Revolutionizing 2,3,4,4'-Tetrahydroxybenzophenone Production with Magnetic Ionic Liquids
Introduction to Advanced Green Synthesis Technologies
The landscape of fine chemical manufacturing is undergoing a paradigm shift towards sustainability and efficiency, driven by the urgent need to reduce environmental footprints while maintaining high product quality. A pivotal development in this domain is disclosed in patent CN109824491B, which introduces a novel production method for 2,3,4,4'-tetrahydroxybenzophenone, a critical intermediate widely utilized in the synthesis of pharmaceuticals, ultraviolet absorbers, and stabilizers. This groundbreaking technology leverages a functionalized magnetic silica gel supported bisimidazole ionic liquid as a heterogeneous catalyst, fundamentally altering the traditional Friedel-Crafts acylation landscape. By replacing hazardous organic solvents with water and enabling magnetic separation of the catalyst, this innovation addresses long-standing challenges regarding toxicity, waste generation, and operational complexity. For industry leaders seeking a reliable pharmaceutical intermediate supplier, understanding the mechanistic and commercial implications of this patent is essential for future-proofing supply chains and achieving superior cost structures in competitive markets.
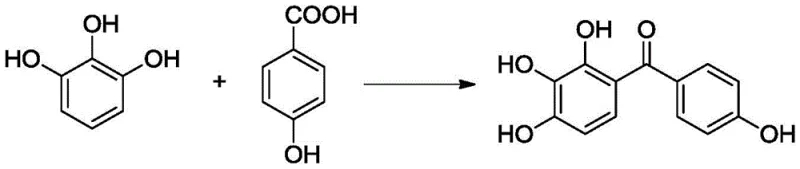
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 2,3,4,4'-tetrahydroxybenzophenone has relied heavily on traditional Friedel-Crafts acylation protocols employing Lewis acid catalysts such as zinc chloride, boron trifluoride, or acidic resins. These legacy methods are fraught with significant technical and environmental drawbacks that hinder modern manufacturing efficiency. For instance, the zinc chloride method often necessitates the addition of phosphorus reagents like phosphorus trichloride, which introduces severe toxicity hazards and complicates downstream purification due to the formation of intractable phosphorous-containing byproducts. Furthermore, boron trifluoride-based processes, while effective, pose serious safety risks as the catalyst can decompose upon contact with moisture to release highly toxic smog, requiring specialized containment infrastructure and rigorous safety protocols. Additionally, these homogeneous or semi-homogeneous systems typically demand high reaction temperatures and extended reaction times, leading to excessive energy consumption and potential thermal degradation of the sensitive polyphenolic substrates. The inability to easily recover and recycle these catalysts results in substantial chemical waste, driving up disposal costs and creating a heavy burden on environmental compliance teams.
The Novel Approach
In stark contrast to these archaic methodologies, the novel approach detailed in the patent utilizes a sophisticated functionalized magnetic silica gel supported bisimidazole ionic liquid catalyst that operates efficiently in an aqueous medium. This system represents a leap forward in green chemistry by eliminating the need for volatile organic compounds (VOCs) and toxic Lewis acids, thereby creating a inherently safer working environment. The unique architecture of the catalyst, which combines the high catalytic activity of imidazolium ionic liquids with the physical robustness of a magnetic silica support, allows for precise control over reaction selectivity and kinetics. Crucially, the magnetic properties of the support enable rapid and complete separation of the catalyst from the reaction mixture simply by applying an external magnetic field, bypassing the need for complex filtration or centrifugation steps. This heterogeneity not only simplifies the work-up procedure but also facilitates the direct reuse of the catalyst for subsequent batches, dramatically reducing the consumption of fresh catalytic material. The result is a streamlined process that delivers high yields and exceptional purity while adhering to the strictest environmental standards, making it an ideal solution for cost reduction in fine chemical manufacturing.
Mechanistic Insights into Magnetic Ionic Liquid Catalysis
The efficacy of this advanced synthesis route lies in the intricate design of the catalyst, which functions as a dual-active site system promoting the acylation of pyrogallic acid with p-hydroxybenzoic acid. The bisimidazole ionic liquid moieties grafted onto the silica surface act as strong Brønsted or Lewis acid sites, activating the carboxylic acid group of the p-hydroxybenzoic acid towards nucleophilic attack by the electron-rich aromatic ring of the pyrogallic acid. The ionic liquid environment stabilizes the transition state and enhances the solubility of the organic reactants within the aqueous phase, effectively creating a micro-environment that accelerates the reaction rate without the need for harsh conditions. Moreover, the silica gel support provides a high surface area that maximizes the exposure of active sites, ensuring efficient mass transfer and minimizing diffusion limitations that often plague heterogeneous catalysis. The presence of the magnetic Fe3O4 core within the silica matrix is the key enabler for the operational simplicity, allowing the entire catalytic bed to be manipulated externally. This structural integrity ensures that the catalyst remains stable under the reaction conditions of 40-100°C, preventing leaching of active species into the product stream and guaranteeing the high purity of the final 2,3,4,4'-tetrahydroxybenzophenone.
Impurity control is another critical aspect where this mechanistic design excels, particularly relevant for R&D directors focused on product specification. Traditional acid catalysts often promote side reactions such as over-acylation or polymerization of the phenolic substrates due to their uncontrolled acidity and lack of selectivity. However, the tunable nature of the ionic liquid anions (such as HSO4-, CF3SO3-, or BF4-) allows for the fine-tuning of acid strength to match the specific reactivity of the substrates, thereby suppressing unwanted side pathways. The aqueous solvent system further aids in impurity management by dissolving polar byproducts and inorganic salts while precipitating the less polar target benzophenone derivative, facilitating a clean crystallization process. The magnetic separation step acts as a final barrier against catalyst contamination, ensuring that no metal residues or support fragments remain in the crude product. This multi-layered approach to purity assurance means that the downstream purification burden is significantly reduced, often requiring only a single recrystallization step to achieve pharmaceutical-grade specifications exceeding 99.7% purity.
