Advanced Solid-Phase Synthesis of Human Relaxin-2: Scaling High-Purity Peptide Hormones for Commercial Production
The pharmaceutical landscape for peptide hormones is constantly evolving, driven by the need for scalable, high-purity manufacturing processes that can meet rigorous regulatory standards. Patent CN102180964B introduces a transformative solid-phase synthesis method for Human Relaxin-2 (RLN2), a critical polypeptide hormone involved in cardiovascular regulation and reproductive physiology. This technology addresses the longstanding challenges associated with the chemical synthesis of complex peptides containing multiple disulfide bridges. By implementing a refined Fmoc-based strategy with orthogonal cysteine protection, the disclosed method achieves remarkable overall yields ranging from 74% to 82%, a substantial improvement over historical chemical methods that struggled with yields as low as 1.4%. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediates supplier, this patent outlines a robust pathway that simplifies production steps while effectively eliminating the impurity profiles typically associated with random chain combination strategies.
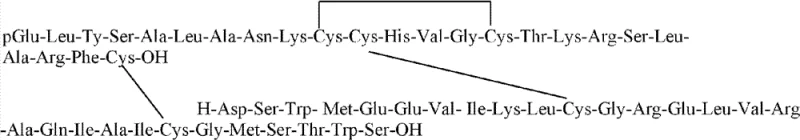
Human Relaxin-2 consists of an A chain with 24 amino acids and an intrachain disulfide bond, and a B chain with 29 amino acids, linked by two interchain disulfide bonds. The structural complexity, similar to insulin but with distinct solubility challenges, has traditionally favored recombinant DNA technology. However, the high cost and time-consuming nature of biotechnological production create a market gap for efficient chemical synthesis. This patent fills that gap by offering a method that is not only economically viable but also technically superior in terms of process control. The ability to produce high-purity RLN2 through total chemical synthesis provides a strategic alternative for supply chain diversification, ensuring continuity even when biological expression systems face bottlenecks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the chemical synthesis of Human Relaxin-2 has been plagued by inefficiencies that render it unsuitable for industrial scale-up. Prior art, such as the methods disclosed in early literature, often relied on a hybrid approach where the A chain was synthesized using Fmoc chemistry while the B chain utilized Boc chemistry. This disjointed strategy necessitated the use of hazardous hydrogen fluoride (HF) for deprotection at multiple stages, significantly increasing operational risks and waste disposal costs. Furthermore, these conventional methods required separate synthesis of chains followed by a complex three-step combination process to form the disulfide bonds. The use of different protecting groups for different cysteine residues added layers of complexity, leading to extensive purification requirements and catastrophic yield losses. In some documented cases, the overall yield calculated based on the B chain input was a mere 1.4%, making the process economically unacceptable for commercial manufacturing. Additionally, the poor water solubility of the RLN2 B-chain often led to aggregation during Fmoc synthesis, resulting in incomplete couplings and deletion sequences that were difficult to remove.
The Novel Approach
The methodology presented in CN102180964B revolutionizes this workflow by adopting a unified Fmoc solid-phase synthesis strategy for both chains, utilizing Wang resin as a versatile carrier. A key innovation lies in the specific selection of orthogonal protecting groups for the cysteine residues: Cys(Trt), Cys(Acm), and Cys(4-MeBzl). This allows for the selective formation of the intrachain disulfide bond in the A-chain while the peptide is still attached to the resin or immediately after partial deprotection, preventing misfolding. The B-chain synthesis is optimized using a specialized solvent system containing Guanidinium hydrochloride, which acts as a chaotropic agent to keep the hydrophobic peptide chain solvated and reactive. By streamlining the deprotection and cleavage steps into a more cohesive workflow using trifluoroacetic acid (TFA) cocktails with scavengers like thioanisole and phenol, the process drastically reduces the number of isolation steps. This results in a significant reduction in cost reduction in pharmaceutical intermediates manufacturing by minimizing solvent consumption and resin usage while maximizing the recovery of the active pharmaceutical ingredient.
