Industrial Scale Production of Asarin Intermediates via Optimized Acylation and Reduction
Introduction to Advanced Asarin Intermediate Manufacturing
The pharmaceutical industry constantly seeks robust, scalable, and cost-effective pathways for synthesizing critical active pharmaceutical ingredients (APIs) and their precursors. A significant breakthrough in this domain is documented in Chinese Patent CN101492351A, which outlines a novel, three-step synthetic route for producing Asarin, chemically known as E-2,4,5-trimethoxy-1-propenylbenzene. This compound serves as a vital expectorant and anti-inflammatory agent, widely utilized in the treatment of respiratory conditions such as bronchitis and asthma. The patent details a transformative approach that shifts away from traditional, hazardous methodologies towards a safer, more economically viable process utilizing 1,2,4-trimethoxybenzene as the starting material. By leveraging standard Friedel-Crafts acylation followed by reduction and dehydration, this technology addresses key pain points in fine chemical manufacturing, including safety, waste generation, and purification complexity. For global procurement leaders and R&D directors, understanding this proprietary methodology offers a strategic advantage in securing reliable supply chains for high-quality respiratory therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of propenyl-2,4,5-trimethoxybenzene has relied heavily on Grignard reactions, a pathway fraught with operational hazards and logistical challenges. As illustrated in the prior art, the conventional route involves reacting 2,4,5-trimethoxybenzaldehyde with ethylmagnesium bromide to generate an alcohol intermediate, which is subsequently dehydrated. This traditional method imposes stringent requirements for anhydrous conditions and necessitates the use of highly flammable solvents such as diethyl ether or tetrahydrofuran (THF). Furthermore, the purification of the final product often requires molecular distillation, a energy-intensive and technically demanding unit operation that complicates industrial scale-up. The reliance on Grignard reagents also introduces significant safety risks due to their pyrophoric nature, demanding specialized equipment and rigorous safety protocols that drive up capital expenditure and operational costs for manufacturing facilities.
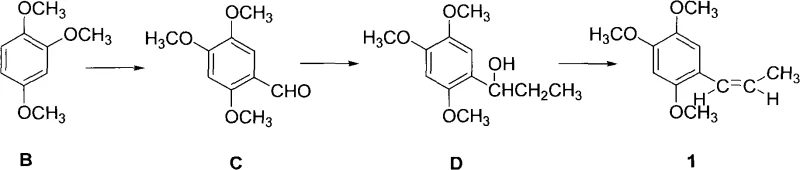
The Novel Approach
In stark contrast, the innovative process disclosed in patent CN101492351A circumvents these difficulties by employing a sequence of acylation, reduction, and dehydration reactions that are inherently safer and more manageable. This new strategy utilizes readily available and inexpensive raw materials such as propionic anhydride or propionyl chloride, coupled with Lewis acid catalysts like anhydrous aluminum chloride. A distinct advantage of this methodology is that the intermediates generated at each stage are solids, which significantly simplifies isolation and purification through standard crystallization techniques rather than complex distillation. This shift not only enhances the overall yield but also drastically reduces the generation of hazardous waste, aligning with modern green chemistry principles. By eliminating the need for sensitive organometallic reagents and high-vacuum distillation, the process offers a streamlined, robust alternative that is ideally suited for continuous, large-volume production environments.