How to Synthesize 2,3,4,4'-Tetrahydroxybenzophenone Efficiently
Implementing this cutting-edge synthesis protocol requires a clear understanding of the operational parameters that govern reaction efficiency and product quality. The process begins with the precise stoichiometric mixing of pyrogallic acid and p-hydroxybenzoic acid in water, followed by the introduction of the magnetic catalyst at a loading of 1-30% relative to the substrate mass. The reaction mixture is then heated to a controlled temperature range of 40-100°C, with optimal results typically observed between 90-100°C, and maintained under stirring for a duration of 1 to 8 hours depending on the specific catalyst variant employed. Upon completion, the reaction vessel is cooled, and a magnet is applied to the exterior to sequester the catalyst, allowing the supernatant containing the crude product to be decanted or filtered effortlessly. The detailed standardized synthesis steps, including specific molar ratios, temperature profiles, and work-up procedures validated by experimental data, are outlined below for technical reference.
- Mix pyrogallic acid, p-hydroxybenzoic acid, water, and the magnetic catalyst in a reaction vessel.
- Heat the mixture to 40-100°C and stir for 1-8 hours to facilitate the Friedel-Crafts acylation.
- Separate the catalyst using an external magnet, then wash and recrystallize the product with ethanol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this magnetic catalytic technology translates into tangible strategic advantages that extend far beyond simple yield improvements. The shift from hazardous organic solvents and corrosive Lewis acids to a benign aqueous system fundamentally alters the cost structure of production by eliminating the expenses associated with solvent recovery, storage, and hazardous waste disposal. The ability to recover and recycle the catalyst magnetically removes the recurring cost of purchasing fresh catalyst for every batch, turning a consumable expense into a fixed capital asset that depreciates over a much longer lifecycle. Furthermore, the simplified work-up procedure reduces the man-hours required for filtration and purification, directly lowering labor costs and increasing the overall throughput of the manufacturing facility. These factors combine to create a robust economic model that offers substantial cost savings and improved margin potential for high-volume production runs.
- Cost Reduction in Manufacturing: The elimination of expensive and toxic organic solvents alongside the removal of phosphorus or boron-based reagents drastically reduces the raw material bill of materials. Since the catalyst can be reused multiple times without significant loss of activity, the effective cost per kilogram of catalyst consumed approaches zero over time, leading to a dramatic decrease in variable production costs. Additionally, the reduced need for complex waste treatment infrastructure lowers the overhead associated with environmental compliance and effluent management.
- Enhanced Supply Chain Reliability: Relying on water as the primary solvent mitigates the risks associated with the supply volatility of petrochemical-derived solvents, ensuring a more stable and predictable production schedule. The non-toxic nature of the reagents simplifies logistics and storage requirements, reducing the regulatory burden and insurance premiums associated with handling hazardous chemicals. This stability ensures consistent delivery timelines for high-purity pharmaceutical intermediates, safeguarding downstream customers from supply disruptions.
- Scalability and Environmental Compliance: The heterogeneous nature of the reaction makes it inherently easier to scale from laboratory benchtop to multi-ton industrial reactors without the heat transfer and mixing limitations often seen in viscous homogeneous systems. The green profile of the process aligns perfectly with increasingly stringent global environmental regulations, future-proofing the manufacturing site against potential bans on traditional Friedel-Crafts reagents. This scalability ensures that the commercial scale-up of complex pharmaceutical intermediates can be achieved rapidly and safely to meet growing market demand.
Frequently Asked Questions (FAQ)
To further clarify the technical and commercial viability of this production method, we have compiled a set of frequently asked questions based on the specific data points and claims found within the patent literature. These answers address common concerns regarding catalyst longevity, product purity, and process safety, providing a transparent view of the technology's capabilities. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the advantages of using magnetic ionic liquid catalysts over traditional Lewis acids?
A: Unlike traditional catalysts like ZnCl2 or BF3 which generate toxic waste and are difficult to separate, magnetic ionic liquids allow for simple magnetic separation, recyclability, and operate in green aqueous solvents.
Q: What is the typical purity achievable with this novel synthesis method?
A: The patented process demonstrates the capability to achieve product purity levels exceeding 99.7% after standard recrystallization, meeting stringent requirements for pharmaceutical intermediates.
Q: Is the catalyst reusable in this production method?
A: Yes, the functionalized magnetic silica gel supported catalyst can be recovered via magnetic adsorption and reused for multiple cycles without significant loss of catalytic activity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3,4,4'-Tetrahydroxybenzophenone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the magnetic ionic liquid catalysis technology disclosed in patent CN109824491B and are fully equipped to leverage these advancements for our global clientele. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this green chemistry approach are realized in practical, large-scale operations. Our state-of-the-art facilities are designed to handle sensitive catalytic processes with precision, utilizing rigorous QC labs to verify that every batch meets stringent purity specifications required by the pharmaceutical and specialty chemical industries. We are committed to delivering high-purity UV absorber intermediates that not only meet but exceed the quality expectations of discerning international partners.
We invite you to collaborate with us to explore how this innovative synthesis route can optimize your supply chain and reduce your overall manufacturing costs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating the economic impact of switching to this greener methodology. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and sample availability, and let us demonstrate why we are the preferred partner for sustainable and efficient chemical manufacturing solutions.
