Advanced One-Step Grignard Route for Ezetimibe Key Intermediate Manufacturing
The pharmaceutical industry continuously seeks more efficient pathways for synthesizing critical cardiovascular therapeutics, and the production of Ezetimibe, a potent cholesterol absorption inhibitor, is no exception. Patent CN102531985A discloses a groundbreaking methodology for preparing the key Ezetimibe intermediate, specifically the (3R,4S)-1-(4-fluorophenyl)-3-[3-(4-fluorophenyl)-3-oxopropyl]-4-substituted phenoxy-2-azetidinone structure. This innovation represents a paradigm shift from traditional multi-step sequences involving precious metal catalysis to a streamlined, one-step direct Grignard addition. For R&D directors and procurement strategists, this development signals a substantial opportunity to optimize supply chains by reducing dependency on volatile palladium markets and complex organometallic reagents. The technical breakthrough lies in the ability to directly convert the ester precursor into the desired ketone functionality without over-reaction or degradation of the sensitive beta-lactam ring, achieving yields that support robust industrial viability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this critical azetidinone intermediate has been plagued by inefficiencies inherent in multi-step linear sequences. As illustrated in prior art such as U.S. Pat 5767115, the conventional route necessitates the hydrolysis of the starting ester to a carboxylic acid, followed by activation to an acyl chloride, and finally a cross-coupling reaction. This final coupling step typically relies on a Negishi reaction, requiring the in-situ preparation of a 4-fluorophenyl zinc reagent and the use of a tetrakis(triphenylphosphine)palladium(0) catalyst. Such a pathway introduces significant logistical burdens, including the handling of moisture-sensitive organozinc species and the stringent requirement for removing trace palladium residues to meet pharmaceutical purity standards. Furthermore, the cumulative yield loss across three distinct isolation and purification stages significantly inflates the cost of goods sold (COGS) and extends the overall production lead time.
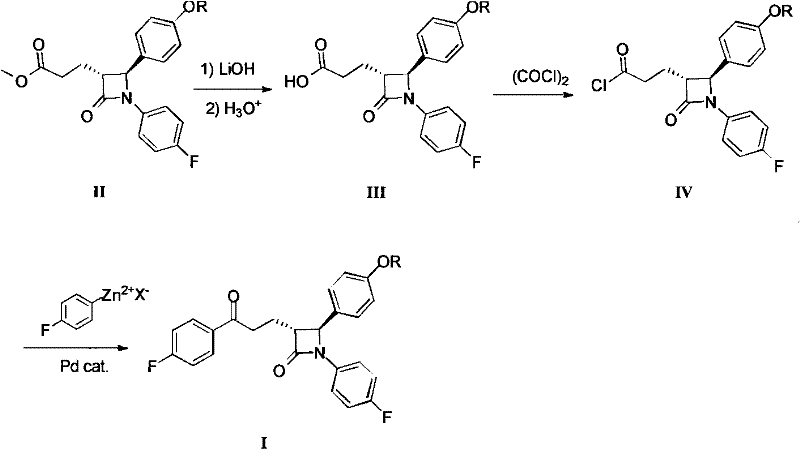
The Novel Approach
In stark contrast, the novel methodology described in the patent data simplifies this complex transformation into a single, elegant operation. By reacting the ester intermediate (II) directly with a 4-fluorophenyl Grignard reagent under carefully controlled alkaline conditions, the process bypasses the need for acid activation and transition metal catalysis entirely. This direct addition strategy not only shortens the synthetic route by three operational steps but also eliminates the capital expenditure associated with recovering expensive palladium catalysts. The reaction proceeds with high chemoselectivity, stopping at the ketone stage rather than proceeding to the tertiary alcohol, which is a common side reaction in Grignard additions to esters. This efficiency translates directly into a more agile manufacturing process, allowing for faster batch turnover and reduced solvent inventory requirements.