Mechanistic Insights into the Three-Step Synthetic Sequence
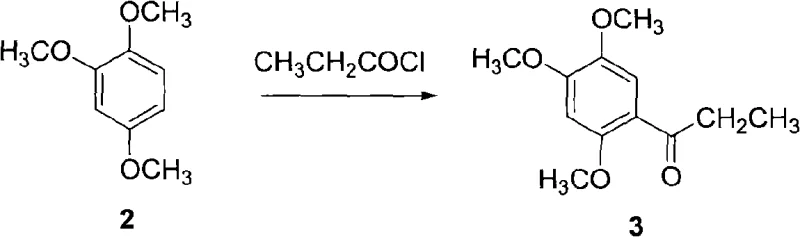
The core of this technological advancement lies in the precise control of reaction parameters across three distinct chemical transformations, beginning with the Friedel-Crafts acylation. In the first step, 1,2,4-trimethoxybenzene undergoes acylation with propionic anhydride or propionyl chloride in the presence of anhydrous aluminum chloride or zinc chloride. The reaction is typically initiated at low temperatures, around 15°C, to control exothermicity, before being warmed to a range of 40-70°C to ensure complete conversion over a period of 2 to 5 hours. The stoichiometry is carefully balanced, with a molar ratio of substrate to acylating agent to catalyst maintained at approximately 1:(1.5-2.0):(2.0-2.5). This step yields 2,4,5-trimethoxypropiophenone as a solid product with high purity, setting a strong foundation for the subsequent reduction phase.
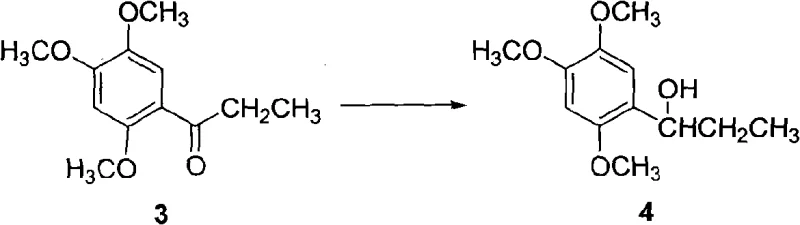
Following acylation, the ketone intermediate is subjected to a reduction reaction to form the corresponding alcohol, 2,4,5-trimethoxyphenylpropyl alcohol. This transformation is achieved using hydride reducing agents such as sodium borohydride, lithium aluminum hydride, or potassium borohydride, with sodium borohydride being preferred for its efficacy and handling safety. The reaction is conducted in solvents like ethyl acetate or THF, starting at cryogenic temperatures of -5°C to manage the reaction rate, and then gradually warming to 45°C to drive the reaction to completion as monitored by TLC. The resulting alcohol is isolated as a solid after solvent removal and crystallization, demonstrating excellent yield characteristics. This solid-state intermediate is crucial for supply chain stability, as solids are generally more stable and easier to transport than liquid oils.
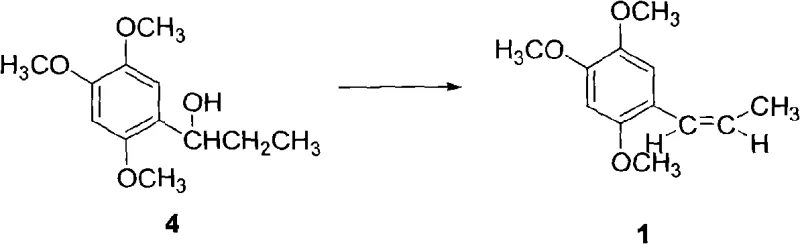
The final stage of the synthesis involves the dehydration of the alcohol intermediate to generate the target alkene, 1-propenyl-2,4,5-trimethoxybenzene. This elimination reaction is facilitated by dehydrating agents such as a combination of anhydrous sodium acetate and acetic anhydride, which are cost-effective compared to alternatives like phosphorus oxychloride or DMSO. The mixture is heated to reflux for approximately 3 hours, promoting the elimination of water to form the double bond. Post-reaction workup involves vacuum distillation to remove volatiles followed by recrystallization from solvents like n-hexane to obtain the final high-purity product. This final step ensures the stereochemical integrity of the E-isomer while avoiding the formation of carcinogenic impurities like safrole, thereby meeting stringent pharmaceutical quality standards.
How to Synthesize 1-Propenyl-2,4,5-trimethoxybenzene Efficiently
Implementing this synthesis route requires careful attention to temperature control and reagent addition rates to maximize yield and safety. The process is designed to be operationally simple, avoiding the need for exotic equipment or extreme conditions. Detailed standard operating procedures for each step, including specific solvent volumes, addition times, and crystallization protocols, are essential for reproducibility. For a comprehensive guide on executing this synthesis with optimal results, please refer to the standardized protocol below.
