Advanced Manufacturing of Montelukast Sodium Side Chain Intermediates for Global Pharma Supply
Advanced Manufacturing of Montelukast Sodium Side Chain Intermediates for Global Pharma Supply
The global demand for respiratory therapeutics continues to drive innovation in the synthesis of critical active pharmaceutical ingredients (APIs) and their precursors. Among these, Montelukast Sodium stands out as a cornerstone treatment for asthma and allergic rhinitis, necessitating a reliable supply of its complex molecular components. A pivotal breakthrough in this domain is documented in Chinese patent CN112094212A, which discloses a highly efficient preparation method for the Montelukast sodium side chain, specifically 1-(mercaptomethyl)cyclopropylacetic acid (MLM). This compound serves as the essential chiral building block that defines the pharmacological efficacy of the final drug. The structural complexity of Montelukast Sodium, featuring a quinoline ring, a vinyl bridge, and the critical cyclopropyl-acetic acid side chain, demands precision in every synthetic step.
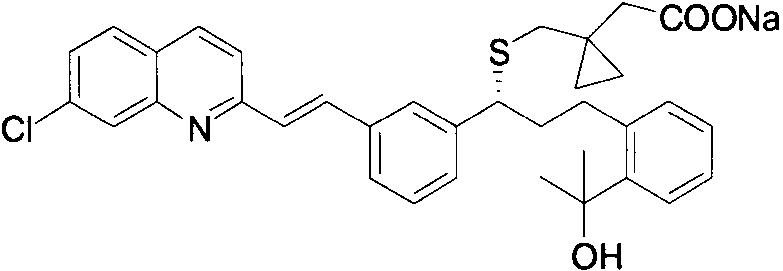
The significance of this patent lies in its ability to overcome historical bottlenecks associated with thiol-containing intermediates, which are notoriously prone to oxidation and difficult to purify. By introducing a novel thiobenzoate protection strategy, the inventors have created a pathway that not only enhances chemical stability during synthesis but also drastically simplifies the workup procedures. For R&D directors and procurement specialists alike, understanding this technological shift is crucial for securing a stable supply chain of high-purity pharmaceutical intermediates. The method described moves away from hazardous reagents and convoluted sequences, offering a streamlined approach that aligns with modern green chemistry principles while maintaining rigorous quality standards required by regulatory bodies worldwide.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1-(mercaptomethyl)cyclopropylacetic acid has been plagued by significant technical and safety challenges that hindered large-scale adoption. Early methodologies, such as those cited in WO9518107 and US5523477, relied on the conversion of 1,1-cyclopropyldimethanol into cyclic sulfites using thionyl chloride, followed by complex ring-opening reactions. These traditional routes suffered from poor atom utilization and generated a multitude of by-products that were exceptionally difficult to separate, often requiring multiple aqueous extractions and solvent swaps that eroded overall yield. Furthermore, other reported approaches, such as those in EP480717, utilized highly toxic and explosive reagents like diazomethane (CH2N2), posing unacceptable safety risks for industrial environments. The reliance on such hazardous materials not only increased operational costs due to specialized containment requirements but also resulted in low overall yields, with some literature reporting efficiencies as low as 28%, rendering them economically unviable for commercial API production.
The Novel Approach
In stark contrast to these legacy methods, the process outlined in CN112094212A introduces a paradigm shift by utilizing 1-bromocyclopropylacetonitrile (MLM-5) as a robust starting material. This innovative route leverages a nucleophilic substitution reaction with thiobenzoic acid under mild alkaline conditions to form a stable thioester intermediate, effectively masking the reactive thiol group until the final stages. This strategic protection prevents premature oxidation, a common failure mode in thiol chemistry, thereby preserving the integrity of the molecule throughout the synthesis. The subsequent in-situ alkaline hydrolysis simultaneously cleaves the thioester protecting group and hydrolyzes the nitrile functionality to the desired carboxylic acid, collapsing what were previously multiple discrete steps into a cohesive, efficient sequence. This consolidation of reaction steps not only minimizes material handling losses but also significantly reduces the volume of solvent and reagents required, directly translating to a more sustainable and cost-effective manufacturing profile.
