Advanced Synthesis of DiFmoc-Lys(plus) for Commercial MAPS Polypeptide Manufacturing
Advanced Synthesis of DiFmoc-Lys(plus) for Commercial MAPS Polypeptide Manufacturing
The landscape of vaccine development and immunological research is constantly evolving, with a significant shift towards synthetic peptide vaccines due to their high safety profiles and design flexibility. However, the inherent limitation of small molecular weight synthetic peptides is their weak immunogenicity, which often necessitates conjugation to carrier proteins. Patent CN111233714A introduces a groundbreaking solution to this challenge through the development of a novel unnatural amino acid, DiFmoc-Lys(plus). This technology enables the construction of Multiple Antigen Peptide Systems (MAPS) with unprecedented structural consistency. By addressing the critical bottleneck of uneven coupling efficiency found in traditional lysine-based cores, this innovation offers a reliable pathway for producing high-purity pharmaceutical intermediates. For R&D directors and procurement specialists alike, understanding the mechanistic advantages of this symmetric linker is crucial for optimizing supply chains and reducing the total cost of ownership in complex peptide manufacturing.
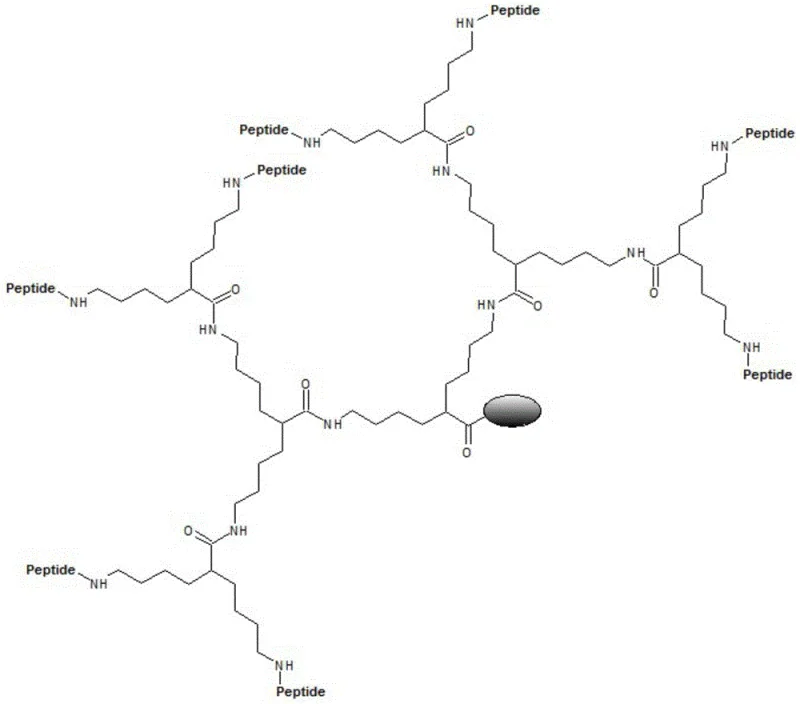
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
In the traditional synthesis of branched multiple antigen peptides, the industry has long relied on the natural amino acid Lysine (Lys) as a core scaffold. While Lysine possesses two amino groups capable of forming peptide bonds, these groups are chemically distinct: the alpha-amino group and the epsilon-amino group are separated by different carbon chain lengths. This structural asymmetry creates a significant technical hurdle during solid-phase synthesis. When antigen peptides are coupled to these divergent arms, the steric hindrance and reaction kinetics differ substantially between the two sites. The shorter alpha-arm often suffers from lower coupling efficiency due to crowding near the carboxyl backbone, while the longer epsilon-arm reacts more freely. This disparity leads to the formation of deletion peptides and incomplete sequences, resulting in a heterogeneous crude product mixture that is notoriously difficult to purify. Furthermore, previous attempts to mitigate this by grafting beta-alanine onto the alpha-amino group, creating Di-Fmoc-Lys(beta-Ala), introduced amide bonds that improved length consistency but failed to achieve perfect reaction uniformity due to the electronic differences introduced by the amide linkage.
The Novel Approach
The invention disclosed in CN111233714A overcomes these historical limitations by introducing DiFmoc-Lys(plus), a meticulously engineered unnatural amino acid designed for perfect symmetry. Unlike natural lysine, DiFmoc-Lys(plus) features two bifurcated arms that are completely identical in both length and chemical structure. Both arms consist of a four-carbon butylamine chain protected by Fmoc groups, radiating from the central alpha-carbon. This geometric symmetry ensures that the subsequent amino acid coupling reactions proceed with identical efficiency on both branches. By eliminating the kinetic disparity between the two growth points, the synthesis of the branched polypeptide becomes highly predictable and uniform. The result is a dramatic improvement in the purity of the crude MAPS polypeptide and a significantly higher success rate in obtaining the target macromolecular structure. This structural innovation transforms the MAPS core from a source of variability into a robust platform for consistent manufacturing.