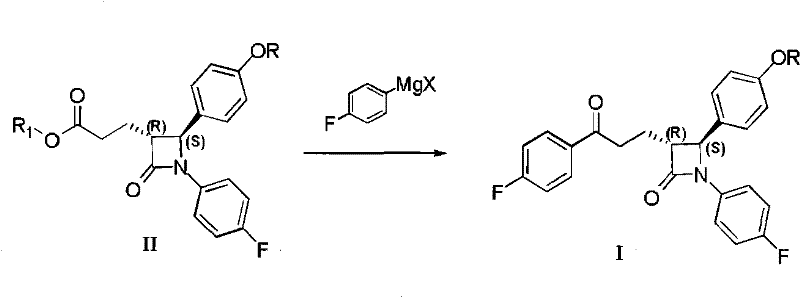
Mechanistic Insights into Chemoselective Grignard Addition
The core scientific achievement of this patent lies in the modulation of Grignard reactivity to achieve ketone selectivity in the presence of an ester functionality. Typically, Grignard reagents are too nucleophilic to stop at the ketone stage when reacting with esters, often leading to double addition and the formation of tertiary alcohols. However, the inventors have discovered that the presence of a stoichiometric amount of organic base, such as triethylamine or N,N-diisopropylethylamine, plays a critical role in tempering the nucleophilicity of the Grignard species. It is hypothesized that the base may coordinate with the magnesium center or interact with the carbonyl oxygen of the intermediate ketone, effectively deactivating it towards further nucleophilic attack while leaving the initial ester susceptible to reaction. This delicate balance allows for the preservation of the sensitive four-membered beta-lactam ring, which is prone to ring-opening under harsh nucleophilic or acidic conditions.
Furthermore, the control of reaction temperature and addition order is paramount to the success of this mechanism. The patent specifies that the reaction should be conducted at low temperatures, preferably between -30°C and 0°C, to kinetically favor the formation of the ketone and minimize thermal degradation. The strategy of adding the ester substrate slowly to a pre-mixed solution of the Grignard reagent and base ensures that the concentration of the electrophile remains low relative to the nucleophile, preventing localized hot spots that could lead to over-addition. This precise control over reaction parameters ensures that the impurity profile remains clean, with minimal formation of the corresponding alcohol byproduct, thereby simplifying downstream purification and enhancing the overall quality of the pharmaceutical intermediate.
How to Synthesize Ezetimibe Key Intermediate Efficiently
The implementation of this novel synthetic route requires strict adherence to anhydrous conditions and precise dosing protocols to maximize yield and purity. The process begins with the preparation of a dry reaction vessel under an inert atmosphere, typically nitrogen or argon, to prevent the quenching of the Grignard reagent by atmospheric moisture. A solution of the organic base and the 4-fluorophenyl magnesium halide is prepared in an aprotic solvent such as toluene or tetrahydrofuran (THF) and cooled to the specified sub-zero temperature range. The ester starting material, dissolved in a compatible solvent, is then added dropwise to this mixture over a controlled period, maintaining the internal temperature within the optimal window to ensure chemoselectivity. Following the completion of the addition, the reaction mixture is stirred for a defined period to allow for full conversion before being quenched with a mild acid solution.
- Prepare a reaction system with anhydrous aprotic solvent (e.g., toluene/THF) and add organic base (triethylamine) and Grignard reagent at low temperature (-30°C to 0°C).
- Slowly add the solution of ester intermediate II to the reaction mixture in batches to control exotherm and chemoselectivity.
- Quench the reaction with acid, extract with organic solvent, and purify via crystallization or chromatography to obtain the high-purity ketone intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this technology offers compelling economic and operational advantages that extend beyond simple yield improvements. The most significant impact is the drastic reduction in raw material costs associated with the elimination of the palladium catalyst system. Palladium complexes and their specialized phosphine ligands represent a high-cost input with significant price volatility; removing this dependency stabilizes the cost structure of the intermediate. Additionally, the removal of the organozinc preparation step eliminates the need for activated zinc metal and the associated safety protocols for handling pyrophoric or highly reactive organometallic species, thereby lowering operational overhead and insurance costs related to hazardous material handling.
