Advanced Synthesis of Tridentate Pyridine Imine Ligands for High-Efficiency Ethylene Oligomerization Catalysts
Advanced Synthesis of Tridentate Pyridine Imine Ligands for High-Efficiency Ethylene Oligomerization Catalysts
The global demand for linear alpha-olefins (LAO) continues to surge, driven by their critical role as comonomers in polyethylene production and as precursors for surfactants and lubricants. At the heart of efficient LAO production lies the catalyst system, specifically the ligand architecture that governs selectivity and activity. Patent CN101580488B introduces a breakthrough preparation method for tridentate pyridine imine ligands, utilizing titanium tetrachloride (TiCl4) as a catalyst under controlled micro-positive pressure. This innovation addresses long-standing challenges in ligand synthesis, offering a robust pathway to high-purity intermediates essential for next-generation iron and cobalt-based oligomerization catalysts. For R&D directors and procurement specialists seeking reliable industrial chemical suppliers, this technology represents a significant leap forward in process efficiency and product consistency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of tridentate pyridine imine ligands has relied on condensation reactions catalyzed by protic acids such as formic acid, acetic acid, or p-toluenesulfonic acid, as documented by research groups like Brookhart and Gibson. While effective to a degree, these conventional methods often suffer from suboptimal yields and limited control over reaction kinetics, particularly when scaling up for commercial applications. The use of protic acids can sometimes lead to side reactions or incomplete condensation, necessitating extensive purification steps that erode overall process economics. Furthermore, comparative data indicates that performing these reactions under normal atmospheric pressure without precise control over catalyst addition can result in yields hovering around 79%, which is insufficient for cost-sensitive high-volume manufacturing environments where every percentage point of yield impacts the bottom line.
The Novel Approach
The novel approach detailed in the patent data revolutionizes this synthesis by employing titanium tetrachloride (TiCl4) as a Lewis acid catalyst within an inert atmosphere maintained at a micro-positive pressure of 0.11 to 0.15 MPa. This specific combination of parameters creates a highly favorable thermodynamic environment for the condensation of 2,6-diacetylpyridine with various aniline or naphthylamine derivatives. By strictly controlling the addition rate of the TiCl4 catalyst (e.g., 0.03 ml/min to 3 ml/min) and utilizing molecular sieves to sequester water byproducts, the process achieves ligand yields exceeding 80%. This method not only enhances the crude yield but also simplifies the downstream purification workflow, as the product can be effectively isolated through rotary evaporation and recrystallization, thereby establishing a new benchmark for cost reduction in industrial chemical manufacturing.
Mechanistic Insights into TiCl4-Catalyzed Ligand Formation and Metal Coordination
The mechanistic superiority of this synthesis lies in the activation of the carbonyl group on the 2,6-diacetylpyridine by the strong Lewis acidity of TiCl4. This activation facilitates a nucleophilic attack by the amine nitrogen of the aniline derivative, promoting the formation of the imine bond (C=N) with high fidelity. The maintenance of a micro-positive pressure inert atmosphere (using nitrogen or argon) is critical, as it prevents the hydrolysis of the sensitive TiCl4 catalyst and protects the forming imine bonds from moisture-induced degradation. This rigorous exclusion of oxygen and water ensures that the resulting ligand possesses the structural integrity required for subsequent metal coordination, minimizing the formation of oxidized impurities that could poison the final oligomerization catalyst.
Once synthesized, these tridentate ligands coordinate with iron or cobalt halides to form the active catalytic species. The steric and electronic properties of the ligand substituents (R groups) play a pivotal role in defining the catalyst's performance. For instance, ligands featuring ortho-methyl groups create a specific steric environment around the metal center that favors the formation of linear alpha-olefins over branched isomers or polymers. 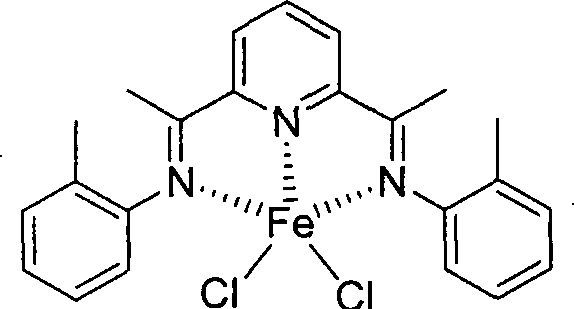 As illustrated in the structure above, the iron center is coordinated by the three nitrogen atoms of the ligand and two chloride ions, forming a distorted geometry that is optimal for ethylene insertion. Variations in the ligand structure, such as the introduction of electron-donating methoxy groups shown below, further tune the electron density at the metal center, allowing for precise control over the oligomerization activity and product distribution.
