Advanced Manufacturing of Carbapenem Intermediates via Novel Acid Addition Salt Technology
The pharmaceutical industry continuously seeks robust methodologies for the production of beta-lactam antibiotics, particularly carbapenems, due to their critical role in treating multi-drug resistant bacterial infections. Patent CN101848911A introduces a transformative approach to synthesizing key synthetic intermediates for these life-saving drugs, specifically focusing on the preparation of acid addition salts. Historically, the manufacturing of carbapenem precursors has been plagued by instability issues and complex purification requirements that hinder large-scale commercialization. This intellectual property details a method to prepare acid addition salts of carbapenem synthetic intermediates, notably compounds of Formula 2 and Formula 3, with exceptional yield and purity profiles. By shifting from traditional free base isolation to direct salt crystallization, the process circumvents the need for resource-intensive column chromatography. This technical breakthrough not only enhances the chemical stability of the intermediates by converting them into solid forms but also aligns perfectly with the demands of modern Good Manufacturing Practice (GMP) facilities seeking reliable carbapenem intermediates supplier partnerships for scalable production.
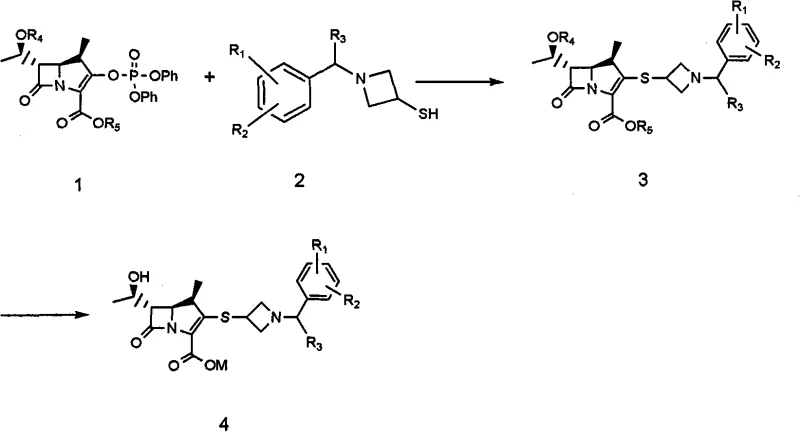
The limitations of conventional methods for producing carbapenem intermediates are well-documented in prior art, often involving the isolation of the intermediate as a free base which exists as a liquid at room temperature. This physical state presents significant logistical challenges, including difficulties in accurate weighing, transfer between reactors, and long-term storage without degradation. Furthermore, the purification of these liquid intermediates traditionally necessitates column chromatography, a technique that is notoriously difficult to scale up for industrial manufacturing due to high solvent consumption, low throughput, and significant waste generation. In contrast, the novel approach described in the patent leverages the chemical properties of acid addition salts to overcome these barriers. By adjusting the pH of the reaction mixture to a specific acidic range, typically between 1 and 5, the intermediate precipitates as a crystalline solid. This transition from liquid to solid state fundamentally changes the handling characteristics, allowing for standard filtration and drying operations instead of complex distillation or chromatographic separation. The result is a process that is inherently more suitable for the commercial scale-up of complex pharmaceutical intermediates, ensuring consistent quality and reduced operational complexity.
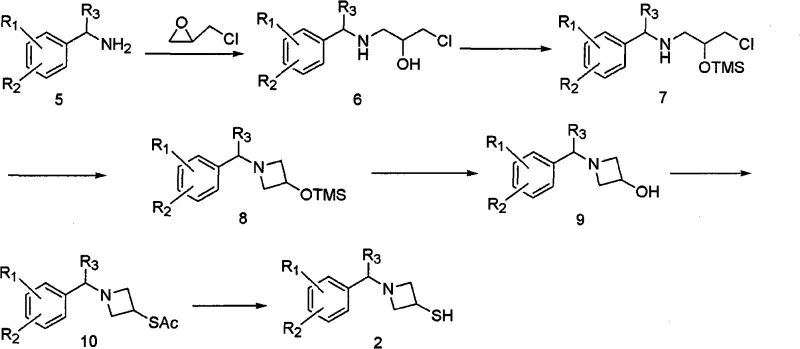
Mechanistic insights into the formation of these acid addition salts reveal a sophisticated control over solubility and crystallization kinetics. The process involves reacting a phosphonate ester precursor with a thiol-containing azetidine derivative in an anhydrous organic solvent such as acetonitrile or dichloromethane. Following the coupling reaction, the introduction of an aqueous-organic mixed solvent system allows for the precise manipulation of the partition coefficient of the product. Acidification using inorganic acids like hydrochloric acid, sulfuric acid, or phosphoric acid protonates the basic nitrogen center of the azetidine ring, forming a stable salt that is less soluble in the organic phase. This drives the equilibrium towards crystallization, effectively purifying the molecule from unreacted starting materials and by-products without the need for silica gel adsorption. Additionally, the patent highlights an improved route for synthesizing the azetidine thiol precursor itself, bypassing the need for silylation protection groups. By reacting the chlorohydrin intermediate directly with a base in a refluxing solvent, the azetidine ring closes efficiently. This simplification reduces the number of synthetic steps and eliminates the use of expensive silylating reagents, contributing to cost reduction in pharmaceutical manufacturing while maintaining high stereochemical integrity essential for antibiotic activity.
