Scalable Synthesis of Diethyl Naphthylmethyl-Tetrahydrofurfurylmalonate via Green MeTHF Chemistry for Commercial API Production
The pharmaceutical industry continuously seeks robust, scalable, and environmentally sustainable pathways for the production of critical cardiovascular intermediates. Patent CN101381350B discloses a highly efficient synthetic method for diethyl naphthylmethyl-tetrahydrofurfurylmalonate, a pivotal precursor in the manufacture of nafronyl oxalate, a potent vasodilator used for treating cerebrovascular diseases. This technical disclosure represents a significant departure from traditional methodologies by replacing hazardous reagents and problematic solvents with a greener, more manageable system centered around 2-methyltetrahydrofuran (MeTHF). For R&D directors and process chemists, this patent offers a blueprint for enhancing reaction safety and simplifying downstream processing, while supply chain leaders will find value in the reported solvent recovery rates exceeding 97%, which directly correlates to reduced raw material consumption and waste disposal costs. The transition to this novel protocol addresses long-standing pain points in the synthesis of complex malonate derivatives, specifically targeting the elimination of emulsion formation and the dangers associated with pyrophoric bases.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of menaphthyl tetrahydrofurfuryl dialkyl malonates has relied heavily on the use of sodium hydride (NaH) suspended in polar aprotic solvents such as dimethylformamide (DMF) or alcoholic systems. While chemically effective on a small laboratory scale, these conventional methods present severe drawbacks when translated to industrial manufacturing environments. Sodium hydride is notoriously pyrophoric, posing substantial fire and explosion risks that necessitate expensive safety infrastructure and rigorous handling protocols, thereby inflating operational expenditures. Furthermore, the use of DMF creates significant downstream processing challenges; because DMF is fully miscible with water, the reaction mixture cannot be simply washed to remove inorganic salts. Instead, manufacturers are forced to introduce a third solvent, such as toluene, to facilitate extraction, which complicates the solvent recovery loop and increases the overall carbon footprint of the process. Additionally, the interaction between DMF, water, and organic byproducts frequently leads to the formation of stubborn emulsions, causing difficult phase separation, prolonged batch times, and potential product loss, all of which erode profit margins and compromise supply chain reliability.
The Novel Approach
The innovative methodology outlined in the patent data fundamentally re-engineers this synthesis by substituting the hazardous sodium hydride with safer sodium alkoxides and replacing DMF with 2-methyltetrahydrofuran (MeTHF). This strategic shift leverages the unique physicochemical properties of MeTHF, a cyclic ether that acts as an excellent Lewis base while remaining immiscible with water. This immiscibility is the cornerstone of the process improvement, as it allows for a homogeneous reaction environment that transitions seamlessly into a biphasic system upon the addition of water for workup. Consequently, the organic product partitions cleanly into the MeTHF layer, leaving inorganic salts in the aqueous phase without the need for auxiliary extraction solvents or the risk of emulsion formation. The reaction proceeds under mild thermal conditions, typically involving a reflux period of 0.5 to 2 hours for enolate formation followed by the addition of the halide at 80 to 100°C. This streamlined approach not only enhances operator safety but also drastically simplifies the isolation procedure, enabling high-purity product recovery through simple distillation of the solvent.