- Cost Reduction in Manufacturing: The consolidation of three discrete chemical transformations into a single reactor charge significantly lowers the variable costs of production. By removing the hydrolysis and acylation steps, the process saves on the consumption of reagents such as thionyl chloride or oxalyl chloride, as well as the solvents required for multiple workups and extractions. The absence of a heavy metal catalyst also removes the expensive and time-consuming step of scavenging trace metals to meet regulatory limits, which often requires specialized resin columns or additional crystallization cycles. Consequently, the overall cost per kilogram of the active intermediate is substantially reduced, improving margin potential for the final drug product.
- Enhanced Supply Chain Reliability: Simplifying the synthetic route inherently reduces the number of potential failure points in the manufacturing supply chain. Long, multi-step syntheses are vulnerable to yield fluctuations at any given stage, which can cascade into significant delays in final delivery. By shortening the route, the lead time from raw material intake to finished intermediate is compressed, allowing for more responsive inventory management. Furthermore, the reliance on commodity chemicals like Grignard reagents and triethylamine, rather than custom-synthesized organozinc species or specialized catalysts, ensures a more robust and diversified supplier base, mitigating the risk of single-source bottlenecks.
- Scalability and Environmental Compliance: From an environmental and safety perspective, this process aligns well with green chemistry principles by reducing the E-factor (mass of waste per mass of product). The elimination of palladium waste streams simplifies effluent treatment and reduces the burden on wastewater processing facilities, which is increasingly critical as environmental regulations tighten globally. The process is also highly amenable to scale-up; the exothermic nature of the Grignard addition can be easily managed in large-scale reactors through controlled dosing and cooling, ensuring consistent quality from pilot plant to commercial tonnage production without the need for complex equipment modifications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel Grignard-based synthesis for Ezetimibe intermediates. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, offering clarity on yield expectations, impurity profiles, and scalability factors. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer and for commercial teams assessing the long-term value proposition of this manufacturing route.
Q: How does this new method improve upon the traditional Palladium-catalyzed route?
A: The traditional route requires three distinct steps: hydrolysis, acylation, and a Palladium-catalyzed Negishi coupling. The new method condenses this into a single direct Grignard addition step, eliminating the need for expensive Palladium catalysts and Zinc reagents, thereby drastically simplifying the workflow and reducing heavy metal contamination risks.
Q: What represents the primary cost-saving mechanism in this synthesis?
A: The primary cost savings arise from the elimination of transition metal catalysts (Palladium) and the associated ligand systems, which are costly and require rigorous removal processes. Additionally, shortening the synthetic sequence from three steps to one significantly reduces solvent consumption, labor hours, and overall processing time.
Q: Is this process scalable for industrial production of cardiovascular drug intermediates?
A: Yes, the process is highly scalable. It utilizes standard Grignard chemistry which is well-understood in industrial settings. The avoidance of sensitive organozinc reagents and the use of robust organic bases like triethylamine make the process safer and easier to manage on a large scale compared to the previous multi-step protocols.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ezetimibe Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to more efficient synthetic routes is critical for maintaining competitiveness in the global cardiovascular drug market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this novel Grignard methodology are fully realized in a GMP-compliant environment. We maintain stringent purity specifications and operate rigorous QC labs equipped to detect trace impurities, guaranteeing that every batch of Ezetimibe intermediate meets the exacting standards required for downstream API synthesis. Our commitment to continuous process improvement allows us to offer clients a supply partner that is not just a vendor, but a strategic ally in cost optimization.
We invite pharmaceutical companies and contract manufacturers to engage with our technical procurement team to discuss how this advanced synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the specific economic benefits tailored to your volume requirements. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with the highest level of technical confidence and commercial security.