Understanding the precise operational parameters for synthesizing these high-purity carbapenem intermediates is crucial for technology transfer and process validation. The patent outlines a sequence where temperature control, specifically maintaining reactions between -20°C and 0°C during the coupling phase, is vital to prevent epimerization or degradation of the sensitive beta-lactam core. Subsequent work-up procedures involve careful pH adjustments to ensure the selective precipitation of the desired salt form, whether it be a hydrochloride, sulfate, or phosphate. The detailed standardized synthesis steps see the guide below provide a roadmap for replicating these results in a pilot or production environment, ensuring that the critical quality attributes defined in the intellectual property are met consistently.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this chromatography-free synthesis route offers substantial strategic advantages regarding cost efficiency and supply continuity. The elimination of column chromatography removes a major bottleneck in the production schedule, significantly reducing the cycle time required to manufacture each batch of intermediates. This efficiency gain translates directly into enhanced supply chain reliability, as facilities can respond more rapidly to fluctuations in market demand for antibiotic raw materials. Moreover, the conversion of the intermediate into a solid acid addition salt drastically simplifies logistics and warehousing. Solid materials are far easier to package, ship, and inventory compared to liquid oils, which often require specialized containers and climate-controlled storage to prevent oxidation or hydrolysis. This physical stability ensures that the material retains its specification throughout the distribution network, reducing the risk of rejected shipments and production delays at the downstream API manufacturing site.
- Cost Reduction in Manufacturing: The removal of column chromatography from the purification workflow results in significant operational expenditure savings. Chromatography requires large volumes of high-purity solvents and expensive stationary phases, both of which contribute heavily to the cost of goods sold. By replacing this with a crystallization-based purification, the consumption of these consumables is drastically reduced. Furthermore, the simplified synthesis of the azetidine precursor, which avoids silylation steps, lowers the raw material costs by eliminating the need for costly protecting group reagents and the associated waste disposal fees. These cumulative efficiencies allow for a more competitive pricing structure without compromising the stringent purity specifications required for pharmaceutical grade materials.
- Enhanced Supply Chain Reliability: The robustness of the solid salt form enhances the overall resilience of the supply chain. Liquid intermediates are prone to degradation over time, which can lead to inventory write-offs and urgent, unplanned production runs to replace spoiled stock. The stable crystalline nature of the acid addition salts extends the shelf life of the inventory, allowing for safer stockpiling and better planning against demand surges. Additionally, the use of water in the initial steps of the side-chain synthesis reduces reliance on volatile organic solvents, mitigating risks associated with solvent supply shortages or regulatory restrictions on emissions, thereby ensuring a more consistent and uninterrupted flow of materials.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this process represents a greener alternative to traditional methods. The reduction in solvent usage and the elimination of silica gel waste align with increasingly strict environmental regulations governing chemical manufacturing. The ability to perform crystallization in standard reactor vessels without specialized chromatography columns facilitates easier scale-up from laboratory to multi-ton production scales. This scalability ensures that the manufacturing process can grow in tandem with the commercial success of the final antibiotic drug, providing a sustainable long-term sourcing solution that meets both economic and ecological compliance standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on how this method improves upon existing industry standards. Understanding these details helps stakeholders evaluate the feasibility of integrating this intermediate into their existing supply chains and production workflows.
Q: Why is the acid addition salt form preferred over the free base for carbapenem intermediates?
A: The free base form of the intermediate is typically a liquid, which makes it difficult to handle, store, and transport on an industrial scale. The acid addition salt converts this into a stable solid crystalline form, significantly improving operational safety and shelf-life stability.
Q: How does this process eliminate the need for column chromatography?
A: Traditional methods rely on column chromatography to purify the liquid free base, which limits throughput. This novel method utilizes pH-controlled crystallization of the acid addition salt, achieving high purity (98% or higher) through simple filtration, making it suitable for mass production.
Q: What are the environmental benefits of the updated azetidine ring formation step?
A: The improved process allows for the formation of the azetidine ring from the chlorohydrin precursor using aqueous conditions or simplified base treatment without silylation protection groups. This reduces the consumption of volatile organic solvents and hazardous silylating reagents.
- React the phosphonate precursor with the azetidine thiol intermediate in anhydrous organic solvent at low temperature.
- Acidify the reaction mixture to pH 1-5 using inorganic acid to form the acid addition salt directly in the organic layer.
- Induce crystallization by adding anti-solvent, followed by filtration and drying to obtain the solid intermediate salt.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbapenem Intermediates Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the global fight against antibiotic-resistant bacteria. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the innovative processes described in patents like CN101848911A can be successfully translated into industrial reality. We maintain stringent purity specifications and operate rigorous QC labs equipped to analyze complex beta-lactam structures, guaranteeing that every batch of carbapenem intermediate meets the exacting standards required by top-tier pharmaceutical companies. Our commitment to quality assurance means that clients can trust our materials to perform consistently in their downstream API synthesis, minimizing the risk of batch failures.
We invite potential partners to engage with our technical procurement team to discuss how these advanced manufacturing capabilities can benefit your specific projects. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how switching to our chromatography-free intermediates can optimize your production budget. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your volume requirements, ensuring a seamless integration of our high-purity carbapenem intermediates into your supply chain.