As illustrated in the structure above, the iron center is coordinated by the three nitrogen atoms of the ligand and two chloride ions, forming a distorted geometry that is optimal for ethylene insertion. Variations in the ligand structure, such as the introduction of electron-donating methoxy groups shown below, further tune the electron density at the metal center, allowing for precise control over the oligomerization activity and product distribution. 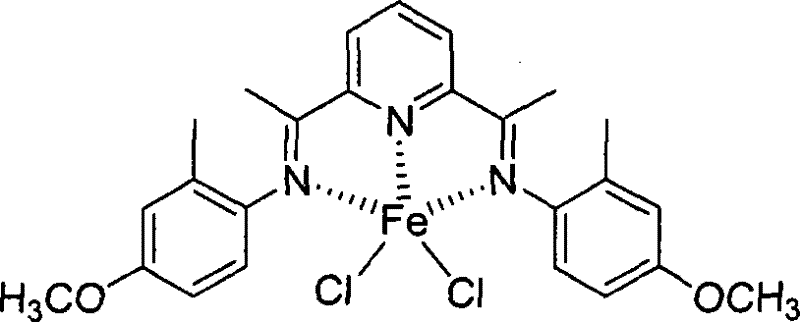 This tunability is essential for producing high-purity OLED material precursors or specialized polymer additives where specific chain lengths are required.
This tunability is essential for producing high-purity OLED material precursors or specialized polymer additives where specific chain lengths are required.
How to Synthesize Tridentate Pyridine Imine Ligand Efficiently
The synthesis protocol outlined in the patent provides a clear, reproducible roadmap for laboratory and pilot-scale production. The process begins with the charging of 2,6-diacetylpyridine and the chosen aniline derivative into a reactor containing an organic solvent such as tetrahydrofuran or toluene, along with molecular sieves. The system is then pressurized with inert gas, and the TiCl4 catalyst is introduced dropwise while maintaining reflux conditions. Following the reaction period of 6 to 72 hours, the workup involves solvent removal, washing, and recrystallization to isolate the pure ligand. The detailed standardized synthesis steps see the guide below.
- Combine 2,6-diacetylpyridine and aniline derivatives in an organic solvent with molecular sieves as a water-retaining agent under an inert gas atmosphere.
- Slowly add titanium tetrachloride (TiCl4) catalyst dropwise while maintaining a micro-positive pressure of 0.11 to 0.15 MPa at reflux temperature for 6 to 72 hours.
- Remove solvent via rotary evaporation, filter the product, wash with organic solvents and water, and finally recrystallize the head product to obtain the ligand with >80% yield.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this TiCl4-catalyzed synthesis route offers compelling strategic advantages beyond mere technical performance. The primary benefit is the substantial improvement in process yield, which directly translates to lower raw material consumption per kilogram of finished ligand. By consistently achieving yields over 80%, manufacturers can significantly reduce the cost of goods sold (COGS), making the final ethylene oligomerization catalyst more competitive in the global market. Additionally, the use of common organic solvents and commercially available starting materials like 2,6-diacetylpyridine and substituted anilines ensures a stable and resilient supply chain, mitigating the risks associated with sourcing exotic or restricted reagents.
- Cost Reduction in Manufacturing: The elimination of complex purification stages and the high crude yield achieved through the TiCl4 method drastically simplify the production workflow. Unlike traditional methods that may require extensive chromatography or multiple recrystallizations to remove acid residues, this novel approach allows for efficient isolation via filtration and washing. This streamlining of unit operations reduces energy consumption, labor hours, and solvent waste, leading to significant operational expenditure savings without compromising the stringent purity specifications required for high-performance catalysis.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, specifically the tolerance for various aniline derivatives and the use of standard inert gas protection, ensures consistent batch-to-batch quality. This reliability is crucial for reducing lead time for high-purity catalyst intermediates, as it minimizes the risk of batch failures or off-spec material that could disrupt downstream catalyst production schedules. Furthermore, the scalability of the process means that suppliers can rapidly ramp up production volumes to meet fluctuating market demands for linear alpha-olefin catalysts.
- Scalability and Environmental Compliance: From an environmental and safety perspective, the closed-system nature of the reaction under micro-positive pressure minimizes the release of volatile organic compounds (VOCs) and hazardous vapors. The ability to recycle solvents like toluene or diethyl ether after rotary evaporation further aligns the process with green chemistry principles. This environmental compliance not only reduces waste disposal costs but also future-proofs the manufacturing facility against increasingly stringent regulatory frameworks governing industrial chemical production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ligand synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for evaluating the feasibility of adopting this method in your own production facilities or supply networks.
Q: What are the advantages of using TiCl4 over traditional acid catalysts for ligand synthesis?
A: According to patent CN101580488B, using TiCl4 under micro-positive pressure significantly improves reaction control and yield compared to formic or acetic acid methods, achieving yields over 80% versus roughly 79% in comparative examples.
Q: What is the selectivity of the linear alpha-olefins produced using these catalysts?
A: The ethylene oligomerization catalysts prepared from these ligands demonstrate exceptional selectivity, producing linear alpha-olefins with a selectivity greater than 96%, with a carbon number distribution primarily between C6 and C20.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the process utilizes standard unit operations such as reflux, filtration, and recrystallization, and employs readily available raw materials, making it highly scalable for industrial manufacturing of specialty chemical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tridentate Pyridine Imine Ligand Supplier
As the chemical industry evolves towards more efficient and selective catalytic processes, the demand for high-quality ligand precursors has never been greater. NINGBO INNO PHARMCHEM stands at the forefront of this transition, leveraging deep expertise in coordination chemistry to deliver superior tridentate pyridine imine ligands. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous volume requirements of global catalyst manufacturers. We operate with stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of ligand meets the exacting standards necessary for high-activity ethylene oligomerization.
We invite you to collaborate with us to optimize your catalyst supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific production needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced synthesis capabilities can drive value and efficiency in your operations.
