Advanced Synthesis of 2,3-Difluoro-4-Methoxyphenylacetic Acid for Commercial Scale-up
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and environmentally benign pathways for constructing complex fluorinated building blocks. Patent CN114380682A introduces a groundbreaking synthetic methodology for 2,3-difluoro-4-methoxyphenylacetic acid, a critical intermediate utilized in the development of anticancer agents targeting hyperproliferative diseases. This innovation addresses a significant gap in the prior art, where the absence of a reliable synthesis route has hindered the broader application of this valuable molecular scaffold. By leveraging a strategic functional group conversion from vinyl to ethanol and finally to acetate, the disclosed method achieves an ideal total yield while maintaining operational simplicity. For R&D directors and procurement specialists, this represents a pivotal shift towards safer, more efficient manufacturing protocols that align with modern green chemistry principles and stringent regulatory standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of similar fluoro-phenylacetic acid derivatives has relied heavily on hazardous reagents that pose substantial risks to both personnel and the environment. Traditional pathways often employ sodium cyanide for cyanation followed by hydrolysis, a process notorious for its extreme toxicity and the potential for fatal accidents even upon trace exposure. Alternatively, methods utilizing diphenyl diselenide for acetate group conversion, while yielding acceptable results, introduce heavy metal contaminants and toxic byproducts that complicate waste management and increase disposal costs. These conventional approaches frequently require harsh reaction conditions, such as cryogenic temperatures around -78°C, which demand significant energy consumption and specialized equipment. Furthermore, the formation of difficult-to-remove byproducts, such as ring-closing impurities, often necessitates complex purification steps that erode overall process efficiency and economic viability.
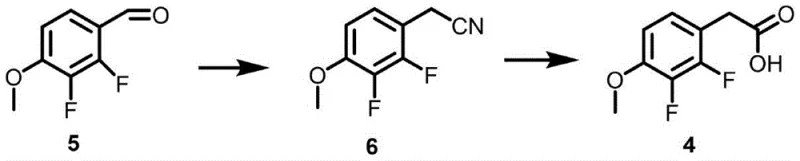
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes 4-bromo-2,3-difluoroanisole, a readily available starting material, to construct the target molecule through a sequence of olefination, hydroboration, and oxidation. This strategy completely circumvents the use of highly toxic cyanides and selenium-based reagents, replacing them with safer vinyl potassium trifluoroborate and borane complexes. The reaction conditions are remarkably mild, operating between 0°C and 75°C, which drastically reduces energy requirements and eliminates the need for expensive cryogenic infrastructure. The process demonstrates high stability and selectivity, avoiding the rearrangement phenomena common in other alkylation strategies. By streamlining the synthetic sequence and utilizing reagents that are easy to handle and source, this method offers a compelling solution for cost reduction in pharmaceutical intermediate manufacturing while ensuring a safer working environment for production teams.
Mechanistic Insights into Pd-Catalyzed Olefination and Oxidation
The core of this synthetic breakthrough lies in the precise execution of a palladium-catalyzed cross-coupling reaction followed by a regioselective hydroboration-oxidation sequence. In the initial step, 4-bromo-2,3-difluoroanisole undergoes olefination with vinyl potassium trifluoroborate in the presence of a palladium catalyst such as Pd(dppf)Cl2·CH2Cl2. This transformation is conducted in a mixed solvent system of tetrahydrofuran and water at 75°C, facilitating the efficient introduction of the vinyl group onto the aromatic ring. The subsequent hydroboration step employs borane-tetrahydrofuran complex at 0°C, followed by oxidative workup with alkaline hydrogen peroxide. This sequence is critical as it follows the anti-Markovnikov rule, ensuring the formation of the primary alcohol intermediate without skeletal rearrangement. The high selectivity of this step minimizes the generation of structural isomers, thereby simplifying downstream purification and enhancing the overall purity profile of the intermediate.
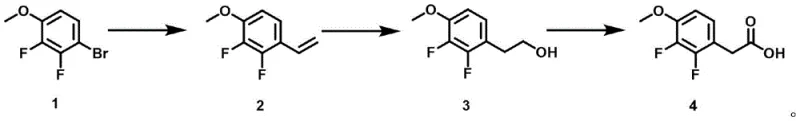
Final conversion to the carboxylic acid is achieved through a controlled oxidation process using chromium trioxide and periodic acid. This oxidation step is performed at low temperatures between 0°C and 5°C, which prevents over-oxidation or degradation of the sensitive fluoro-methoxy substituents on the aromatic ring. The use of periodic acid as a co-oxidant enhances the efficiency of chromium trioxide, allowing for high conversion rates under mild conditions. From an impurity control perspective, the avoidance of high-temperature conditions and toxic reagents significantly reduces the formation of thermal degradation products and heavy metal residues. The resulting crude product can be efficiently purified via column chromatography to achieve purity levels exceeding 95%, meeting the rigorous specifications required for active pharmaceutical ingredient (API) synthesis. This mechanistic robustness ensures batch-to-batch consistency, a key factor for supply chain reliability.
