Advanced One-Pot Synthesis of Dibenzothiazole Disulfide and Triphenylphosphine for Pharmaceutical Manufacturing
Advanced One-Pot Synthesis of Dibenzothiazole Disulfide and Triphenylphosphine for Pharmaceutical Manufacturing
The pharmaceutical industry constantly seeks sustainable pathways to manage complex waste streams generated during the synthesis of critical active pharmaceutical ingredients (APIs). A groundbreaking approach detailed in patent CN101357908B offers a transformative solution for the simultaneous preparation of Dibenzothiazole Disulfide (DM) and Triphenylphosphine (TPP) through an efficient one-pot methodology. This innovation specifically targets the valorization of Triphenylphosphine Oxide (TPPO) and 2-Mercaptobenzothiazole (M), two significant byproducts often encountered in the production of cephalosporin active esters. By integrating waste recycling with high-value chemical synthesis, this technology not only mitigates severe environmental pollution associated with traditional disposal methods but also establishes a robust economic model for reliable pharmaceutical intermediates supplier networks. The process leverages Bis(trichloromethyl) carbonate (BTC) as a key activating agent, enabling the conversion of oxidized phosphine waste back into useful triphenylphosphine while concurrently oxidizing the mercaptan species into the disulfide dimer. This dual-output strategy represents a paradigm shift in green chemistry, aligning perfectly with the global demand for cost reduction in API manufacturing through circular economy principles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the regeneration of Triphenylphosphine from its oxide form (TPPO) has been a formidable technical challenge characterized by harsh reaction conditions and prohibitive costs. Traditional reduction protocols frequently rely on aggressive reagents such as trichlorosilane, diisobutylaluminum hydride, or complex silane derivatives, which pose significant safety risks due to their pyrophoric nature and moisture sensitivity. Furthermore, these conventional pathways often suffer from low atom economy, generating substantial quantities of stoichiometric waste that requires energy-intensive separation and disposal procedures. In parallel, the oxidation of 2-Mercaptobenzothiazole to its disulfide form typically employs hydrogen peroxide or nitrite-based oxidants, processes that can be operationally tedious and prone to over-oxidation side reactions. The separation of these distinct reaction streams necessitates multiple unit operations, increasing capital expenditure and extending production lead times. Consequently, many manufacturers have historically opted to discard TPPO and mercaptan wastes, leading to severe ecological burdens and lost economic potential, a practice that is increasingly untenable under modern environmental regulations.
The Novel Approach
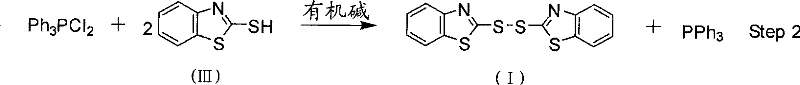
The methodology disclosed in the patent introduces a streamlined, one-pot synthetic route that elegantly bypasses the limitations of stepwise processing by coupling the reduction of TPPO and the oxidation of the mercaptan in a single reactor vessel. By utilizing Bis(trichloromethyl) carbonate (BTC) as a chlorinating agent, the process first converts TPPO into the reactive intermediate dichlorotriphenylphosphine (Ph3PCl2) under mild thermal conditions ranging from -30°C to 90°C. This activated phosphine species then reacts directly with 2-Mercaptobenzothiazole in the presence of an organic base, facilitating the formation of the disulfide bond while regenerating the valuable triphenylphosphine. This integrated approach eliminates the need for external reducing agents and separate oxidation steps, drastically simplifying the workflow. The result is a process with superior atom economy, where the oxygen atom from the phosphine oxide is effectively utilized to drive the oxidative coupling of the sulfur species. This innovation not only enhances the overall yield of both target compounds but also significantly reduces the solvent volume and energy consumption required per kilogram of product, marking a substantial advancement in commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into BTC-Mediated Phosphine Regeneration and Disulfide Coupling
The core of this technological breakthrough lies in the unique reactivity of Bis(trichloromethyl) carbonate (BTC) towards Triphenylphosphine Oxide. Under the specified reaction conditions, BTC acts as a source of phosgene equivalents, effectively deoxygenating the TPPO to generate the highly electrophilic dichlorotriphenylphosphine intermediate. This transformation is critical because it avoids the direct reduction of the P=O bond, which typically requires high-energy inputs. Instead, the process leverages the nucleophilicity of the 2-Mercaptobenzothiazole thiol group to attack the phosphorus center, displacing chloride ions. This nucleophilic attack triggers a cascade where the sulfur atoms are oxidized to form the disulfide linkage, simultaneously reducing the phosphorus species back to the trivalent state found in Triphenylphosphine. The presence of an organic base, such as triethylamine or pyridine, is essential to neutralize the hydrochloric acid byproduct formed during this exchange, driving the equilibrium towards the desired products. This mechanistic pathway ensures that the reaction proceeds with high selectivity, minimizing the formation of sulfonic acid byproducts or phosphine oxides that could contaminate the final stream.
