Advanced Molten Salt Catalysis for Scalable 5-Chloro-2,3-dihydro-1-indanone Manufacturing
Advanced Molten Salt Catalysis for Scalable 5-Chloro-2,3-dihydro-1-indanone Manufacturing
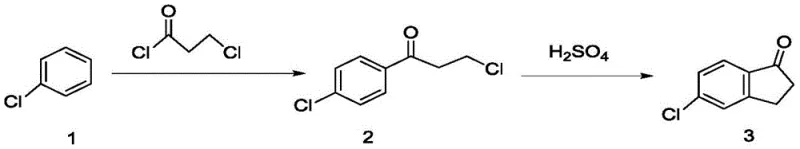
The chemical industry is constantly seeking more efficient, environmentally benign, and cost-effective pathways for synthesizing critical intermediates used in both pharmaceutical and agrochemical sectors. A significant breakthrough in this domain is detailed in patent CN109293488B, which outlines a novel preparation method for 5-chloro-2,3-dihydro-1-indanone, a pivotal building block for the pesticide indoxacarb and various anti-inflammatory medications. This technology replaces traditional, hazardous catalytic systems with a sophisticated mixed molten salt approach, fundamentally altering the economic and safety profile of the production process. By utilizing a eutectic mixture of potassium chloride, sodium chloride, and aluminum chloride, the process achieves a liquid catalytic phase at moderate temperatures, effectively solving the historical issue of aluminum chloride sublimation. This innovation not only boosts the reaction conversion rate to an impressive 98% but also elevates the final product yield to 75%, representing a substantial leap forward in process chemistry efficiency.
For global procurement teams and R&D directors, the implications of this patent extend far beyond simple yield improvements; it signals a shift towards sustainable manufacturing practices that align with modern regulatory standards. The ability to utilize mixed molten salts as both catalyst and solvent removes the necessity for massive quantities of corrosive acids like sulfuric acid or trifluoromethanesulfonic acid, which are notoriously difficult to handle and dispose of. Consequently, this method offers a reliable agrochemical intermediate supplier pathway that minimizes environmental footprint while maximizing output. As we delve deeper into the technical specifics, it becomes clear that this methodology provides a robust framework for the commercial scale-up of complex pharmaceutical intermediates, ensuring supply chain continuity and reducing the total cost of ownership for downstream manufacturers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 5-chloroindanone has been plagued by significant technical and operational challenges that hindered its widespread industrial adoption. Traditional routes, such as the one utilizing m-chlorocinnamic acid, suffer from poor selectivity during the ring-closing step, leading to the formation of unwanted isomers like 7-chloro-2,3-dihydro-1-indanone, which complicates purification and lowers overall efficiency. Another common approach involves the use of p-chlorobenzoyl chloride under high pressure with ethylene, a method that requires specialized, expensive equipment and poses inherent safety risks due to the pressurization requirements. Furthermore, the widely cited route employing concentrated sulfuric acid as a catalyst for the cyclization of 5-chloroketone is particularly problematic; it demands harsh reaction conditions, generates vast amounts of acidic waste liquid that is difficult to treat, and results in a dismal total yield of merely 36.7%. These legacy methods impose heavy burdens on waste management systems and increase the risk of equipment failure due to corrosion, making them economically unviable for large-scale production in a modern, regulated environment.
The Novel Approach
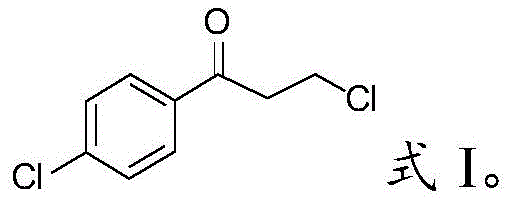
In stark contrast to these outdated methodologies, the novel approach disclosed in the patent leverages the unique properties of mixed molten salts to create a streamlined, high-efficiency synthesis route. By selecting a specific combination of potassium chloride, sodium chloride, and aluminum chloride, the inventors have created a catalytic medium that remains liquid at approximately 100°C, thereby maintaining the aluminum chloride in an active state without the losses associated with sublimation. This system allows the reaction between chlorobenzene and 3-chloropropionyl chloride to proceed smoothly at temperatures between 120-130°C, achieving a conversion rate of 98% and a product yield of 75%. This represents a dramatic improvement over the sulfuric acid method, effectively doubling the yield while simultaneously eliminating the need for excessive amounts of corrosive liquid acids. The result is a cleaner, safer, and more economically attractive process that significantly simplifies post-reaction workup and reduces the environmental impact associated with hazardous waste disposal.