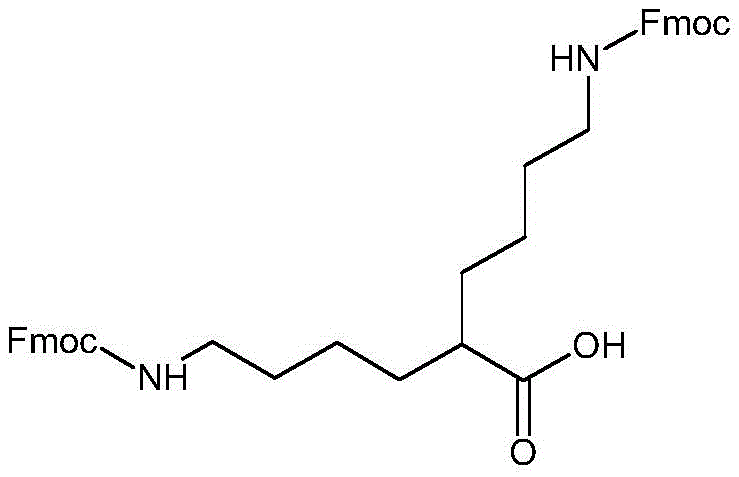
Mechanistic Insights into Grignard-Mediated Symmetric Core Construction
The synthesis of DiFmoc-Lys(plus) represents a sophisticated application of organometallic chemistry tailored for peptide intermediate production. The core strategy involves the construction of the symmetric side chain via a Grignard coupling reaction, a method chosen for its reliability and scalability in fine chemical manufacturing. The process begins with the conversion of Fmoc-butanediamine into a reactive electrophile. Through a diazotization reaction utilizing sodium nitrite (NaNO2) and iodine (I2), or alternatively using sodium bromide (NaBr) and nitrous acid (HNO2) depending on the specific step, the amino groups are transformed into halides. Specifically, Fmoc-butanediamine is converted to N-Fmoc-4-bromobutylamine, which is then treated with metallic magnesium to generate the corresponding Grignard reagent. This nucleophilic species is key to extending the carbon skeleton with high precision.
Simultaneously, the lysine backbone is prepared as an electrophilic partner. The alpha-amino group of Fmoc-Lys-OtBu is converted into an iodide functionality (I-Lys(Fmoc)-Otbu) using NaNO2 and I2 catalysis. The subsequent coupling of the Fmoc-butylamine Grignard reagent with this iodo-lysine derivative forms the critical carbon-carbon bond that establishes the second symmetric arm. This step is pivotal, as it replaces the native epsilon-amino side chain of lysine with a synthetic butylamine chain that mirrors the alpha-side modification. Finally, acidolysis with trifluoroacetic acid (TFA) removes the tert-butyl ester protection, yielding the free carboxylic acid form of DiFmoc-Lys(plus). This multi-step sequence avoids the use of transition metal catalysts that often leave toxic residues, thereby simplifying the purification profile and ensuring the final intermediate meets stringent pharmaceutical standards.
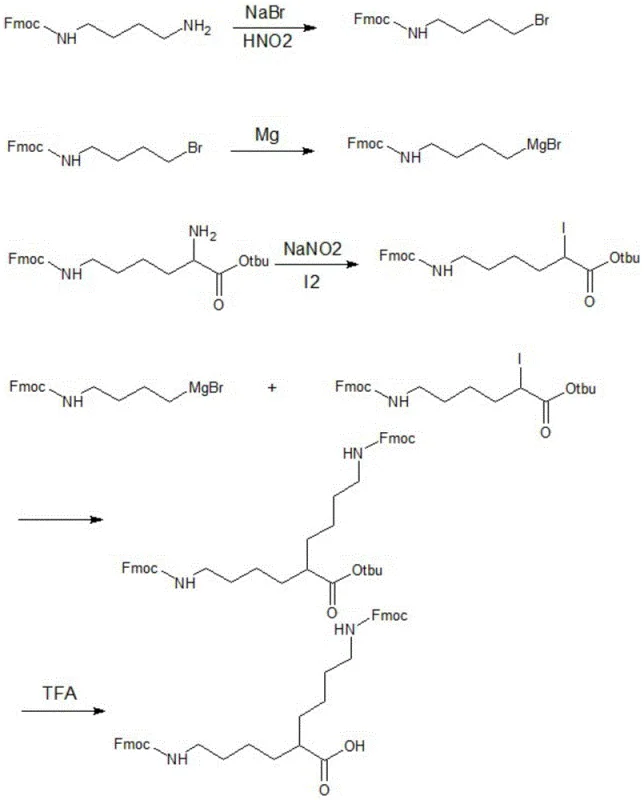
How to Synthesize DiFmoc-Lys(plus) Efficiently
The preparation of this novel linker requires precise control over reaction conditions to maximize yield and minimize byproduct formation. The patent outlines a robust five-step protocol that transitions from simple starting materials to the complex symmetric core. The process leverages standard protecting group strategies compatible with Fmoc solid-phase synthesis, ensuring seamless integration into existing peptide manufacturing workflows. Operators must pay close attention to the stoichiometry of the Grignard formation and the subsequent coupling to prevent homocoupling or incomplete reactions. The detailed standardized synthesis steps, including specific molar ratios and workup procedures, are provided in the guide below to facilitate immediate technology transfer.