Mechanistic Insights into Orthogonal Disulfide Bond Formation
The core technical breakthrough of this synthesis lies in the precise management of the three disulfide bonds inherent to the Relaxin structure. The process begins with the assembly of the linear A-chain precursor on Wang resin, incorporating Cys(Trt) at positions 10 and 15, Cys(Acm) at position 11, and Cys(4-MeBzl) at position 24. The mechanistic elegance is revealed during the cyclization step, where a mixture of hydrogen bromide and acetic acid (3:7 v/v) is employed to selectively remove the acid-labile Trityl (Trt) groups without affecting the Acm or 4-MeBzl protections. Once the free thiols at Cys10 and Cys15 are exposed, the resin is treated with a water/acetonitrile/DMSO mixture, facilitating air oxidation to form the correct intrachain disulfide bridge. This on-resin or semi-on-resin cyclization ensures that the A-chain adopts the correct conformation early in the process, which is thermodynamically favorable for the subsequent folding of the complete hormone.
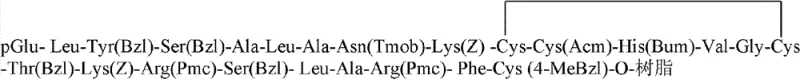
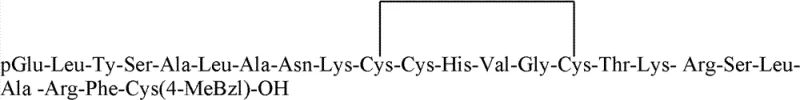
Following the formation of the monocyclic A-chain, the side-chain protecting groups are removed using a cocktail of trimethylammonium bromosilane, thioanisole, and TFA, yielding the free A-chain with the Cys(4-MeBzl) group intentionally retained at the C-terminus. This retention is critical for the final ligation step. Similarly, the B-chain is synthesized with Cys(Acm) and Cys(4-MeBzl) protections strategically placed to match the pairing requirements of the native hormone. When the purified A and B chains are mixed in a DMSO/DMF/TFA solvent system at a molar ratio of 1:1.2, the specific protecting groups guide the formation of the interchain disulfide bonds. The reaction conditions promote the selective interaction between the free thiols and the protected cysteines or between two free thiols generated in situ, ultimately driving the equilibrium towards the native, biologically active structure with high fidelity. This controlled oxidative folding minimizes the formation of scrambled isomers, which are the primary source of impurities in peptide synthesis.
How to Synthesize Human Relaxin-2 Efficiently
The synthesis of Human Relaxin-2 via this patented route requires strict adherence to the specified coupling protocols and deprotection sequences to ensure the high yields reported. The process leverages standard solid-phase peptide synthesis (SPPS) equipment but differentiates itself through the specific reagent ratios and solvent compositions detailed in the examples. For instance, the use of TBTU/HOBt/DIEA for the A-chain and PyBOP/HOBt for the B-chain optimizes coupling efficiency for different sequence contexts. The following guide outlines the critical operational phases derived from the patent data, serving as a foundational reference for process chemists aiming to replicate this high-efficiency route. Detailed standardized synthesis steps are provided in the section below.
- Synthesize the monocyclic A-chain on Wang resin using orthogonal Cys protection (Trt/Acm/4-MeBzl), performing on-resin cyclization between Cys10 and Cys15 using HBr/AcOH deprotection followed by air oxidation.
- Assemble the linear B-chain using PyBOP/HOBt coupling with specific side-chain protections (Acm/4-MeBzl) to prevent premature disulfide formation, followed by global deprotection and cleavage.
- Ligate the purified A and B chains in a DMSO/DMF/TFA solvent system at a 1: 1.2 molar ratio, allowing spontaneous formation of the interchain disulfide bonds to yield native Human Relaxin-2.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis method translates into tangible operational improvements beyond simple yield metrics. The shift from a multi-step, hazardous HF-based process to a streamlined TFA-based Fmoc protocol significantly enhances workplace safety and reduces the regulatory burden associated with handling anhydrous hydrogen fluoride. This simplification of the chemical workflow directly correlates to a more robust supply chain, as the reliance on specialized, hazard-intensive infrastructure is minimized. Furthermore, the high purity achieved through the orthogonal protection strategy reduces the load on downstream purification processes, such as preparative HPLC, which are often the bottleneck in peptide manufacturing. By reducing the complexity of impurity profiles, manufacturers can achieve faster batch release times and lower overall production costs.
