Advanced Catalytic Fluorination Technology for High-Purity Polyfluorobenzonitrile Manufacturing
Introduction to Next-Generation Fluorination Technology
The global demand for high-purity polyfluorobenzonitrile derivatives has surged due to their critical role as intermediates in the synthesis of advanced pharmaceuticals, agrochemicals, and liquid crystal materials. Patent CN111004149B introduces a groundbreaking method for preparing these compounds through the catalytic fluorination of polychlorinated benzonitrile, addressing long-standing inefficiencies in traditional halogen exchange processes. This technology leverages electron-withdrawing substituted phenylboronic acid esters to activate fluoride salts, enabling reactions to proceed at significantly lower temperatures ranging from 80°C to 120°C. By shifting away from the extreme thermal conditions historically required for nucleophilic aromatic substitution, this innovation not only enhances safety profiles but also dramatically improves reaction selectivity and overall yield. For R&D directors and process engineers, this represents a pivotal opportunity to optimize synthetic routes for complex fluorinated aromatics, ensuring that production scales meet the rigorous quality standards of modern fine chemical manufacturing without compromising on economic viability or operational safety.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the direct fluorination of polychlorinated benzonitriles using potassium fluoride has been plagued by severe thermodynamic and kinetic barriers that necessitate harsh reaction conditions. Traditional protocols often require temperatures exceeding 300°C to overcome the high activation energy associated with displacing chlorine atoms on the electron-deficient benzene ring. Such extreme thermal environments impose immense stress on reactor vessels, requiring specialized high-pressure equipment and expensive oil-bath heating systems that are far more complex and costly to operate than standard steam heating infrastructure. Furthermore, high-temperature reactions invariably lead to increased side reactions, including the formation of tars and decomposition products, which severely compromise the purity of the final product and complicate downstream purification processes. The combination of high energy consumption, significant safety risks associated with high-pressure operations, and low selectivity has made conventional fluorination a bottleneck in the supply chain for critical fluorinated intermediates, driving up costs and limiting scalability for commercial manufacturers.
The Novel Approach
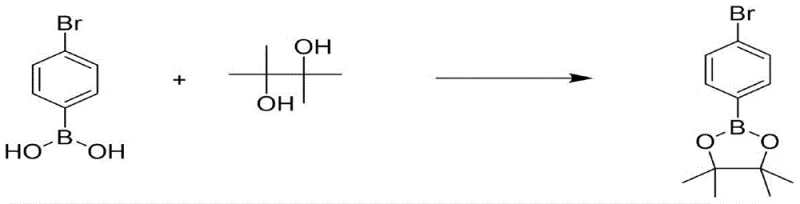
In stark contrast to legacy methods, the novel catalytic approach disclosed in CN111004149B utilizes a sophisticated boron-mediated activation strategy that fundamentally alters the reaction landscape. By introducing electron-withdrawing substituted phenylboronic acid esters, such as p-bromophenylboronic acid pinacol ester, the system forms a highly reactive complex with the fluoride salt that facilitates nucleophilic attack at much milder temperatures between 80°C and 120°C. This drastic reduction in operating temperature allows for the use of standard steam heating systems, eliminating the need for costly oil baths and reducing the overall energy footprint of the manufacturing process. Additionally, the integration of reactive distillation, where the product is continuously removed from the reaction mixture as it forms, drives the equilibrium forward according to Le Chatelier's principle, resulting in exceptional conversion rates and yields exceeding 94%. This method effectively solves the technical difficulties of low conversion and medium selectivity often encountered with low-activity mononitrile compounds, providing a robust and scalable solution for industrial production.
