Advanced Synthesis of Menthyl Glycol Ester: A Breakthrough in Cooling Agent Intermediate Manufacturing
The landscape of cooling agent intermediate manufacturing is undergoing a significant transformation driven by the need for higher purity and more sustainable processing methods. Patent CN107793315B introduces a groundbreaking synthesis method for menthyl glycol ester, a critical precursor in the production of high-performance cooling agents like WS-3 and WS-23 used extensively in the flavor, fragrance, and personal care industries. This innovative approach utilizes p-toluenesulfonyl chloride (TsCl) activation to facilitate the esterification of menthanecarboxylic acid with ethylene glycol, bypassing the severe limitations of traditional acyl chloride or sodium salt routes. By operating under mild temperature conditions ranging from -10°C to 25°C during the activation phase, the process minimizes thermal degradation and ensures exceptional selectivity. The strategic use of TsCl not only enhances reaction kinetics but also fundamentally alters the impurity profile, offering a robust solution for manufacturers seeking to optimize their production lines for complex fine chemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of menthyl glycol esters has been plagued by inefficient pathways that compromise both yield and product quality. One prevalent conventional method involves converting menthyl formic acid into an acyl chloride intermediate using aggressive reagents, which subsequently reacts with glycol; however, this route frequently triggers unwanted coupling reactions that generate substantial byproduct loads and drastically reduce overall yield. Another common technique employs the formation of a sodium salt from menthyl formate followed by reaction with chloroethanol under pyridine catalysis, but this approach is notoriously unstable due to the propensity of chlorohydrin to undergo cyclization reactions in the presence of base. These cyclization events lead to the formation of cyclic ethers rather than the desired linear ester, resulting in extremely low conversion rates that can render the process commercially unviable. Furthermore, the harsh conditions required for acyl chloride formation often necessitate specialized corrosion-resistant equipment and rigorous safety protocols, inflating capital expenditure and operational complexity for production facilities.
The Novel Approach
In stark contrast to these legacy methods, the novel TsCl-mediated pathway described in the patent offers a streamlined, one-step synthesis that effectively circumvents the pitfalls of cyclization and coupling. By utilizing p-toluenesulfonyl chloride as an activating agent in the presence of pyridine, the reaction proceeds through a highly reactive mixed anhydride or activated ester intermediate that is rapidly intercepted by the nucleophilic ethylene glycol. This mechanism ensures that the esterification occurs swiftly and selectively before side reactions can take place, leveraging the excellent leaving group ability of the tosylate moiety. The process operates under significantly milder thermal conditions, initially maintaining temperatures between -10°C and 25°C to control exotherms, followed by a moderate heating phase to drive completion. This gentle yet effective protocol not only simplifies the operational workflow but also dramatically improves the purity of the crude product, thereby reducing the burden on downstream purification units.
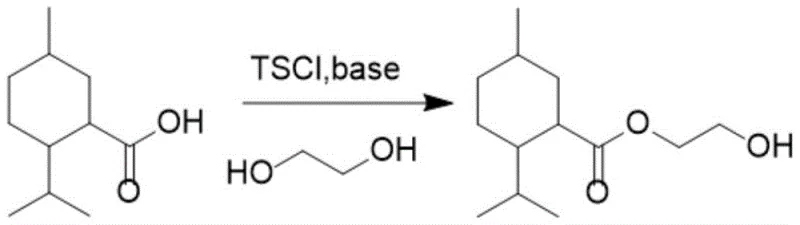
Mechanistic Insights into TsCl-Mediated Esterification
The core of this technological advancement lies in the sophisticated mechanistic role played by p-toluenesulfonyl chloride within the reaction matrix. Upon addition to the mixture of menthanecarboxylic acid and pyridine, TsCl reacts to form an activated species, likely a mixed anhydride or an acyl pyridinium salt, which possesses a much higher electrophilicity than the parent carboxylic acid. The bulky nature of the p-toluenesulfonyl group provides a critical steric shield that prevents the ethylene glycol from engaging in self-epoxidation or intermolecular crosslinking, which are common failure modes in glycol esterifications. As the ethylene glycol attacks the activated carbonyl carbon, the tosylate group departs as a stable anion, driving the equilibrium forward towards the formation of the menthyl glycol ester. This precise control over the reaction trajectory ensures that the hydroxyl groups of the glycol remain available for the intended esterification rather than participating in deleterious side reactions, thereby securing high selectivity even in the presence of excess diol.
Impurity control is further enhanced by the specific workup procedure designed to remove residual reagents and byproducts efficiently. Following the reaction, the mixture is treated with hydrochloric acid to adjust the pH to between 1 and 3, which effectively protonates and solubilizes the pyridine base and any unreacted amine species into the aqueous phase. Subsequent neutralization with potassium or sodium hydroxide to a pH of 13 ensures the removal of any acidic impurities or unreacted starting materials that might interfere with the final product stability. The use of non-polar solvents like n-hexane or toluene for extraction allows for the selective partitioning of the lipophilic menthyl glycol ester into the organic layer, leaving polar salts and inorganic residues behind. This rigorous purification strategy, combined with the inherent selectivity of the TsCl activation, results in a final product with exceptional chemical purity, minimizing the need for energy-intensive distillation columns or chromatographic separation techniques.
