Scalable Synthesis of Oxacephem Nucleus Intermediate ACB-7 via Continuous Chlorination and Cyclization
The pharmaceutical industry constantly seeks robust pathways for generating beta-lactam antibiotics, particularly the oxacephem class which includes vital drugs like flomoxef and moxalactam. Patent CN114805395B introduces a groundbreaking preparation method for the oxacephem parent nucleus intermediate, specifically designated as ACB-7. This technology represents a significant leap forward in synthetic efficiency, transitioning from fragmented, low-yield batch processes to a streamlined continuous reaction sequence starting from ACB-3. For R&D directors and process chemists, the core innovation lies in the ability to bypass the isolation of unstable hydroxyl intermediates, thereby drastically reducing purification burdens and enhancing overall throughput. The method not only addresses the historical challenges of low selectivity but also aligns with modern green chemistry principles by eliminating the use of highly corrosive and toxic reagents such as boron trifluoride complexes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of oxacephem nuclei has been plagued by discontinuous reaction steps that suffer from poor atom economy and hazardous reagent profiles. As illustrated in prior art such as Patent JP2005179336, traditional routes often involve a complex sequence of chlorine addition, elimination, hydrolysis, and ring closure, resulting in an overall yield of merely 54 percent. 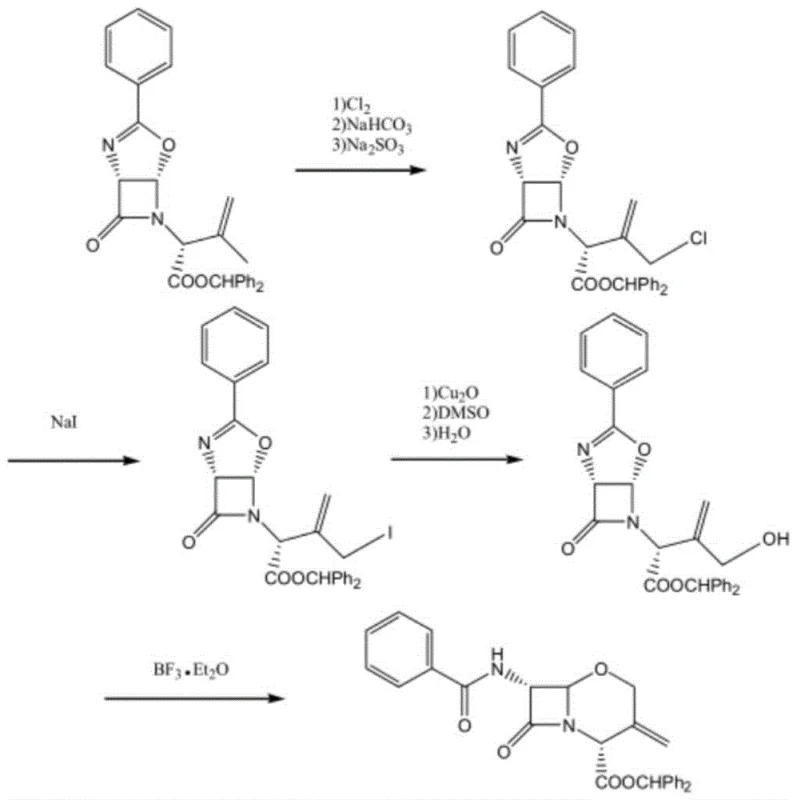 Furthermore, these legacy methods frequently rely on boron trifluoride etherate for the critical cyclization step, a substance that is not only highly corrosive and dangerous to handle but also strictly regulated in many chemical manufacturing jurisdictions due to its environmental impact. The necessity for anhydrous conditions in the final step, juxtaposed with aqueous hydrolysis in earlier steps, creates a logistical nightmare for process engineers, requiring extensive solvent swaps and drying protocols that inflate production costs and extend lead times significantly.
Furthermore, these legacy methods frequently rely on boron trifluoride etherate for the critical cyclization step, a substance that is not only highly corrosive and dangerous to handle but also strictly regulated in many chemical manufacturing jurisdictions due to its environmental impact. The necessity for anhydrous conditions in the final step, juxtaposed with aqueous hydrolysis in earlier steps, creates a logistical nightmare for process engineers, requiring extensive solvent swaps and drying protocols that inflate production costs and extend lead times significantly.
