Advanced Low-Temperature Isomerization for High-Purity Diphenyl Phenylphosphonate Manufacturing
The global demand for high-performance, halogen-free flame retardants has necessitated a re-evaluation of synthetic routes for aryl phosphates, specifically targeting the robust structure of diphenyl phenylphosphonate. Patent CN111635431A introduces a transformative preparation method that addresses the critical limitations of historical synthesis protocols, offering a pathway to superior purity and yield through a controlled isomerization mechanism. This technical breakthrough centers on the strategic use of bromobenzene as an initiator within a Raney nickel catalytic system, allowing the reaction to proceed efficiently at moderated temperatures between 220-260°C. By fundamentally altering the reaction kinetics and thermodynamics, this approach not only enhances the stability of the P-C bond formation but also integrates a unique impurity conversion strategy that turns potential waste streams into valuable product. For R&D directors and procurement specialists seeking reliable polymer additive supplier partnerships, this methodology represents a significant leap forward in process chemistry, ensuring consistent quality for engineering plastics and phenolic resin applications.
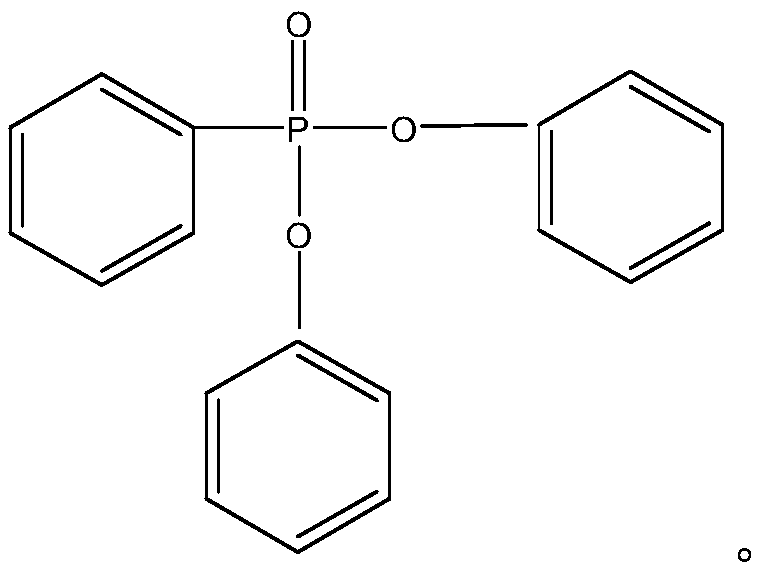
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of diphenyl phenylphosphonate has been plagued by severe thermodynamic and economic inefficiencies that hinder scalable production. Early methodologies, such as those described in U.S. Pat. No. 3,430,3807, relied heavily on benzene iodide as an initiator, which presented prohibitive costs due to the high price of iodine derivatives and resulted in disappointingly low yields that were commercially unviable. Subsequent improvements by major chemical corporations attempted to utilize benzene bromide with Raney nickel catalysts but were forced to operate at dangerously high temperatures ranging from 300-320°C to achieve acceptable conversion rates. These extreme thermal conditions introduced significant safety hazards, particularly regarding the pyrophoric nature of Raney nickel which becomes prone to ignition at such elevated temperatures, alongside the thermal degradation of triphenyl phosphite into difficult-to-remove triphenyl phosphate impurities. Furthermore, alternative transesterification routes often generated complex mixtures of ethyl phosphate impurities that required energy-intensive and costly refining processes to separate from the target molecule, creating a bottleneck for high-purity manufacturing.
The Novel Approach
The innovative protocol detailed in the patent data overcomes these legacy challenges by precisely controlling the molar ratio of triphenyl phosphite to benzene bromide between 1:0.5 and 1:2, effectively driving the isomerization equilibrium forward at much milder conditions. By maintaining the reaction temperature strictly within the 220-260°C window, the process successfully avoids the thermal oxidation pathways that generate triphenyl phosphate, thereby preserving the integrity of the phosphorus center and simplifying the downstream purification profile. A distinct advantage of this novel approach is the intentional generation of phenol as a primary byproduct during the isomerization phase, which is not treated as waste but rather as a reactive intermediate for the subsequent step. This strategic design allows for the seamless integration of a second reaction stage where phenylphosphonic dichloride is introduced to consume the phenol, chemically converting what would traditionally be an impurity into additional quantities of the desired diphenyl phenylphosphonate, thus drastically boosting overall atom economy and process yield without requiring complex separation technologies.