How to Synthesize 2,3-Difluoro-4-Methoxyphenylacetic Acid Efficiently
Implementing this synthesis route requires careful attention to reagent stoichiometry and temperature control to maximize yield and safety. The process begins with the dissolution of the bromo-anisole starting material in a THF-water mixture, followed by the addition of base and vinyl boron salt under inert atmosphere. The reaction is heated to reflux conditions to drive the coupling to completion before workup. Subsequent steps involve precise dropwise addition of borane and oxidants to manage exotherms and ensure selectivity. The standardized synthesis steps outlined below provide a clear roadmap for laboratory and pilot-scale execution, ensuring that technical teams can replicate the high yields reported in the patent data.
- Perform Pd-catalyzed olefination of 4-bromo-2,3-difluoroanisole with vinyl potassium trifluoroborate at 75°C to form the vinyl intermediate.
- Execute hydroboration-oxidation using borane-THF and alkaline hydrogen peroxide to convert the vinyl group into a primary alcohol.
- Oxidize the primary alcohol to the target carboxylic acid using chromium trioxide and periodic acid under mild temperature conditions.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers profound strategic advantages beyond mere technical feasibility. The elimination of highly toxic reagents like sodium cyanide and diphenyl diselenide translates directly into reduced regulatory burden and lower costs associated with hazardous waste disposal and safety compliance. By utilizing commercially available and stable starting materials, the risk of supply disruption is minimized, ensuring a continuous flow of raw materials for production schedules. The mild reaction conditions reduce energy consumption significantly, contributing to lower operational expenditures and a smaller carbon footprint. Furthermore, the simplified purification process reduces solvent usage and processing time, enhancing overall throughput capacity without compromising quality standards.
- Cost Reduction in Manufacturing: The removal of expensive and hazardous reagents such as diphenyl diselenide eliminates the need for specialized heavy metal removal processes, which are often costly and time-consuming. The use of common solvents and catalysts allows for bulk purchasing advantages, while the high selectivity of the reaction reduces material loss due to byproduct formation. Additionally, the avoidance of cryogenic conditions removes the capital and operational expenses associated with maintaining ultra-low temperature reactors. These factors collectively drive down the cost of goods sold, making the final intermediate more competitive in the global market.
- Enhanced Supply Chain Reliability: Sourcing 4-bromo-2,3-difluoroanisole and vinyl potassium trifluoroborate is straightforward due to their widespread availability in the fine chemical market. Unlike specialized toxic reagents that may face shipping restrictions or supply bottlenecks, these materials can be procured from multiple qualified vendors. The robustness of the reaction against moisture and air in certain steps also reduces the risk of batch failure due to environmental factors. This reliability ensures that production timelines are met consistently, reducing lead time for high-purity pharmaceutical intermediates and strengthening partnerships with downstream API manufacturers.
- Scalability and Environmental Compliance: The process is designed with industrial scale-up in mind, featuring simple unit operations such as extraction and crystallization that are easily transferred from laboratory to plant scale. The absence of highly toxic gases or unstable intermediates simplifies the engineering controls required for large-scale production. Waste streams are less hazardous, facilitating easier treatment and compliance with increasingly strict environmental regulations. This scalability ensures that the supply can grow in tandem with market demand, supporting the commercial expansion of complex drug candidates that rely on this key building block.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of 2,3-difluoro-4-methoxyphenylacetic acid. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation. Understanding these aspects helps stakeholders make informed decisions regarding process adoption and integration into existing manufacturing workflows.
Q: Why is this synthesis method safer than conventional cyanation routes?
A: Conventional methods often rely on highly toxic sodium cyanide or diphenyl diselenide, posing severe health and environmental risks. This patented route eliminates these hazardous reagents, utilizing low-toxicity vinyl boron compounds and mild oxidants instead.
Q: What are the key advantages for industrial scale-up?
A: The process operates under mild temperatures (0-75°C) without strict dehydration requirements. The use of commercially available raw materials and robust reaction conditions ensures high stability and easier purification, facilitating seamless transition from lab to plant.
Q: How does this route impact impurity profiles?
A: The hydroboration step follows anti-Markovnikov addition with high selectivity, minimizing rearrangement byproducts. Combined with efficient column chromatography purification, this results in a final product with superior purity suitable for sensitive pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3-Difluoro-4-Methoxyphenylacetic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of life-saving medications. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 2,3-difluoro-4-methoxyphenylacetic acid meets the highest industry standards. We are committed to delivering not just a product, but a comprehensive solution that supports your R&D and commercial goals.
We invite you to collaborate with our technical procurement team to explore how this innovative synthesis route can benefit your specific projects. Contact us today to request a Customized Cost-Saving Analysis tailored to your volume requirements. Our experts are ready to provide specific COA data and route feasibility assessments to help you optimize your supply chain and accelerate your time to market.
