Revolutionizing 2-Phenethyl Alcohol Production: A Deep Dive into Novel Enzymatic Cascades for Commercial Scale
The global demand for high-purity aroma chemicals continues to surge, driven by the consumer shift towards 'green and natural' labeling in the food, cosmetic, and pharmaceutical sectors. At the forefront of this transition is 2-phenethyl alcohol, a compound renowned for its delicate rose-like fragrance, which serves as a critical flavor & fragrance intermediate. However, traditional manufacturing methods have long been plagued by environmental toxicity and low efficiency. A groundbreaking technical disclosure, detailed in patent CN106957878B, introduces a sophisticated biocatalytic strategy that fundamentally redefines the production landscape. This method leverages a specifically engineered tri-enzyme cascade within recombinant Escherichia coli to convert L-phenylalanine directly into 2-phenethyl alcohol. By bypassing the limitations of chemical synthesis and earlier fermentation attempts, this technology offers a robust pathway for achieving commercial scale-up of complex biocatalytic pathways while maintaining exceptional product purity.
The structural elegance of 2-phenethyl alcohol belies the complexity of its efficient synthesis. As illustrated in the molecular structure, the integration of an aromatic ring with a hydrophilic alcohol group requires precise control to avoid side reactions. The patented approach addresses this by utilizing a whole-cell biocatalyst system where wet thalli of E. coli transformed with phenylalanine dehydrogenase (Pdh), 2-ketoacid decarboxylase (Kdc), and alcohol dehydrogenase (ADH) genes act in concert. This biological machinery operates under mild aqueous conditions, eliminating the need for hazardous organic solvents typically associated with petrochemical-derived routes, thereby positioning suppliers who adopt this technology as leaders in sustainable flavor & fragrance intermediate manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of 2-phenethyl alcohol has relied heavily on chemical synthesis starting from benzene or styrene, processes fraught with significant drawbacks including high raw material toxicity and the generation of persistent pollutants. Even within the realm of biotechnology, previous attempts to synthesize this molecule via microbial transformation faced substantial metabolic bottlenecks. For instance, conventional enzymatic routes often utilized aromatic aminotransferases to convert L-phenylalanine into phenylpyruvic acid. As depicted in the comparative pathway analysis, this traditional transaminase approach necessitates the stoichiometric addition of an alpha-keto acid acceptor, such as oxaloacetic acid.
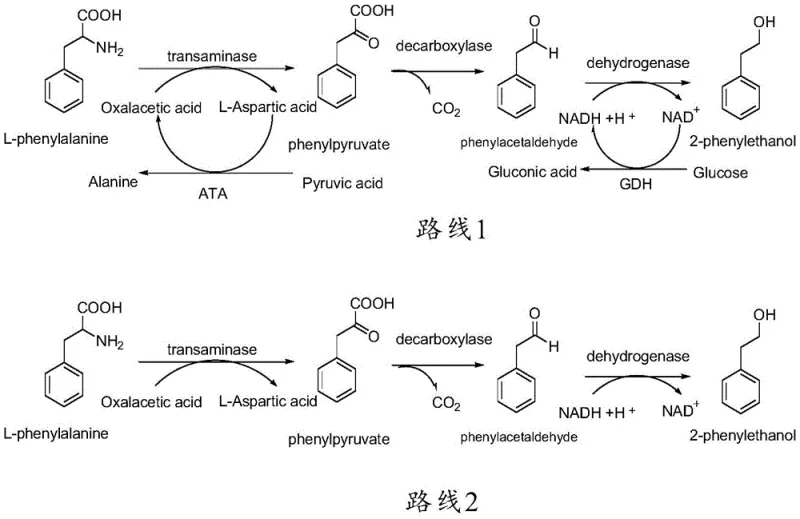
The reliance on oxaloacetic acid creates a cascade of inefficiencies: it increases raw material costs and generates equimolar amounts of L-aspartic acid as a byproduct. This amino acid byproduct complicates downstream processing, requiring energy-intensive separation steps to isolate the target alcohol, which drastically inflates production costs. Furthermore, maintaining the redox balance in these older systems often required the continuous addition of expensive coenzyme NADH or the coupling of auxiliary systems like glucose dehydrogenase, which in turn produced gluconic acid waste. These cumulative inefficiencies render conventional biocatalytic routes economically unviable for cost reduction in natural flavor manufacturing at a competitive scale.
The Novel Approach
The innovation disclosed in patent CN106957878B circumvents these historical hurdles by employing phenylalanine dehydrogenase (Pdh) as the primary catalyst for the initial deamination step. Unlike transaminases, Pdh catalyzes the direct oxidative deamination of L-phenylalanine to phenylpyruvic acid, releasing ammonia rather than transferring the amino group to a keto acid. This fundamental shift eliminates the need for oxaloacetic acid and prevents the formation of L-aspartic acid, thereby streamlining the reaction mixture. The subsequent steps involve a seamless handoff to 2-ketoacid decarboxylase (Kdc), which removes the carboxyl group to form phenylacetaldehyde, followed by reduction via alcohol dehydrogenase (ADH) to yield the final 2-phenethyl alcohol product.