Mechanistic Insights into Raney Nickel Catalyzed Isomerization
The core of this synthesis lies in the rearrangement of the phosphite ester, a transformation that is kinetically sluggish under standard conditions but is dramatically accelerated by the specific interaction between the bromobenzene initiator and the nickel surface. In the first stage, the bromobenzene acts as a source of phenyl radicals or activated species that facilitate the migration of a phenyl group from the oxygen to the phosphorus atom, establishing the critical P-C bond that defines the stability of the final product. This isomerization proceeds via a concerted mechanism where the triphenyl phosphite backbone reorganizes to form diphenyl phenylphosphonate while cleaving one P-O bond to release a phenol molecule, a process that is highly sensitive to temperature and catalyst activity. The presence of Raney nickel provides the necessary active sites to lower the activation energy barrier, allowing this rearrangement to occur rapidly at 220-260°C rather than the >300°C required in uncatalyzed or less efficient systems, ensuring that the reaction mixture remains stable and free from charring or decomposition products that typically contaminate high-temperature organic syntheses.
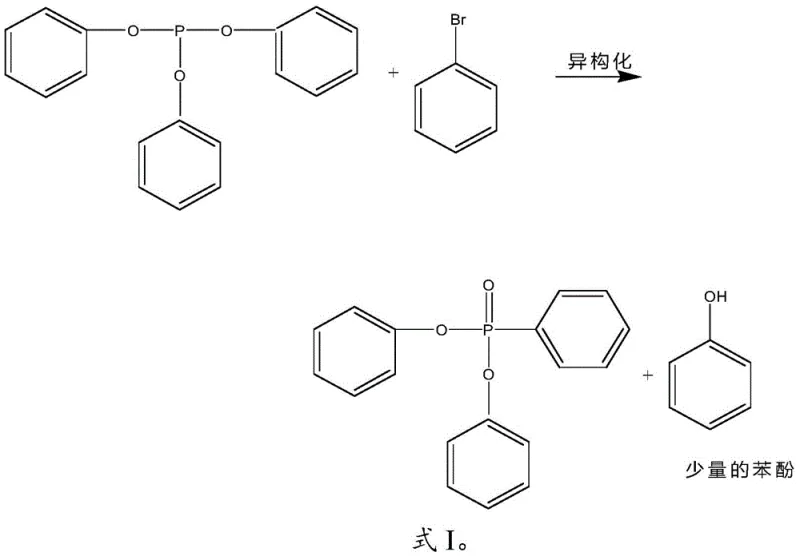
Following the initial isomerization, the process employs a sophisticated impurity management mechanism that distinguishes it from prior art, specifically targeting the phenol byproduct generated in situ. Instead of attempting to separate the phenol through energy-intensive distillation columns which often lead to product loss, the method utilizes the inherent reactivity of the hydroxyl group in phenol to participate in a secondary esterification reaction. Upon the addition of phenylphosphonic dichloride, the phenol acts as a nucleophile, attacking the electrophilic phosphorus center of the dichloride to form a new P-O-C linkage, effectively stitching the byproduct back into the target molecular architecture. This tandem reaction sequence ensures that the theoretical yield is maximized because every mole of phenol produced in the first step is theoretically capable of generating more target product in the second step, provided the stoichiometry of phenylphosphonic dichloride is carefully managed to match the phenol content. The result is a reaction profile where impurity levels are minimized not by physical separation alone, but by chemical conversion, leading to a crude product stream that is exceptionally rich in the desired diphenyl phenylphosphonate.
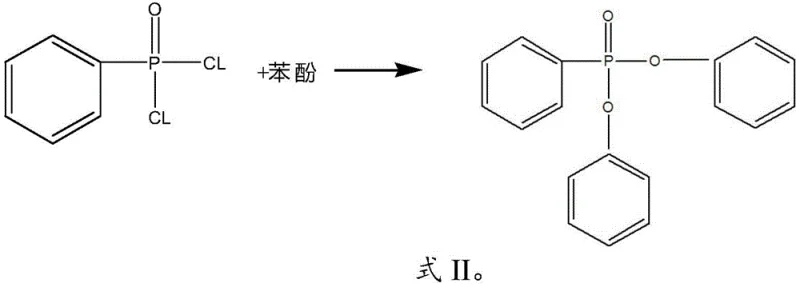
How to Synthesize Diphenyl Phenylphosphonate Efficiently
The execution of this synthesis requires precise control over reaction parameters to ensure the successful transition from isomerization to esterification without compromising product quality. The process begins with the dehydration of the Raney nickel catalyst under vacuum to prevent hydrolysis of the phosphite starting material, followed by the controlled dropwise addition of bromobenzene to manage the exotherm and maintain the optimal 220-260°C range. Once the isomerization is complete, indicated by the disappearance of triphenyl phosphite via liquid chromatography, the reaction mass undergoes reduced pressure distillation to recover excess bromobenzene for recycling, followed by filtration to remove the solid nickel catalyst for reuse. The resulting liquid filtrate, containing the isomerized product and phenol, is then heated and treated with phenylphosphonic dichloride in the presence of a Lewis acid catalyst such as magnesium chloride or titanium tetrachloride to drive the final esterification to completion, ensuring phenol levels drop below 0.5 wt% before final high-vacuum distillation yields the pure crystalline product.