Mechanistic Insights into Boron-Mediated Catalytic Fluorination
The core innovation of this technology lies in the unique interaction between the electron-withdrawing phenylboronic acid ester and the fluoride source, which generates a transient, highly nucleophilic fluorinating species. Unlike simple fluoride salts which are often poorly soluble and kinetically sluggish in organic media, the boron-fluoride complex exhibits enhanced solubility and reactivity, allowing for efficient displacement of chlorine substituents on the aromatic ring. The electron-withdrawing nature of the substituents on the phenylboronic acid ester, such as bromine or nitro groups, further increases the Lewis acidity of the boron center, thereby strengthening the B-F bond interaction and facilitating the transfer of fluoride to the substrate. This mechanistic pathway bypasses the high-energy transition states typical of uncatalyzed halogen exchange, enabling the reaction to proceed smoothly under mild thermal conditions. For process chemists, understanding this catalytic cycle is crucial for optimizing catalyst loading and selecting the appropriate boronate ester to maximize turnover frequency and minimize catalyst consumption during large-scale campaigns.
Furthermore, the impurity control mechanism inherent in this process is driven by the combination of mild reaction temperatures and continuous product removal. In traditional high-temperature fluorination, thermal degradation and polymerization of the nitrile group or the aromatic ring are common, leading to complex impurity profiles that are difficult to separate. By maintaining the reaction temperature below 120°C, the thermal stability of both the substrate and the product is preserved, significantly reducing the formation of tar and heavy byproducts. The simultaneous rectification of the product ensures that the desired polyfluorobenzonitrile is immediately separated from the reaction zone, preventing it from undergoing further unwanted side reactions or reversible de-fluorination. This dynamic separation technique not only boosts yield but also simplifies the workup procedure, as the crude distillate obtained is already of high purity, often exceeding 99.97%, thus reducing the burden on final crystallization or chromatography steps and ensuring a cleaner impurity profile for downstream pharmaceutical applications.
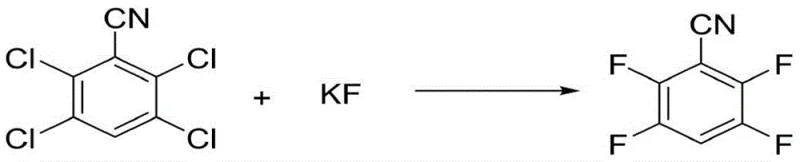
How to Synthesize Polyfluorobenzonitrile Efficiently
The implementation of this catalytic fluorination route requires precise control over reaction parameters to fully realize its potential for cost reduction in fine chemical manufacturing. The process begins with the activation of the catalyst system, where fluoride salt, organic solvent, and the phenylboronic acid ester are heated together to form the active species before the substrate is introduced. Following activation, the polychlorinated benzonitrile is added, and the mixture is maintained at 80-120°C while vacuum is applied to distill off the product as it forms. This continuous operation mode allows for the supplementation of fresh substrate and fluoride salt in subsequent batches without replacing the solvent or catalyst, creating a semi-continuous loop that maximizes resource efficiency. Detailed standardized synthesis steps see the guide below.
- Activate the catalyst system by heating fluoride salt, organic solvent, and substituted phenylboronic acid ester to 110-120°C under nitrogen protection.
- Add polychlorinated benzonitrile to the activated mixture and maintain temperature at 80-120°C while simultaneously distilling the product to drive the reaction forward.
- Supplement additional polychlorinated benzonitrile and potassium fluoride for subsequent batches, utilizing solvent recycling to maximize efficiency and yield.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this catalytic fluorination technology offers transformative advantages that extend far beyond simple yield improvements. The ability to operate at significantly lower temperatures translates directly into reduced utility costs, as the process can utilize standard low-pressure steam rather than expensive thermal oil systems, which require higher maintenance and energy inputs. Moreover, the extended lifetime of reactor equipment due to reduced thermal stress lowers capital expenditure on replacements and repairs, contributing to a more stable and predictable operational budget. The simplicity of the post-treatment process, which involves basic centrifugation and solvent recycling, minimizes labor hours and waste disposal costs, further enhancing the overall economic attractiveness of the route. These factors combine to create a supply chain that is not only more cost-effective but also more resilient against fluctuations in energy prices and equipment availability.