- Perform Friedel-Crafts acylation of 1,2,4-trimethoxybenzene with propionic anhydride or propionyl chloride using anhydrous aluminum chloride catalyst at 40-70°C to form 2,4,5-trimethoxypropiophenone.
- Reduce the resulting ketone intermediate using sodium borohydride or lithium aluminum hydride in solvents like ethyl acetate or THF at temperatures ranging from -5°C to 45°C to yield the corresponding alcohol.
- Execute dehydration of the alcohol intermediate using acetic anhydride and anhydrous sodium acetate under reflux conditions to finalize the synthesis of 1-propenyl-2,4,5-trimethoxybenzene.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented synthesis route offers substantial benefits for procurement managers and supply chain directors focused on cost optimization and reliability. The shift from Grignard chemistry to acylation/reduction significantly lowers the barrier to entry for manufacturing, as it utilizes commodity chemicals that are widely available in the global market. This reduces dependency on specialized reagent suppliers and mitigates the risk of supply disruptions associated with hazardous materials. Furthermore, the simplified workup procedures, which rely on crystallization rather than distillation, translate directly into lower energy consumption and reduced processing time, enhancing overall plant throughput.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the substitution of expensive and hazardous Grignard reagents with low-cost acylating agents like propionic anhydride and common Lewis acids. Additionally, the ability to isolate intermediates as solids eliminates the need for complex molecular distillation equipment, resulting in significant capital expenditure savings and lower maintenance costs. The high reaction yields reported in the patent examples further contribute to cost efficiency by maximizing the output per unit of raw material input, thereby reducing the effective cost of goods sold.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of robust, non-sensitive reagents that do not require specialized storage conditions such as strict inert atmospheres or refrigeration. The formation of solid intermediates at every stage facilitates easier quality control testing and inventory management, as solids are less prone to degradation during storage compared to liquid oils. This stability ensures consistent product quality over time and simplifies logistics, allowing for more flexible shipping and warehousing options without the need for hazardous material classifications associated with pyrophoric reagents.
- Scalability and Environmental Compliance: The process is inherently scalable due to its straightforward reaction conditions and minimal generation of hazardous waste, aligning with increasingly strict environmental regulations. The absence of heavy metal catalysts and the use of recyclable solvents simplify waste treatment protocols, reducing the environmental footprint of the manufacturing facility. This compliance advantage not only lowers disposal costs but also future-proofs the supply chain against tightening regulatory frameworks, ensuring long-term operational continuity for pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Asarin intermediates using this optimized pathway. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, offering clarity on performance and feasibility. Understanding these details is critical for technical teams evaluating the integration of this route into existing production portfolios.
Q: What are the primary advantages of this new Asarin synthesis route over traditional Grignard methods?
A: The new route eliminates the need for hazardous Grignard reagents and strict anhydrous ether conditions. It utilizes cheaper raw materials like propionic anhydride and produces solid intermediates that are easier to purify via crystallization rather than complex molecular distillation.
Q: Does this process avoid carcinogenic impurities like safrole?
A: Yes, the patented process is specifically designed to produce high-purity E-2,4,5-trimethoxy-1-propenylbenzene without containing carcinogenic substances such as safrole or beta-propenyl isomers, ensuring superior quality compared to extraction methods.
Q: Is this synthesis method suitable for large-scale industrial manufacturing?
A: Absolutely. The process features simple post-treatment, high reaction yields at each step, and generates minimal three wastes. The use of common solvents and the formation of solid products make it highly adaptable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Propenyl-2,4,5-trimethoxybenzene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes to maintain competitiveness in the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. By leveraging the efficiencies of the acylation-reduction-dehydration route, we can offer our partners a secure and cost-effective supply of this essential respiratory therapeutic intermediate.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis can benefit your specific project requirements. Contact us today to request a Customized Cost-Saving Analysis tailored to your volume needs. Our experts are ready to provide specific COA data and route feasibility assessments to demonstrate how we can support your supply chain goals with reliability and precision.