Mechanistic Insights into Molten Salt-Catalyzed Cyclization
The core of this technological advancement lies in the dual role of the mixed molten salt, which functions simultaneously as a Lewis acid catalyst and a reaction solvent. In the initial stage, aluminum chloride facilitates the Friedel-Crafts acylation of chlorobenzene with 3-chloropropionyl chloride to form the intermediate 3-chloro-1-(4'-chlorophenyl)-1-propanone. The structural integrity of this intermediate is crucial for the subsequent cyclization step, and the molten salt environment provides a stable medium that prevents premature decomposition or polymerization. Unlike traditional solvents that might coordinate too strongly with the catalyst or degrade under acidic conditions, the inorganic salt melt offers a non-coordinating, high-temperature stable environment that promotes the desired electrophilic aromatic substitution. This ensures that the acyl group is directed specifically to the para-position relative to the chlorine atom on the benzene ring, setting the stage for the intramolecular cyclization that follows.
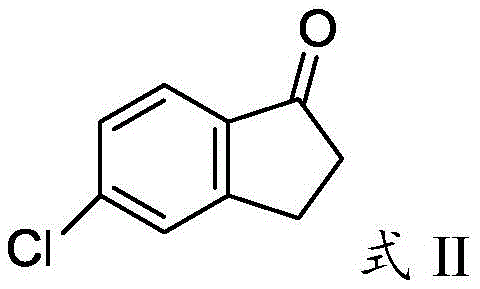
Following the formation of the intermediate ketone, the reaction mixture is subjected to elevated temperatures within the molten salt bath to induce cyclization. The mechanism involves the activation of the carbonyl group by the aluminum species within the melt, followed by an intramolecular electrophilic attack on the aromatic ring to close the five-membered indanone ring. The presence of potassium and sodium chlorides is critical here, as they modify the acidity and physical properties of the aluminum chloride, preventing it from subliming away before the reaction is complete. This precise control over the catalytic species ensures that the cyclization proceeds with high regioselectivity, minimizing the formation of byproducts and maximizing the yield of the target 5-chloro-2,3-dihydro-1-indanone. The final product, characterized by its specific bicyclic structure, is then isolated through a straightforward quenching and filtration process, yielding a high-purity solid suitable for further pharmaceutical or agrochemical synthesis.
How to Synthesize 5-Chloro-2,3-dihydro-1-indanone Efficiently
The practical implementation of this synthesis route is designed for scalability and ease of operation, making it highly attractive for contract development and manufacturing organizations (CDMOs). The process begins with the careful addition of aluminum chloride to chlorobenzene under stirring, followed by the dropwise addition of 3-chloropropionyl chloride at a controlled temperature of 30-60°C to manage the exothermic acylation reaction. Once the intermediate is formed, the entire mixture is transferred into a pre-heated vessel containing the molten salt mixture, where the temperature is raised to 120-130°C to drive the cyclization to completion. Detailed standardized synthetic steps see the guide below.
- Perform Friedel-Crafts acylation by reacting chlorobenzene with 3-chloropropionyl chloride in the presence of aluminum chloride at 30-60°C to form the intermediate ketone.
- Transfer the reaction mixture into a pre-heated mixed molten salt (KCl/NaCl/AlCl3) and maintain at 120-130°C to facilitate cyclization.