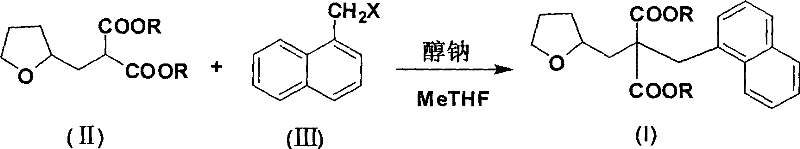
Mechanistic Insights into Sodium Alkoxide-Mediated Alkylation in MeTHF
The core chemical transformation involves the generation of a stabilized carbanion/enolate species from the tetrahydrofurfuryl malonate derivative, which subsequently undergoes nucleophilic substitution with 1-halogenated methyl naphthalene. In the presence of sodium alkoxide, the acidic alpha-protons of the malonate ester are deprotonated to form the corresponding sodium enolate. MeTHF serves as an ideal medium for this step due to its ability to solvate the sodium cation effectively without overly stabilizing the anionic nucleophile, thus maintaining high reactivity towards the electrophilic benzylic halide. The reaction temperature is carefully controlled, initially kept between 20 and 45°C during the base addition to prevent exothermic runaway, and then elevated to 80-100°C during the alkylation phase to overcome the activation energy barrier for the SN2 displacement. The use of a slight excess of base (molar ratio of 1.0:1.0~1.3 relative to the malonate) ensures complete conversion of the starting material, minimizing the presence of unreacted ester which could complicate purification. Furthermore, the steric and electronic environment provided by the MeTHF solvent shell likely contributes to the high regioselectivity observed, preventing side reactions such as O-alkylation or elimination that might occur in more polar or protic media.
Impurity control is intrinsically built into this solvent system through the physical separation mechanisms enabled by MeTHF. In traditional DMF processes, trace impurities often co-elute or remain trapped in the solvent matrix, requiring extensive chromatographic purification or recrystallization steps that reduce overall yield. In contrast, the MeTHF process allows for a rigorous aqueous wash immediately following the reaction. Since MeTHF does not mix with water, any water-soluble impurities, including excess sodium alkoxide, sodium halide salts generated during the reaction, and polar byproducts, are efficiently partitioned into the aqueous waste stream. The patent data indicates that a simple brine wash followed by drying over anhydrous sodium sulfate is sufficient to prepare the organic phase for solvent recovery. This efficiency in impurity rejection is evidenced by the reported gas phase purity levels consistently exceeding 99.0% across multiple embodiments. The stability of the tetrahydrofuran ring under these basic conditions is also preserved, as MeTHF is less prone to ring-opening polymerization or degradation compared to THF in the presence of strong bases, ensuring the structural integrity of the furfuryl moiety in the final API intermediate.
How to Synthesize Diethyl Naphthylmethyl-Tetrahydrofurfurylmalonate Efficiently
To implement this synthesis effectively, operators must adhere to precise stoichiometric ratios and thermal profiles as validated by the patent examples. The process begins with the charging of the tetrahydrofurfuryl diester and MeTHF into a reactor, followed by the controlled addition of the base. Detailed standard operating procedures regarding agitation speeds, addition rates, and specific distillation parameters are critical for reproducing the high yields and solvent recovery rates documented. For a comprehensive breakdown of the exact experimental conditions, reagent grades, and equipment specifications required to replicate this process safely, please refer to the standardized synthesis guide below.
- Charge tetrahydrofurfuryl diester malonate and 2-methyltetrahydrofuran into a reactor, then add sodium alkoxide in batches at 20-45°C.
- Heat the mixture to reflux for 0.5-2 hours to form the enolate, then slowly drip 1-halogenated methyl naphthalene at 80-100°C.
- Reflux for 0.5-5 hours, cool, add water to separate layers, extract the aqueous phase with MeTHF, and recover the solvent via distillation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this MeTHF-based synthetic route offers tangible economic and logistical benefits that extend beyond mere chemical yield. The primary advantage lies in the drastic simplification of the workup procedure, which translates directly into reduced cycle times and lower utility consumption. By eliminating the need for solvent exchanges and complex emulsion-breaking steps, manufacturing facilities can increase batch throughput significantly without requiring additional capital investment in new equipment. Furthermore, the inherent safety profile of using sodium alkoxides instead of pyrophoric sodium hydride reduces the regulatory burden and insurance costs associated with handling hazardous materials, making the supply chain more resilient to safety audits and compliance inspections. The ability to recover and reuse the reaction solvent at high efficiency also insulates the production cost from volatility in raw material pricing, providing a more predictable cost of goods sold (COGS) structure for long-term contracts.