- Conduct isomerization of triphenyl phosphite using bromobenzene initiator and Raney nickel catalyst at 220-260°C under nitrogen protection.
- Perform reduced pressure distillation to recover bromobenzene and solid-liquid separation to recycle the Raney nickel catalyst.
- React the separated liquid containing phenol with phenylphosphonic dichloride under Lewis acid catalysis to convert impurities into the target product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers substantial strategic benefits that extend far beyond simple yield improvements, fundamentally altering the cost structure and reliability of the supply base. The ability to operate at significantly lower temperatures reduces the energy load on reactor systems and minimizes the wear and tear on equipment, leading to extended asset life and reduced maintenance downtime which translates directly into operational cost savings. Furthermore, the closed-loop nature of the process, where both the expensive bromobenzene initiator and the heterogeneous Raney nickel catalyst are recovered and reused, drastically reduces the consumption of raw materials and lowers the volume of hazardous waste requiring disposal, aligning perfectly with modern sustainability mandates and reducing the total cost of ownership for the manufacturing facility.
- Cost Reduction in Manufacturing: The elimination of expensive iodine-based initiators and the recycling of bromobenzene create a leaner raw material profile that insulates production costs from volatile halogen market fluctuations. By converting the phenol byproduct into saleable product rather than treating it as waste, the process effectively increases the output per batch without increasing the input of the primary phosphite feedstock, delivering a profound improvement in material efficiency that drives down the unit cost of goods sold.
- Enhanced Supply Chain Reliability: The simplified purification train, which relies on straightforward distillation and filtration rather than complex chromatographic separations or recrystallizations, shortens the overall cycle time per batch and increases the throughput capacity of existing manufacturing infrastructure. This operational simplicity reduces the risk of batch failures due to purification bottlenecks, ensuring a more consistent and predictable delivery schedule for downstream customers who rely on just-in-time inventory models for their polymer compounding operations.
- Scalability and Environmental Compliance: Operating at 220-260°C instead of >300°C significantly lowers the safety risk profile associated with handling pyrophoric catalysts at extreme temperatures, making the process easier to scale from pilot plant to multi-ton commercial production without requiring exotic high-temperature alloys or specialized safety containment systems. The reduction in hazardous waste generation and the ability to recycle key reagents also streamline environmental permitting and compliance reporting, facilitating faster regulatory approval for new production lines in stringent jurisdictions.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis route, providing clarity on the operational parameters that define its success. Understanding these nuances is critical for technical teams evaluating the feasibility of technology transfer or contract manufacturing agreements, as they highlight the specific control points that ensure product consistency and safety. The answers provided are derived directly from the experimental data and process descriptions within the patent literature, offering a factual basis for decision-making.
Q: How does this process manage the phenol byproduct generated during isomerization?
A: Unlike traditional methods where phenol is a waste impurity, this novel process utilizes phenylphosphonic dichloride to react with the generated phenol in a secondary esterification step, effectively converting the byproduct back into the target diphenyl phenylphosphonate, thereby maximizing yield.
Q: What are the safety advantages of the new isomerization temperature range?
A:
Q: Can the catalyst and initiator be recycled in this manufacturing route?
A: Yes, the process is designed for circular efficiency. The solid-liquid separation step allows for the recovery and reuse of the Raney nickel catalyst, while reduced pressure distillation enables the recovery of unreacted bromobenzene, substantially lowering raw material consumption and environmental impact.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diphenyl Phenylphosphonate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate balance of temperature, catalysis, and stoichiometry required for this isomerization process is maintained with absolute precision at scale. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify the absence of triphenyl phosphate and phenol impurities, guaranteeing that every shipment meets the exacting standards required for high-performance engineering plastics and flame retardant applications.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific volume and quality requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized process can reduce your total landed costs while securing a stable supply of high-purity materials. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to validate our capabilities against your internal benchmarks and move forward with confidence in a supply relationship built on transparency and technical excellence.