- Convert Fmoc-butanediamine to N-Fmoc-4-bromobutylamine using NaBr and HNO2, then react with Magnesium to form the Grignard reagent.
- Transform NH2-Lys(Fmoc)-Otbu into I-Lys(Fmoc)-Otbu utilizing NaNO2 and Iodine catalysis.
- Couple the Grignard reagent with the Iodo-Lys derivative, followed by TFA acidolysis to remove the tert-butyl ester and yield DiFmoc-Lys(plus).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of DiFmoc-Lys(plus) translates into tangible operational efficiencies and risk mitigation. The primary value driver is the substantial reduction in downstream processing costs associated with peptide purification. In conventional MAPS synthesis, the heterogeneity of the crude product necessitates extensive and expensive preparative HPLC runs to isolate the correct isomer from deletion sequences. By ensuring uniform coupling efficiency through symmetric arm design, the new method significantly increases the purity of the crude polypeptide. This enhancement directly reduces the load on purification columns, decreases solvent consumption, and shortens the overall production cycle time. Furthermore, the elimination of complex enzymatic steps or exotic catalysts in the linker synthesis ensures a more stable and predictable supply of raw materials.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the drastic simplification of the purification workflow. Because the crude MAPS polypeptide produced using DiFmoc-Lys(plus) contains fewer deletion peptides and structural impurities, the yield of the final refined product is markedly improved. This efficiency gain means that less starting material is wasted, and the cost per gram of the active pharmaceutical ingredient is effectively lowered. Additionally, the synthesis route utilizes commodity chemicals like magnesium, bromides, and iodides, avoiding the price volatility associated with precious metal catalysts. This stability in raw material costs allows for more accurate long-term budgeting and pricing strategies for large-scale contracts.
- Enhanced Supply Chain Reliability: Supply continuity is critical for vaccine and therapeutic development programs. The synthetic route for DiFmoc-Lys(plus) is based on well-established organic transformations that are easily scalable from kilogram to tonne quantities. Unlike biotechnological methods that rely on fermentation and are susceptible to biological variability, this chemical synthesis approach offers consistent batch-to-batch reproducibility. The robustness of the Grignard reaction and the stability of the Fmoc-protected intermediates ensure that manufacturers can maintain safety stocks and meet tight delivery windows without compromising quality. This reliability is essential for supporting clinical trials and commercial launches where material availability cannot be a bottleneck.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, the process offers distinct advantages. The avoidance of heavy metal catalysts simplifies the waste stream treatment and reduces the environmental footprint of the manufacturing facility. The solvents used, such as THF, DMF, and acetone, are standard in the industry and can be efficiently recovered and recycled. The high atom economy of the coupling steps further contributes to a greener manufacturing profile. As regulatory bodies increasingly scrutinize the environmental impact of pharmaceutical production, adopting a synthesis route that minimizes hazardous waste and energy consumption positions the supply chain for long-term compliance and sustainability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating DiFmoc-Lys(plus) into their current development pipelines.
Q: Why is DiFmoc-Lys(plus) superior to traditional Lysine cores for MAPS synthesis?
A: Traditional Lysine cores have inconsistent arm lengths (alpha vs. epsilon amines), leading to uneven coupling efficiency and deletion peptides. DiFmoc-Lys(plus) features two completely symmetric butylamine arms, ensuring uniform reaction kinetics and significantly higher crude purity.
Q: What is the expected purity of MAPS polypeptides synthesized using this method?
A: According to patent data, the use of DiFmoc-Lys(plus) allows for the production of refined MAPS polypeptides with purity exceeding 95% to 98% after HPLC purification, drastically reducing the burden of downstream processing.
Q: Is this synthesis route scalable for industrial production?
A: Yes, the route relies on robust chemical transformations such as Grignard reactions and standard diazotization, avoiding exotic catalysts. This makes the process highly amenable to scale-up from laboratory grams to multi-kilogram commercial batches.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable DiFmoc-Lys(plus) Supplier
The development of DiFmoc-Lys(plus) represents a significant leap forward in the field of peptide vaccine intermediates, offering a solution to the long-standing challenge of structural heterogeneity in MAPS systems. NINGBO INNO PHARMCHEM stands at the forefront of translating such innovative academic and patent technologies into commercial reality. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the sensitive organometallic reactions required for this synthesis, ensuring that every batch meets stringent purity specifications. With our rigorous QC labs and commitment to quality, we guarantee that the DiFmoc-Lys(plus) supplied for your MAPS projects will deliver the consistent performance necessary for successful clinical outcomes.
We invite you to collaborate with us to leverage this advanced technology for your next-generation vaccine candidates. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and process constraints. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and competitive quotations. Let us help you accelerate your development timeline with a supply partner that understands the critical importance of purity and reliability in pharmaceutical manufacturing.