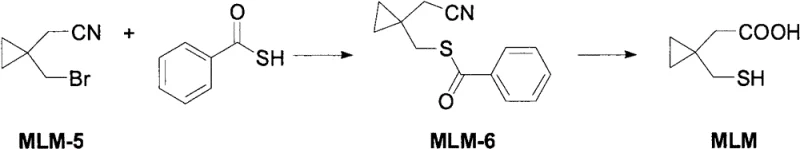
Mechanistic Insights into Thiobenzoate-Mediated Substitution and Hydrolysis
The core chemical innovation of this process rests on the dual functionality of thiobenzoic acid acting as both a sulfur source and a temporary protecting group. In the initial phase, the bromide leaving group on the cyclopropyl ring of MLM-5 is displaced by the thiobenzoate anion in a classic SN2-type mechanism, facilitated by the presence of inorganic bases such as lithium hydroxide or sodium hydroxide. The choice of alcoholic solvents like ethanol or isopropanol is critical here, as they provide the necessary polarity to dissolve the ionic reagents while maintaining a homogeneous reaction medium that supports efficient mass transfer. The formation of the S-benzoyl protected intermediate (MLM-6) is thermodynamically favorable and kinetically rapid at moderate temperatures ranging from 30°C to 50°C, ensuring high conversion rates without the need for energy-intensive heating or cooling cycles. This step is pivotal because it installs the sulfur atom in a protected state, rendering the intermediate stable against air oxidation, which is a persistent issue when handling free thiols in earlier synthetic attempts.
Following the substitution, the reaction mixture undergoes a transformative hydrolysis step upon the addition of aqueous base and elevation of temperature to between 60°C and 90°C. Under these conditions, the hydroxide ions attack both the thioester carbonyl and the nitrile carbon. The hydrolysis of the thioester releases the free thiol group and benzoic acid (as a salt), while the hydrolysis of the nitrile proceeds through an amide intermediate to finally yield the carboxylic acid moiety. The elegance of this mechanism lies in its orthogonality; the conditions are tuned such that the cyclopropane ring remains intact despite the harsh alkaline environment, avoiding ring-opening side reactions that could compromise the structural fidelity of the final product. The resulting crude product is then isolated by simple acidification to pH 3-4, which precipitates the target acid while keeping impurities in the aqueous phase, followed by a single recrystallization from isopropanol to achieve purity levels exceeding 99%, demonstrating exceptional control over the impurity profile.
How to Synthesize 1-(Mercaptomethyl)cyclopropylacetic Acid Efficiently
The practical implementation of this synthesis requires precise control over stoichiometry and thermal parameters to maximize yield and minimize impurity formation. The process begins with the suspension of 1-bromocyclopropylacetonitrile in a suitable alcohol solvent, followed by the sequential addition of the inorganic base and thiobenzoic acid under an inert nitrogen atmosphere to prevent any potential oxidative degradation. Maintaining the reaction temperature within the specified window of 30°C to 50°C for approximately 6 hours is essential to ensure complete consumption of the starting bromide before proceeding to the hydrolysis phase. Once the substitution is complete, the addition of aqueous base triggers the second transformation, requiring a temperature increase to 60-90°C and a holding time of roughly 8 hours to drive the hydrolysis to completion. The detailed standardized operating procedures, including specific molar ratios and workup protocols, are critical for reproducibility and are outlined in the technical guide below.
- Conduct a nucleophilic substitution reaction between 1-bromocyclopropylacetonitrile and thiobenzoic acid using an inorganic base in an alcoholic solvent at 30-50°C for 6 hours.
- Without isolation, add aqueous inorganic base to the reaction mixture and heat to 60-90°C for 8 hours to effect simultaneous hydrolysis of the nitrile and thioester groups.