The Novel Approach
In stark contrast, the methodology disclosed in CN114805395B offers a cohesive, continuous pathway that transforms ACB-3 directly into the target ACB-7 with remarkable efficiency. 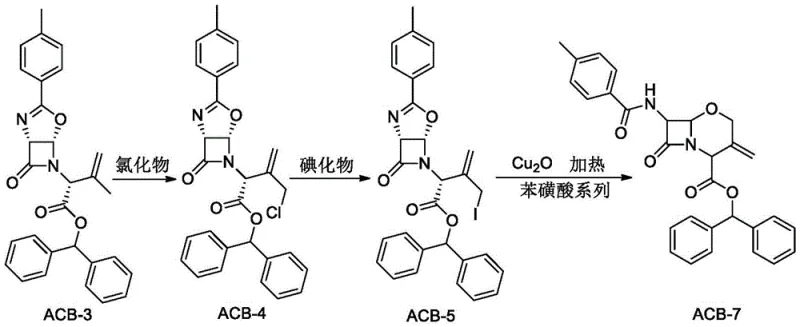 This novel approach integrates chlorination, iodination, and a unique copper-catalyzed cyclization into a unified workflow. By optimizing the reaction parameters, the yield for the conversion of ACB-3 to the chlorinated intermediate ACB-4 can reach 91.5 percent with 98.6 percent purity. Subsequently, the transformation to the iodide ACB-5 achieves yields up to 96.5 percent. Most critically, the final cyclization step avoids the isolation of the hydroxyl compound ACB-6 entirely. Instead, by employing a cuprous oxide and dimethyl sulfoxide system with a benzenesulfonic acid catalyst under controlled anhydrous conditions, the process drives the reaction directly to the bicyclic oxacephem structure. This eliminates the need for intermediate crystallization and solvent exchange, representing a paradigm shift in how these complex antibiotic cores are manufactured.
This novel approach integrates chlorination, iodination, and a unique copper-catalyzed cyclization into a unified workflow. By optimizing the reaction parameters, the yield for the conversion of ACB-3 to the chlorinated intermediate ACB-4 can reach 91.5 percent with 98.6 percent purity. Subsequently, the transformation to the iodide ACB-5 achieves yields up to 96.5 percent. Most critically, the final cyclization step avoids the isolation of the hydroxyl compound ACB-6 entirely. Instead, by employing a cuprous oxide and dimethyl sulfoxide system with a benzenesulfonic acid catalyst under controlled anhydrous conditions, the process drives the reaction directly to the bicyclic oxacephem structure. This eliminates the need for intermediate crystallization and solvent exchange, representing a paradigm shift in how these complex antibiotic cores are manufactured.
Mechanistic Insights into Copper-Catalyzed Cyclization and Halogen Exchange
The mechanistic elegance of this process is rooted in the precise control of halogen reactivity and the catalytic environment during ring closure. The initial chlorination of the allylic position in ACB-3 is carefully managed using chlorine gas in ethyl acetate at moderate temperatures (30-45°C), ensuring high regioselectivity and minimizing poly-chlorination side reactions. Following this, the substitution of chlorine with iodine using sodium iodide serves a dual purpose: it activates the allylic position for the subsequent nucleophilic attack and leverages the superior leaving group ability of iodide. The pivotal mechanistic breakthrough occurs in the final stage, where the inventors discovered that the presence of water typically leads to the stable hydroxyl intermediate ACB-6, necessitating a separate, harsh cyclization step. However, by rigorously excluding water and utilizing a Cu2O/DMSO/benzenesulfonic acid system, the reaction trajectory is altered. The copper species likely facilitates the activation of the allylic iodide, promoting an intramolecular nucleophilic attack by the proximal oxygen or nitrogen functionality to close the six-membered oxazine ring directly. This one-pot transformation not only simplifies the operation but also prevents the degradation often associated with isolating sensitive beta-lactam intermediates.
Impurity control is another cornerstone of this mechanistic design. In traditional routes, the use of boron trifluoride often leads to polymerization or decomposition of the sensitive beta-lactam ring if moisture is inadvertently introduced. The new copper-catalyzed system operates under milder acidic conditions provided by organic sulfonic acids like p-toluenesulfonic acid, which are far less aggressive than Lewis acids like BF3. This gentler environment preserves the stereochemical integrity of the chiral centers, which is paramount for the biological activity of the final antibiotic. Furthermore, the continuous nature of the process means that reactive intermediates are consumed immediately upon formation, reducing the residence time of unstable species and thereby suppressing the formation of oligomeric impurities. The result is a product profile with significantly higher purity, often exceeding 98 percent without the need for extensive chromatographic purification, which is a major advantage for GMP manufacturing environments.
How to Synthesize ACB-7 Efficiently
The synthesis of ACB-7 via this patented route is designed for operational simplicity and high reproducibility, making it an ideal candidate for technology transfer. The process begins with the dissolution of the starting material ACB-3 in ethyl acetate, followed by the controlled introduction of chlorine gas. Once the chlorination is complete, the reaction mixture is treated with an iodide source to effect the halogen exchange. The resulting solution of ACB-5 is then concentrated slightly to remove excess solvent before being introduced into the cyclization reactor containing the copper catalyst system. The detailed standardized synthesis steps, including specific stoichiometric ratios, temperature ramps, and workup procedures, are outlined below to ensure consistent high-quality output.