This streamlined architecture not only simplifies the reaction stoichiometry but also enables a closed-loop cofactor recycling system. The NADH consumed in the final reduction step is regenerated during the initial oxidation step catalyzed by Pdh. Consequently, the system requires only a catalytic amount of NAD to be added once at the beginning, rather than continuous supplementation. This self-sustaining redox cycle significantly lowers the operational expenditure associated with cofactors, making the process highly attractive for reliable flavor & fragrance intermediate supplier networks aiming to optimize their cost structures without compromising on the 'natural' status of the final product.
Mechanistic Insights into the Tri-Enzyme Cascade
To fully appreciate the technical superiority of this method, one must examine the intricate interplay of the three recombinant enzymes within the E. coli host. The process initiates with the Pdh enzyme, derived from sources such as Bacillus badius or Thermoactinomyces intermedius, which binds L-phenylalanine and NAD+. This interaction facilitates the removal of the amino group as ammonium ions, concurrently reducing NAD+ to NADH and generating phenylpyruvic acid. This step is critical because it sets the redox balance for the entire cascade. The phenylpyruvic acid then diffuses to the active site of the Kdc enzyme, sourced from organisms like Lactococcus lactis or Pychrobacter cryohalolentis.
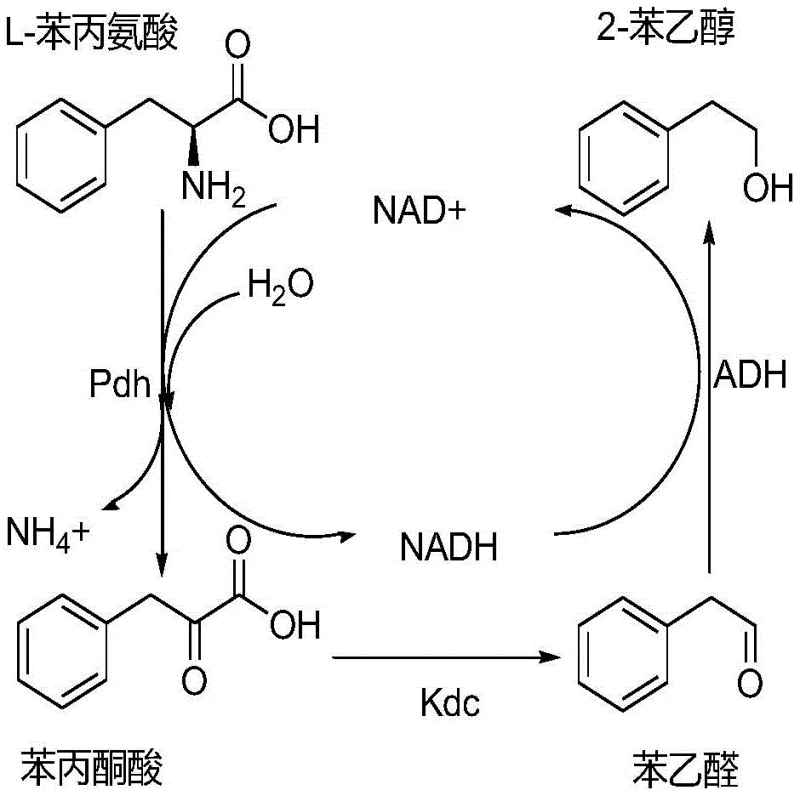
Within the Kdc active site, the alpha-keto acid undergoes non-oxidative decarboxylation, a reaction dependent on the cofactor thiamine pyrophosphate (TPP), releasing carbon dioxide and forming phenylacetaldehyde. This aldehyde intermediate is highly reactive and potentially toxic to the cell if allowed to accumulate; however, the presence of the third enzyme, ADH (from E. coli YahK or yeast Sfa1), ensures its rapid conversion. The ADH enzyme utilizes the NADH generated in the first step to reduce the aldehyde carbonyl group to a hydroxyl group, yielding 2-phenethyl alcohol and regenerating NAD+. This elegant mechanistic loop ensures that the concentration of toxic intermediates remains negligible, protecting cell viability and driving the equilibrium towards the product. The result is a highly efficient conversion where substrate utilization can reach up to 99%, demonstrating the potential for high-purity 2-phenethyl alcohol production with minimal impurity profiles.
How to Synthesize 2-Phenethyl Alcohol Efficiently
Implementing this biocatalytic route requires precise control over reaction parameters to maximize the synergy between the three enzymatic activities. The patent outlines a robust protocol where wet thalli of the recombinant strains are resuspended in specific buffer systems—such as PBS or Tris-HCl—and combined with the L-phenylalanine substrate. The optimization of the mass ratio of the three bacterial strains (e.g., Pdh:Kdc:ADH at 3:2:1) is paramount to prevent bottlenecking at any specific step of the cascade. Additionally, the maintenance of pH between 7 and 8 through the controlled addition of dilute acid helps neutralize the ammonia released during deamination, further stabilizing the enzymatic environment. For a comprehensive guide on the specific preparation of the recombinant strains and the exact formulation of the reaction buffers, please refer to the standardized synthesis protocol below.