- Cost Reduction in Manufacturing: The implementation of this green chemistry protocol drives cost efficiency primarily through solvent management and waste reduction. Since MeTHF serves as both the reaction medium and the extraction solvent, the need for purchasing secondary solvents like toluene or hexane is completely eliminated, reducing the total volume of chemicals required per kilogram of product. Moreover, the patent reports a solvent recovery rate of over 97%, meaning that the vast majority of the MeTHF can be distilled and recycled back into the process, drastically lowering the recurring cost of raw materials. The avoidance of emulsions also prevents product loss during the aqueous workup, ensuring that the theoretical yield is closely matched by the isolated yield, which maximizes the return on investment for every batch produced.
- Enhanced Supply Chain Reliability: From a logistics perspective, the switch to non-pyrophoric reagents simplifies the transportation and storage of raw materials. Sodium alkoxides are generally more stable and easier to handle than sodium hydride, reducing the risk of shipping delays caused by hazardous material restrictions. Additionally, the robustness of the reaction conditions, which tolerate a range of temperatures and molar ratios without significant degradation in quality, ensures consistent production output even if minor variations occur in feedstock quality. This process reliability is crucial for maintaining continuous supply to downstream API manufacturers, minimizing the risk of stockouts that could disrupt the production of life-saving cardiovascular medications.
- Scalability and Environmental Compliance: The environmental profile of this process aligns perfectly with modern green chemistry initiatives, facilitating easier permitting and regulatory approval for scale-up. MeTHF is derived from renewable resources and has a lower toxicity profile compared to chlorinated solvents or DMF, reducing the environmental liability of the manufacturing site. The high recovery rate of the solvent minimizes volatile organic compound (VOC) emissions and liquid waste generation, simplifying wastewater treatment requirements. This sustainability advantage not only future-proofs the supply chain against tightening environmental regulations but also enhances the brand reputation of the supplier as a responsible partner in the pharmaceutical value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of diethyl naphthylmethyl-tetrahydrofurfurylmalonate. These answers are derived directly from the experimental data and technical specifications provided in the patent literature, ensuring accuracy and relevance for process development teams. Understanding these nuances is essential for evaluating the feasibility of technology transfer and for optimizing the commercial production strategy.
Q: Why is 2-Methyltetrahydrofuran (MeTHF) preferred over DMF for this alkylation?
A: MeTHF is immiscible with water, allowing for direct phase separation during workup without the need for additional extraction solvents like toluene. Unlike DMF, which forms difficult emulsions and requires complex removal, MeTHF offers a recovery rate exceeding 97%, significantly simplifying the purification process and reducing environmental impact.
Q: What are the safety advantages of using sodium alkoxide instead of sodium hydride?
A: Sodium hydride (NaH) is pyrophoric and poses significant fire and explosion risks, especially on an industrial scale. Substituting it with sodium alkoxide (such as sodium methoxide or ethoxide) eliminates these severe safety hazards, making the process much more suitable for large-scale commercial manufacturing and reducing insurance and safety compliance costs.
Q: What purity levels can be achieved with this synthetic route?
A: According to the patent data, this method consistently achieves gas phase purity levels above 99.0%, with yields ranging typically between 80% and 86%. The efficient removal of impurities is facilitated by the clean phase separation properties of the MeTHF solvent system.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diethyl Naphthylmethyl-Tetrahydrofurfurylmalonate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global supply of cardiovascular therapeutics. Our technical team has extensively analyzed the MeTHF-based synthetic route described in CN101381350B and possesses the engineering expertise to translate this laboratory-scale innovation into a robust, commercial-scale manufacturing process. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications, consistently delivering material that meets or exceeds the 99% purity benchmarks established in the patent data.
We invite you to collaborate with us to optimize your supply chain for nafronyl oxalate precursors. By leveraging our process development capabilities, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how the adoption of this green solvent technology can improve your bottom line. Please contact our technical procurement team today to request specific COA data, discuss route feasibility assessments, and explore how our manufacturing excellence can support your long-term strategic goals.
