product quality and sustainable manufacturing practices.
We invite you to engage with our technical procurement team to discuss how we can tailor our production capabilities to your specific needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this optimized supply chain. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive your product development forward. Partner with us to secure a reliable, cost-effective, and high-quality supply of this essential flavor compound.