- Cost Reduction in Manufacturing: The elimination of high-temperature oil bath heating systems in favor of standard steam heating represents a substantial decrease in both capital investment and ongoing operational energy costs. By avoiding the need for specialized high-temperature infrastructure, manufacturers can leverage existing general-purpose reactors, thereby accelerating the timeline for technology transfer and scale-up. Additionally, the high catalytic activity allows for lower catalyst loading relative to the substrate, and the ability to recycle the solvent for multiple batches drastically reduces the consumption of expensive organic solvents. These cumulative efficiencies result in a significantly lower cost of goods sold (COGS), providing a competitive edge in the pricing of high-value fluorinated intermediates for the global market.
- Enhanced Supply Chain Reliability: The robustness of this catalytic system ensures consistent production output, mitigating the risks of batch failures that are common in harsh, high-temperature processes. The use of commercially available and stable reagents, such as potassium fluoride and readily synthesized phenylboronic acid esters, secures the raw material supply chain against volatility. Furthermore, the simplified purification process reduces the dependency on complex downstream processing units, minimizing bottlenecks that could delay shipment to customers. This reliability is critical for maintaining just-in-time delivery schedules for pharmaceutical clients who require uninterrupted supplies of key intermediates to support their own drug substance manufacturing timelines.
- Scalability and Environmental Compliance: From an environmental perspective, the lower energy consumption and reduced generation of tarry waste align perfectly with modern green chemistry principles and regulatory requirements. The process generates minimal hazardous waste, as the inorganic salts produced can be easily separated and disposed of, and the organic solvent can be recovered and reused with high efficiency. This reduced environmental footprint simplifies the permitting process for new production lines and ensures compliance with increasingly stringent environmental regulations. The ease of scaling this reaction from laboratory glassware to multi-ton industrial reactors without significant re-engineering makes it an ideal candidate for rapid commercialization to meet growing market demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalytic fluorination technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent literature, providing a reliable foundation for decision-making. Understanding these details is essential for evaluating the feasibility of integrating this process into existing manufacturing portfolios. We encourage technical teams to review these points closely to assess the alignment with their specific production goals and quality requirements.
Q: What is the primary advantage of using phenylboronic acid esters in this fluorination process?
A: The use of electron-withdrawing phenylboronic acid esters significantly lowers the activation energy required for the fluorination reaction, allowing the process to proceed at 80-120°C instead of the traditional >300°C, thereby reducing energy consumption and equipment stress.
Q: Can the solvent be recycled in this catalytic system?
A: Yes, the patent explicitly details a solvent recycling protocol where benzonitrile or sulfolane can be recovered and reused for 6-10 cycles after centrifugation of inorganic salts, drastically reducing waste and raw material costs.
Q: What purity levels are achievable with this method?
A: Experimental data within the patent demonstrates that this method consistently yields polyfluorobenzonitrile with purity levels exceeding 99.97%, meeting the stringent specifications required for pharmaceutical and electronic grade applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polyfluorobenzonitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of polyfluorobenzonitrile as a versatile building block for next-generation pharmaceuticals and agrochemicals. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from bench-scale discovery to full-scale manufacturing. Our facilities are equipped with state-of-the-art reactors capable of handling the precise temperature and pressure controls required for this advanced catalytic fluorination, guaranteeing stringent purity specifications for every batch. With our rigorous QC labs and commitment to process excellence, we deliver high-purity polyfluorobenzonitrile that meets the exacting standards of the global life sciences industry, securing your supply chain against disruptions.
We invite you to collaborate with us to optimize your sourcing strategy and achieve significant operational efficiencies. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. By leveraging our expertise in catalytic fluorination, we can help you reduce lead time for high-purity polyfluorobenzonitrile and secure a stable supply of this critical intermediate. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our technological capabilities can drive value for your organization.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