- Cost Reduction in Manufacturing: The dramatic increase in overall yield from historical lows of 1.4% to the 74-82% range reported in this patent fundamentally alters the cost structure of RLN2 production. Higher yields mean that less raw material, specifically expensive Fmoc-protected amino acids and resin, is required to produce the same amount of final API. Additionally, the elimination of multiple intermediate purification steps and the reduction in solvent volume due to fewer reaction cycles contribute to substantial cost savings. The use of commercially available Fmoc-amino acids further ensures that raw material sourcing remains stable and competitive, avoiding the price volatility often associated with custom-synthesized building blocks.
- Enhanced Supply Chain Reliability: The robustness of the Fmoc solid-phase strategy allows for greater flexibility in production scheduling. Unlike recombinant methods that require long fermentation cycles and complex biological containment, chemical synthesis can be initiated and completed within a predictable timeframe using standard reactor vessels. The method's tolerance for scale-up, supported by the use of Wang resin which is widely available in bulk quantities, ensures that supply can be ramped up quickly to meet market demand. The reduced risk of batch failure due to aggregation or misfolding means that delivery timelines are more reliable, mitigating the risk of stockouts for downstream drug product manufacturers.
- Scalability and Environmental Compliance: From an environmental perspective, replacing HF with TFA-based cleavage cocktails significantly reduces the generation of highly corrosive and toxic waste streams. The process is compatible with microwave-assisted synthesis, which can further reduce reaction times and energy consumption, aligning with green chemistry principles. The simplified workflow also facilitates easier validation and technology transfer between manufacturing sites, ensuring consistent quality across different production scales. This scalability is crucial for meeting the growing global demand for peptide therapeutics while maintaining strict adherence to environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this solid-phase synthesis technology. These answers are derived directly from the experimental data and claims within patent CN102180964B, providing clarity on the feasibility and advantages of this specific manufacturing route. Understanding these details is essential for stakeholders evaluating the potential for licensing or contracting this technology for commercial production.
Q: What is the primary advantage of this solid-phase method over recombinant DNA technology for RLN2?
A: While recombinant technology is common, this chemical synthesis method described in CN102180964B offers a streamlined alternative that avoids complex biological fermentation purification. It achieves significantly higher chemical yields (up to 82%) compared to traditional mixed-strategy chemical syntheses which often yielded as low as 1.4%, providing a more cost-effective route for specific high-purity batches.
Q: How does the patent address the difficulty of synthesizing the hydrophobic B-chain?
A: The patent utilizes a specific solvent system containing Guanidinium hydrochloride, DMF, and ethanol during the B-chain assembly. This unique mixture improves the solvation of the growing peptide chain, preventing aggregation and ensuring high coupling efficiency even for the difficult, hydrophobic sequences found in the Relaxin B-chain.
Q: What orthogonal protection strategy is used for the three disulfide bonds?
A: The method employs a sophisticated orthogonal protection scheme using Cys(Trt), Cys(Acm), and Cys(4-MeBzl). The intrachain A-chain bond is formed first by selectively removing Trt groups. The remaining Acm and 4-MeBzl groups protect the other cysteines during cleavage, allowing for controlled interchain bonding in the final ligation step without scrambling.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Human Relaxin-2 Supplier
The technical advancements detailed in CN102180964B represent a significant leap forward in the chemical manufacturing of complex peptide hormones. At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating such high-potency laboratory methodologies into robust, commercial-scale processes. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate disulfide bridging and orthogonal protection strategies required for RLN2 are executed with precision. Our facilities are equipped with state-of-the-art peptide synthesizers and rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of Human Relaxin-2 meets the highest international standards for safety and efficacy.
We invite pharmaceutical partners to collaborate with us to leverage this advanced synthesis route for your pipeline projects. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and comprehensive route feasibility assessments, ensuring that your supply of high-purity Human Relaxin-2 is secure, cost-effective, and ready for clinical or commercial deployment.