- Chlorinate ACB-3 using chlorine gas in ethyl acetate at 30-45°C to form ACB-4.
- React ACB-4 with sodium iodide in ethyl acetate at 35°C to generate the iodide intermediate ACB-5.
- Perform a one-pot cyclization of ACB-5 using cuprous oxide, DMSO, and p-toluenesulfonic acid without water isolation to yield ACB-7.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers profound strategic advantages beyond mere technical superiority. The elimination of boron trifluoride removes a significant bottleneck in raw material sourcing and hazardous waste disposal. Boron trifluoride complexes are not only expensive but also require specialized storage and handling infrastructure, increasing the overhead cost of manufacturing facilities. By replacing this with readily available cuprous oxide and organic sulfonic acids, the process drastically simplifies the supply chain and reduces the regulatory burden associated with toxic reagent management. Additionally, the ability to recycle the ethyl acetate solvent throughout the continuous process contributes to substantial cost savings in solvent procurement and waste treatment, aligning with sustainability goals that are increasingly important for multinational pharmaceutical partners.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the significant increase in overall yield and the reduction of unit operations. Traditional methods with yields around 54 percent require nearly double the amount of starting material to produce the same quantity of API intermediate compared to this high-yield route. Furthermore, by collapsing multiple steps into a continuous sequence and avoiding the isolation of intermediate ACB-6, the process saves on labor, energy, and equipment usage. The avoidance of expensive chlorinating agents like chlorosuccinimide in favor of elemental chlorine further drives down the raw material cost base, ensuring a more competitive pricing structure for the final intermediate.
- Enhanced Supply Chain Reliability: Supply continuity is critical for antibiotic production, and this method enhances reliability by utilizing commodity chemicals. Chlorine gas, sodium iodide, and ethyl acetate are globally available bulk chemicals with stable supply chains, unlike specialized reagents that may face geopolitical or logistical disruptions. The robustness of the reaction conditions, which tolerate a broader range of parameters without compromising quality, also reduces the risk of batch failures. This reliability ensures that downstream API manufacturers can maintain consistent production schedules, mitigating the risk of drug shortages in the market.
- Scalability and Environmental Compliance: From an environmental perspective, the process is inherently greener. The reduction in solvent usage through recycling and the elimination of fluorine-containing waste streams simplify the effluent treatment process. The moderate reaction temperatures (ranging from 10°C to 60°C) reduce energy consumption for heating and cooling compared to cryogenic or high-temperature alternatives. These factors make the technology highly scalable, allowing for seamless transition from pilot plant to multi-ton commercial production without the need for exotic reactor materials or complex safety systems, thus accelerating time-to-market for new antibiotic formulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oxacephem synthesis technology. These insights are derived directly from the experimental data and process optimization studies detailed in the patent documentation, providing clarity on yield expectations, safety protocols, and scalability potential for prospective manufacturing partners.
Q: How does this new method improve upon traditional oxacephem synthesis?
A: Traditional methods often rely on discontinuous steps with low yields (around 54%) and require highly corrosive boron trifluoride. This patented process achieves yields up to 96.5% per step and eliminates the need for toxic boron trifluoride by using a copper-catalyzed system.
Q: What are the critical reaction conditions for the final cyclization step?
A: The final conversion of ACB-5 to ACB-7 requires a specific anhydrous environment using dimethyl sulfoxide (DMSO) as the solvent, cuprous oxide as the catalyst, and a benzenesulfonic acid derivative. Crucially, water must be excluded to prevent the formation of the hydroxyl intermediate ACB-6, allowing direct ring closure.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is designed for industrial scalability. It utilizes common solvents like ethyl acetate which can be recycled, avoids hazardous reagents that complicate waste treatment, and operates at moderate temperatures (30-60°C), making it safer and more cost-effective for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable ACB-7 Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global supply of life-saving antibiotics. Our technical team has thoroughly analyzed the potential of the CN114805395B pathway and is prepared to leverage our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We understand that translating a patent into a robust commercial process requires more than just following a recipe; it demands rigorous process engineering and deep chemical intuition. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of verifying the structural integrity and impurity profile of complex beta-lactam intermediates like ACB-7, ensuring that every batch meets the exacting standards required by top-tier pharmaceutical companies.
We invite you to collaborate with us to optimize this technology for your specific production needs. Our experts can provide a Customized Cost-Saving Analysis to quantify the economic benefits of switching to this continuous manufacturing route. We encourage you to contact our technical procurement team to request specific COA data from our pilot runs and to discuss route feasibility assessments tailored to your facility's capabilities. Together, we can secure a sustainable and efficient supply chain for the next generation of oxacephem antibiotics.