- Acidify the mixture to pH 3-4, extract with ethyl acetate, and purify the crude product via recrystallization from isopropanol to obtain high-purity MLM.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers tangible strategic benefits that extend beyond mere chemical curiosity. The transition from hazardous, multi-step legacy processes to this streamlined thiobenzoate method fundamentally alters the cost structure and risk profile of producing Montelukast intermediates. By eliminating the need for specialized equipment to handle toxic gases like diazomethane or complex distillation setups for difficult separations, capital expenditure and operational overheads are significantly reduced. Furthermore, the use of commodity chemicals such as thiobenzoic acid and common inorganic bases ensures a resilient supply chain that is less susceptible to the volatility often seen with exotic reagents. This robustness translates directly into improved supply continuity, allowing manufacturers to meet fluctuating market demands for asthma medications without the fear of production stoppages due to raw material shortages.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the drastic simplification of the synthetic sequence. By combining the sulfur introduction and functional group transformation into a telescoped operation, the process eliminates intermediate isolation steps, which are typically labor-intensive and solvent-heavy. The removal of expensive transition metal catalysts or hazardous reagents further lowers the bill of materials, while the high yield (>92%) ensures that raw material costs are amortized over a greater output of saleable product. Additionally, the ability to use standard stainless steel reactors instead of lined vessels required for corrosive or toxic chemistries reduces maintenance costs and extends equipment lifespan, contributing to substantial long-term savings in pharmaceutical intermediate manufacturing.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the accessibility and stability of the raw materials employed in this route. Unlike precursors that require custom synthesis or have short shelf-lives, 1-bromocyclopropylacetonitrile and thiobenzoic acid are commercially available in bulk quantities from multiple global suppliers. This diversification of the supply base mitigates the risk of single-source dependency, a critical factor for procurement strategies aiming to secure long-term contracts. Moreover, the mild reaction conditions reduce the likelihood of batch failures due to thermal runaways or sensitivity to moisture, ensuring consistent production schedules and reliable delivery timelines for downstream API manufacturers who depend on just-in-time inventory models.
- Scalability and Environmental Compliance: As regulatory scrutiny on pharmaceutical waste intensifies, the environmental profile of a synthesis route becomes a key determinant of its commercial viability. This method generates wastewater with significantly lower ammonia nitrogen content compared to traditional ammonolysis routes, simplifying effluent treatment and reducing the burden on wastewater management infrastructure. The high atom economy and reduced solvent usage align with green chemistry metrics, facilitating easier regulatory approval and minimizing the environmental footprint of the manufacturing site. This compliance advantage not only avoids potential fines but also enhances the corporate sustainability profile, which is increasingly important for partnerships with major multinational pharmaceutical companies committed to ESG goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this method compares to established industry standards. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer and for commercial teams assessing the value proposition of this intermediate.
Q: What are the primary advantages of the thiobenzoate route over conventional methods?
A: The thiobenzoate route described in CN112094212A eliminates the need for toxic diazomethane and complex multi-step protections found in older patents. It offers a direct substitution-hydrolysis sequence that significantly improves atom economy and simplifies downstream purification, resulting in yields exceeding 92%.
Q: How does this process address environmental concerns in pharmaceutical manufacturing?
A: Traditional methods often generate significant hazardous waste, including ammonia nitrogen. This novel process utilizes mild inorganic bases and alcohol solvents, resulting in wastewater with low ammonia nitrogen content that is easier to treat biochemically, aligning with stricter environmental compliance standards.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the process is specifically designed for industrial scalability. It uses commercially available raw materials like 1-bromocyclopropylacetonitrile and avoids cryogenic conditions or highly unstable intermediates, making it robust for metric-ton scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-(Mercaptomethyl)cyclopropylacetic acid Supplier
The technical advancements detailed in patent CN112094212A represent a significant leap forward in the manufacture of respiratory drug intermediates, yet realizing this potential requires a partner with deep process engineering expertise. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to bring such innovations to the global market. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that ensure every batch of 1-(mercaptomethyl)cyclopropylacetic acid meets the exacting standards required for GMP API synthesis. We understand that consistency is key in pharmaceutical supply, and our state-of-the-art facilities are designed to handle sensitive thiol chemistry with the utmost care and precision.
We invite procurement leaders and R&D heads to engage with us to explore how this optimized synthesis route can enhance your supply chain efficiency. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your volume requirements. We encourage you to contact our technical procurement team today to索取 specific COA data and route feasibility assessments, ensuring that your next project is built on a foundation of chemical excellence and commercial reliability.