Impurity control in this system is inherently robust due to the crystalline nature of the products and the orthogonality of the reaction phases. The Dibenzothiazole Disulfide typically precipitates out of the reaction mixture as a solid filter cake, allowing for easy physical separation from the soluble Triphenylphosphine remaining in the filtrate. This phase separation acts as a primary purification step, ensuring that the disulfide product achieves high purity levels, often exceeding 99% as indicated by HPLC analysis in the patent examples. The Triphenylphosphine in the filtrate can be further purified through simple solvent recovery and recrystallization techniques, yielding material with purity specifications suitable for sensitive catalytic applications. The choice of solvent systems, such as chlorobenzene or toluene, plays a pivotal role in managing the solubility profiles of the intermediates, ensuring that side reactions are suppressed and that the final products can be isolated with minimal downstream processing. This level of control is vital for maintaining the integrity of the supply chain for high-purity pharmaceutical intermediates.
How to Synthesize Dibenzothiazole Disulfide Efficiently
Implementing this one-pot synthesis requires precise control over stoichiometry and thermal parameters to maximize the dual yield of both DM and TPP. The process begins with the activation of TPPO using BTC in a suitable organic solvent, followed by the controlled addition of the mercaptan-base mixture. The patent outlines specific molar ratios, suggesting a TPPO to BTC ratio of approximately 1.0:0.3 to 0.6, and a TPPO to Mercaptan ratio of 1.0:1.5 to 3.0, to ensure complete conversion without excessive reagent waste. Temperature management is equally critical, with optimal results observed in the range of 20°C to 50°C, balancing reaction kinetics with safety profiles. The detailed standardized synthesis steps below provide a comprehensive guide for replicating this high-efficiency protocol in a pilot or production setting, ensuring consistent quality and yield.
- Activate Triphenylphosphine Oxide (TPPO) by reacting with Bis(trichloromethyl) carbonate (BTC) in an organic solvent at temperatures between -30°C and 90°C to form the dichloro intermediate.
- Introduce 2-Mercaptobenzothiazole (M) dissolved in organic solvent along with an organic base to the reaction mixture, maintaining temperature control for oxidative coupling.
- Upon completion, perform suction filtration to isolate Dibenzothiazole Disulfide, then recover solvent from the filtrate and recrystallize the residue to obtain high-purity Triphenylphosphine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented technology offers profound strategic benefits that extend beyond simple chemical synthesis. By transforming waste byproducts into high-value commodities, manufacturers can fundamentally alter their cost structures, turning a liability (waste disposal) into a revenue stream. The elimination of expensive and hazardous reducing agents like silanes or aluminum hydrides directly translates to lower raw material expenditures and reduced insurance premiums associated with handling dangerous goods. Furthermore, the simplified one-pot nature of the reaction reduces the number of required processing units, thereby lowering capital investment and maintenance costs for production facilities. This efficiency gain allows for reducing lead time for high-purity pharmaceutical intermediates, enabling faster response to market demands and more agile inventory management. The ability to source precursors from internal waste streams also insulates the supply chain from external volatility in raw material pricing, ensuring greater continuity and reliability in production schedules.