How to Synthesize Menthyl Glycol Ester Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for replicating this high-yield process in a commercial setting, emphasizing precise stoichiometric control and temperature management. Operators must carefully dissolve the menthanecarboxylic acid and ethylene glycol in a suitable solvent such as toluene or tetrahydrofuran before initiating the activation sequence with TsCl. The detailed standardized synthesis steps below outline the exact molar ratios and thermal profiles required to achieve the reported yields of over 80%, ensuring consistency across different batch sizes.
- Dissolve menthanecarboxylic acid and ethylene glycol in a solvent like toluene and cool the mixture to between -10°C and 25°C.
- Add pyridine as a base, then slowly drip in a solution of p-toluenesulfonyl chloride (TsCl) while maintaining temperature control.
- Raise the reaction temperature to 45-90°C for 2-8 hours, followed by acid-base workup and distillation to isolate the pure ester.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this TsCl-based synthesis route presents a compelling value proposition centered around operational efficiency and cost predictability. By eliminating the need for hazardous acyl chloride precursors or unstable sodium salt intermediates, the process reduces the reliance on specialized raw materials that are subject to volatile market pricing and supply disruptions. The simplified one-pot nature of the reaction minimizes unit operations, translating directly into reduced labor costs and lower energy consumption per kilogram of product produced. Furthermore, the high selectivity of the reaction means that less raw material is wasted on byproduct formation, optimizing the atom economy and reducing the volume of chemical waste that requires treatment and disposal. These factors collectively contribute to a more resilient and cost-effective supply chain capable of meeting the demanding specifications of the global flavor and fragrance market.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and the avoidance of multi-step activation sequences significantly lowers the direct material costs associated with production. By utilizing commodity chemicals like TsCl and pyridine instead of proprietary or exotic reagents, manufacturers can leverage established supply networks to secure favorable pricing. Additionally, the high yield exceeding 80% ensures that the throughput of the reactor is maximized, effectively spreading fixed overhead costs over a larger volume of saleable product. The simplified workup procedure also reduces the consumption of acids, bases, and solvents during purification, further driving down the variable costs per unit.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route against minor fluctuations in reaction conditions makes it highly suitable for large-scale manufacturing where process consistency is paramount. Unlike methods prone to sudden cyclization failures, this TsCl-mediated process offers a predictable reaction profile that minimizes the risk of batch rejection and production delays. The use of common solvents like toluene and n-hexane ensures that raw material availability remains stable even during periods of global supply chain stress. This reliability allows supply chain planners to maintain leaner inventory levels while still guaranteeing on-time delivery to downstream customers who depend on continuous flows of cooling agent intermediates.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of heavy metal contaminants simplify the environmental compliance landscape for manufacturing facilities. Scaling this process from pilot plant to commercial production does not require exponential increases in safety infrastructure, as the thermal hazards are well-managed within the specified temperature ranges. The aqueous waste streams generated during the acid-base workup are relatively straightforward to treat compared to those containing heavy metals or persistent organic pollutants. This alignment with green chemistry principles not only reduces regulatory burdens but also enhances the sustainability profile of the final product, appealing to end-users who prioritize eco-friendly sourcing in their supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method, derived directly from the patent specifications and industry best practices. Understanding these details is crucial for R&D teams evaluating the feasibility of technology transfer and for procurement specialists assessing the long-term viability of this supply source. The answers provided reflect the specific advantages of the TsCl activation strategy over traditional esterification techniques.
Q: Why is TsCl preferred over thionyl chloride for this esterification?
A: TsCl activation avoids the harsh conditions and corrosive byproducts associated with acyl chloride formation using thionyl chloride, leading to milder reaction profiles and easier purification.
Q: How does this method prevent ethylene glycol cyclization?
A: The bulky p-toluenesulfonyl group acts as a superior leaving group that facilitates rapid esterification before the ethylene glycol can undergo intramolecular cyclization or self-epoxidation side reactions.
Q: What is the typical yield for this synthesis route?
A: According to patent data, this optimized protocol consistently achieves yields exceeding 80%, significantly outperforming traditional sodium salt methods which often suffer from low conversion rates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Menthyl Glycol Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the fine chemical sector. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering high-purity menthyl glycol ester that meets stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical instrumentation to verify every batch. Our capability to implement the TsCl-mediated synthesis described in CN107793315B allows us to offer a product with superior consistency and reduced impurity profiles compared to market alternatives.
We invite potential partners to engage with our technical procurement team to discuss how this optimized route can benefit your specific application requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to our supply chain. We encourage you to contact us directly to obtain specific COA data and route feasibility assessments tailored to your volume needs, ensuring that your production of cooling agents and flavor intermediates remains uninterrupted and cost-efficient.