- Prepare a reaction system containing L-phenylalanine substrate (5-50 g/L), coenzyme NAD (3-10 mM), and cofactor TPP (2-5 mM) in a buffered solution (pH 6.0-10.5).
- Introduce wet thalli of recombinant E. coli strains expressing Phenylalanine Dehydrogenase (Pdh), 2-Ketoacid Decarboxylase (Kdc), and Alcohol Dehydrogenase (ADH) at optimized mass ratios (e.g., 3: 2:1).
- Incubate the mixture at 25-40°C with shaking (150-250 rpm) for 2-24 hours, optionally controlling pH to 7-8, followed by centrifugation and extraction.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel biocatalytic platform represents a strategic opportunity to mitigate risk and enhance margin stability. The elimination of stoichiometric co-substrates like oxaloacetic acid removes a significant variable cost component that plagues traditional enzymatic routes. Furthermore, the avoidance of toxic chemical precursors such as benzene or styrene aligns the supply chain with increasingly stringent global environmental regulations, reducing the liability and cost associated with hazardous waste disposal. The ability to produce a 'natural' label-compliant ingredient using a fermentation-based process also opens up premium market segments that are inaccessible to chemically synthesized competitors, providing a distinct competitive advantage in the marketplace.
- Cost Reduction in Manufacturing: The proprietary tri-enzyme system fundamentally alters the cost structure by eliminating the need for expensive auxiliary substrates. In conventional transaminase routes, the requirement for alpha-keto acids acts as a tax on every mole of product produced; this new method removes that tax entirely. Additionally, the internal recycling of the NAD cofactor means that manufacturers do not need to purchase large quantities of this expensive reagent or invest in complex coupled-enzyme systems for regeneration. The reduction in downstream processing complexity, due to the absence of amino acid byproducts like L-aspartic acid, further drives down the cost per kilogram by simplifying purification and increasing overall yield.
- Enhanced Supply Chain Reliability: Reliance on petrochemical feedstocks exposes the supply chain to volatile oil prices and geopolitical instability. By shifting to L-phenylalanine, a widely available fermentation-derived amino acid, the production of 2-phenethyl alcohol becomes decoupled from fossil fuel markets. The use of robust E. coli expression systems ensures that the biocatalysts can be produced at scale with high consistency, guaranteeing a steady supply of the active enzyme components. This biological foundation provides a more predictable and resilient supply chain, capable of scaling rapidly to meet surges in demand without the long lead times associated with building new chemical synthesis infrastructure.
- Scalability and Environmental Compliance: The process operates under mild aqueous conditions (25-40°C, atmospheric pressure), which significantly reduces energy consumption compared to high-temperature chemical synthesis. The absence of heavy metal catalysts and chlorinated solvents simplifies wastewater treatment and ensures compliance with rigorous environmental standards in key manufacturing hubs. The high substrate conversion rates reported in the patent data indicate that the process is highly atom-economical, minimizing waste generation. This environmental efficiency not only reduces disposal costs but also enhances the brand value of the final product, appealing to eco-conscious consumers and corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this biocatalytic technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these details is crucial for R&D teams evaluating the feasibility of integrating this route into existing production lines.
Q: How does this biocatalytic method improve upon traditional transaminase routes?
A: Unlike conventional transaminase pathways that require expensive alpha-keto acid co-substrates (like oxaloacetic acid) and generate difficult-to-separate amino acid byproducts (like L-aspartic acid), this novel method utilizes phenylalanine dehydrogenase. This eliminates the need for additional keto acids and prevents the formation of stoichiometric amino acid waste, significantly simplifying downstream purification.
Q: What are the key advantages regarding cofactor usage in this process?
A: The process achieves efficient in-situ recycling of the coenzyme NAD. By coupling the oxidative deamination step with the final reduction step, the system only requires a catalytic amount of NAD to be added once, avoiding the continuous supplementation of expensive cofactors or the need for auxiliary enzyme systems like glucose dehydrogenase.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the method demonstrates high substrate conversion rates (up to 99%) and significant yield improvements (up to 15.5 g/L in optimized conditions) using robust recombinant E. coli strains. The mild reaction conditions and elimination of toxic chemical reagents make it highly scalable and compliant with strict environmental regulations for flavor and fragrance manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Phenethyl Alcohol Supplier
The technical breakthroughs detailed in patent CN106957878B underscore the immense potential of modern biocatalysis to transform the production of high-value aroma chemicals. At NINGBO INNO PHARMCHEM, we possess the extensive expertise required to translate such complex laboratory innovations into robust industrial realities. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot scale to full manufacturing is seamless and efficient. We maintain stringent purity specifications across all our facilities, supported by rigorous QC labs that utilize advanced analytical techniques to verify the identity and quality of every batch, guaranteeing that our clients receive a product that meets the highest global standards.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific application needs. Whether you require a Customized Cost-Saving Analysis to evaluate the economic benefits of switching to this biocatalytic route or need to review specific COA data for our current inventory, our technical procurement team is ready to assist. Contact us today to request route feasibility assessments and discover how we can support your supply chain with high-quality, sustainably produced 2-phenethyl alcohol.