- Cost Reduction in Manufacturing: The primary economic driver of this technology is the substitution of costly stoichiometric reducing agents with the more economical Bis(trichloromethyl) carbonate, coupled with the valorization of waste TPPO. By avoiding the purchase of dedicated reducing agents and minimizing waste disposal fees, the overall cost of goods sold (COGS) is significantly compressed. Additionally, the high atom economy means that a larger proportion of the input mass is converted into saleable product, maximizing the return on investment for every kilogram of raw material purchased. The process also reduces solvent consumption through the one-pot design, further lowering utility and procurement costs associated with solvent recovery and make-up.
- Enhanced Supply Chain Reliability: Integrating this recycling loop into the production of cephalosporin active esters creates a closed-loop system that reduces dependency on external suppliers for Triphenylphosphine and Dibenzothiazole Disulfide. This self-sufficiency mitigates the risk of supply disruptions caused by geopolitical issues or market shortages of key reagents. Since the feedstocks (TPPO and Mercaptobenzothiazole) are generated in situ from the main production line, the availability of these intermediates is intrinsically linked to the plant's own output, providing a predictable and stable supply baseline. This stability is crucial for long-term contract negotiations with downstream pharmaceutical clients who require guaranteed delivery schedules.
- Scalability and Environmental Compliance: The reaction conditions described in the patent, operating at mild temperatures and atmospheric pressure, are inherently scalable from laboratory benchtops to multi-ton industrial reactors without requiring specialized high-pressure equipment. The use of common organic solvents like toluene and chlorobenzene facilitates easy integration into existing infrastructure. From an environmental perspective, the process drastically reduces the E-factor (mass of waste per mass of product) by eliminating heavy metal catalysts and toxic reducing agents. This alignment with Green Chemistry principles simplifies regulatory compliance and permitting processes, accelerating the time-to-market for new production lines and enhancing the corporate sustainability profile.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this one-pot synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for evaluating the feasibility of this route for your specific manufacturing needs. Understanding these nuances is essential for R&D teams planning pilot runs and for procurement officers assessing the total cost of ownership.
Q: What are the primary advantages of this one-pot method over traditional TPPO reduction?
A: Unlike traditional methods requiring expensive and hazardous reducing agents like silanes or aluminum hydrides, this patent utilizes Bis(trichloromethyl) carbonate (BTC) under mild conditions, significantly lowering operational hazards and raw material costs while achieving high atom economy.
Q: How does this process address environmental concerns in cephalosporin production?
A: The process effectively recycles Triphenylphosphine Oxide (TPPO) and 2-Mercaptobenzothiazole, which are major waste byproducts in cephalosporin active ester synthesis, transforming them into valuable commodities and eliminating the need for difficult waste disposal.
Q: What purity levels can be expected for the final Triphenylphosphine product?
A: According to the experimental data in the patent, the recrystallized Triphenylphosphine achieves HPLC purity levels exceeding 98.5%, with yields consistently above 80% across various solvent systems, ensuring suitability for sensitive pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dibenzothiazole Disulfide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of waste-to-value technologies like the one described in CN101357908B for optimizing pharmaceutical supply chains. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory concepts are successfully translated into robust industrial realities. Our facility is equipped with rigorous QC labs and stringent purity specifications that guarantee every batch of Dibenzothiazole Disulfide and Triphenylphosphine meets the exacting standards required for API synthesis. We are committed to delivering not just chemicals, but comprehensive solutions that enhance your operational efficiency and sustainability goals.
We invite you to engage with our technical team to explore how this recycling methodology can be adapted to your specific production context. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this greener route. Our experts are ready to provide specific COA data and route feasibility assessments tailored to your volume requirements. Contact our technical procurement team today to discuss how we can support your journey towards more sustainable and cost-effective pharmaceutical manufacturing.