- Quench the reaction in ice water, filter the resulting brown solid, and refine via recrystallization to obtain high-purity 5-chloro-2,3-dihydro-1-indanone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this molten salt catalysis method offers profound strategic benefits that extend well beyond the laboratory bench. The elimination of concentrated sulfuric acid and trifluoromethanesulfonic acid from the process workflow removes a major bottleneck related to hazardous material handling, storage, and disposal. This shift not only enhances workplace safety for operators but also significantly reduces the regulatory burden and insurance costs associated with managing highly corrosive substances. Furthermore, the simplified workup procedure, which involves a simple ice-water quench and filtration, drastically cuts down on processing time and utility consumption, leading to substantial cost savings in the overall manufacturing budget. These operational efficiencies make the supply of this critical intermediate more resilient and less prone to disruptions caused by environmental compliance issues or waste treatment capacity limitations.
- Cost Reduction in Manufacturing: The replacement of expensive and hazardous catalysts like trifluoromethanesulfonic acid with inexpensive, commodity inorganic salts results in a direct reduction in raw material costs. Additionally, the high conversion rate of 98% ensures that expensive starting materials like chlorobenzene and 3-chloropropionyl chloride are utilized with maximum efficiency, minimizing waste and lowering the cost per kilogram of the final product. The avoidance of complex purification steps required to remove isomeric impurities further contributes to a leaner, more cost-effective production model that enhances profit margins for downstream users.
- Enhanced Supply Chain Reliability: By relying on readily available inorganic salts and common organic feedstocks, this method mitigates the risk of supply shortages often associated with specialized reagents. The robustness of the molten salt system allows for consistent batch-to-batch quality, ensuring that pharmaceutical and agrochemical manufacturers receive a steady stream of high-purity intermediates without unexpected delays. This reliability is crucial for maintaining continuous production lines for end-products like indoxacarb, where any interruption in the supply of key precursors can have cascading effects on the global market availability of the final pesticide.
- Scalability and Environmental Compliance: The mild reaction conditions and the absence of volatile organic solvents or strong mineral acids make this process inherently safer and easier to scale from pilot plant to full commercial production. The reduction in hazardous waste generation aligns perfectly with increasingly stringent global environmental regulations, allowing manufacturers to operate with a smaller environmental footprint. This sustainability advantage not only future-proofs the supply chain against tightening regulations but also enhances the brand reputation of companies adopting greener chemistry practices in their sourcing strategies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the molten salt synthesis of 5-chloro-2,3-dihydro-1-indanone, providing clarity for stakeholders evaluating this technology for integration into their supply chains. These answers are derived directly from the experimental data and technical disclosures found in the patent literature, ensuring accuracy and relevance for decision-makers. Understanding these nuances is essential for assessing the feasibility of adopting this method for large-scale manufacturing operations.
Q: What are the advantages of using mixed molten salt over concentrated sulfuric acid?
A: The use of mixed molten salt eliminates the need for large volumes of concentrated sulfuric acid, thereby drastically reducing equipment corrosion, hazardous waste generation, and post-treatment complexity while improving yield from ~36% to 75%.
Q: How does this method prevent aluminum chloride sublimation?
A: By mixing aluminum chloride with potassium chloride and sodium chloride, the melting point of the catalyst system is lowered to approximately 100°C, keeping the catalyst in a liquid state during the reaction and preventing loss via sublimation.
Q: What purity levels can be achieved with this synthesis route?
A: Following the refinement step described in the patent, the final 5-chloro-2,3-dihydro-1-indanone product achieves a purity of greater than 99% as determined by high-performance liquid chromatography (HPLC).
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Chloro-2,3-dihydro-1-indanone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and sustainable synthesis routes for high-value intermediates like 5-chloro-2,3-dihydro-1-indanone. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the molten salt catalysis method are fully realized in a practical, industrial setting. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify every batch. Our capability to adapt and optimize complex chemical processes allows us to offer a reliable pharma intermediates supplier partnership that guarantees both quality and consistency.
We invite potential partners to engage with our technical procurement team to discuss how this innovative manufacturing route can be tailored to your specific volume and quality requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this greener synthesis method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is optimized for the future of fine chemical manufacturing.
