Advanced Anhydrous Synthesis of Fosfomycin Trometamol for Global Pharmaceutical Supply Chains
The pharmaceutical industry continuously seeks robust manufacturing routes for critical antibiotics, and the preparation of Fosfomycin Trometamol (CAS 78964-85-9) stands as a prime example of process innovation driving commercial viability. As detailed in patent CN109608497B, a novel preparation method has been developed that fundamentally addresses the stability and purity challenges inherent in previous synthetic routes. This technology leverages a sophisticated anhydrous salt exchange mechanism, utilizing acetic anhydride to meticulously control the reaction environment, thereby preventing the degradation of the sensitive epoxide moiety. For R&D directors and procurement specialists, this represents a significant leap forward, offering a pathway to high-purity API intermediates with yields exceeding 95%, while simultaneously mitigating the environmental and safety risks associated with traditional aqueous or toxic reagent-based processes. The structural integrity of the final product is paramount for its efficacy in treating urogenital infections, and this patented approach ensures that the molecular architecture remains intact throughout the synthesis.
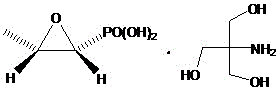
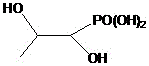
The limitations of conventional methods for synthesizing Fosfomycin Trometamol are well-documented and pose substantial risks to both product quality and operational safety. Historically, processes such as those disclosed in EP 2845859A1 relied on highly toxic reagents like isocyanates and ketenes, which require stringent low-temperature storage and present explosion hazards, creating a nightmare for supply chain continuity and labor protection. Furthermore, aqueous solution methods, while seemingly benign, suffer from a critical chemical flaw: the instability of the fosfomycin molecule in water leads to rapid ring-opening hydrolysis. As illustrated in the degradation pathway, the epoxide ring opens to form a diol impurity, which accumulates rapidly—reaching over 3% within just one hour at room temperature, far exceeding the pharmacopoeia limit of 0.3%. Other methods involving strong acids for pH neutralization often fail to precisely control the endpoint, resulting in products with broad melting ranges and significant impurity profiles that necessitate costly and yield-reducing refining steps.
The novel approach presented in CN109608497B offers a definitive solution to these longstanding industry pain points by shifting the paradigm to a mild, anhydrous organic solvent system. Instead of battling water-induced hydrolysis, this method employs absolute methanol and ethanol coupled with acetic anhydride, a reagent that effectively scavenges trace moisture and maintains the integrity of the epoxide ring. The reaction proceeds at a remarkably mild temperature range of 30-35°C, eliminating the need for energy-intensive cryogenic conditions or complex ion-exchange columns. By reacting the fosfomycin precursor with tromethamine in absolute ethanol, followed by the critical acetic anhydride treatment, the process achieves a pristine white solid with a sharp melting point of 122.6-123.1°C. This indicates a highly crystalline and pure product, directly addressing the purity concerns of R&D teams while simplifying the downstream processing requirements for manufacturing facilities.
Mechanistic Insights into Anhydrous Salt Exchange and Epoxide Stabilization
To fully appreciate the technical superiority of this synthesis, one must delve into the mechanistic interplay between the reactants and the solvent system. The core of the innovation lies in the prevention of nucleophilic attack on the strained three-membered epoxide ring of the fosfomycin molecule. In traditional aqueous environments, water molecules act as nucleophiles, attacking the electrophilic carbons of the epoxide to form the inactive diol byproduct. In this patented process, the use of acetic anhydride serves a dual purpose: it acts as a chemical desiccant to remove any trace water introduced during the salt exchange, and it potentially acetylates transient hydroxyl species that could catalyze degradation. The reaction begins with the dissolution of tromethamine in absolute ethanol, creating a basic environment conducive to salt formation with the levofosfomycin dextro-ammoniate precursor. This initial step is crucial for establishing the correct stoichiometry before the introduction of the stabilizing agent.
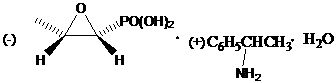
Following the initial salt formation, the addition of acetic anhydride in methanol triggers the stabilization phase. The exothermic nature of this reaction is carefully managed by controlling the temperature rise to no more than 30-35°C, ensuring that thermal energy does not overcome the activation barrier for epoxide ring opening. The molar ratio of fosfomycin trometamol to acetic anhydride is tightly controlled between 1:1.01 and 1:1.10, providing a slight excess of the anhydride to guarantee complete moisture scavenging without introducing excessive acetylation byproducts. This precise control allows for the isolation of the target molecule with exceptional fidelity. Furthermore, the process facilitates the recovery of the chiral auxiliary, dextro phenethylamine, from the filtrate. Since this amine does not participate chemically in the final stabilization step, it remains available for recycling, which is a massive advantage for atom economy and cost efficiency in large-scale operations.
How to Synthesize Fosfomycin Trometamol Efficiently
The operational protocol for this synthesis is designed for seamless integration into existing fine chemical manufacturing infrastructure, requiring only standard reactor setups capable of handling organic solvents and mild heating. The process begins with the preparation of the intermediate salt by refluxing tromethamine and the fosfomycin precursor in absolute ethanol, followed by cooling to induce crystallization. The critical second stage involves the suspension of this intermediate in methanol and the dropwise addition of acetic anhydride under strict temperature monitoring. After the reaction is complete, the solvent is removed via reduced-pressure rotary evaporation, and the product is precipitated by the addition of absolute ethanol at 0°C. This straightforward sequence eliminates the need for specialized equipment like low-temperature ion exchange columns, making it accessible for a wide range of reliable pharmaceutical intermediate suppliers looking to upgrade their production capabilities.
- Dissolve tromethamine in absolute ethanol and react with levofosfomycin dextro-ammoniate under reflux to form the intermediate salt.
- Treat the intermediate with acetic anhydride in methanol at mild temperatures (30-35°C) to scavenge moisture and prevent ring-opening.
- Crystallize the final product by adding absolute ethanol, cooling to 0°C, and recovering the valuable chiral amine from the filtrate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel manufacturing route offers profound strategic benefits that extend far beyond simple yield improvements. The elimination of hazardous reagents such as isocyanates and ketenes drastically reduces the regulatory burden and insurance costs associated with storing and transporting dangerous chemicals. By utilizing common, low-cost industrial solvents like methanol and ethanol, along with inexpensive acetic anhydride, the raw material cost profile is significantly optimized compared to legacy processes that rely on exotic or unstable reagents. Moreover, the ability to recover and recycle the expensive dextro phenethylamine auxiliary directly impacts the bottom line, turning what was once a consumable cost into a reusable asset. This circular approach to raw material usage not only lowers the cost of goods sold but also insulates the supply chain from volatility in the pricing of chiral amines.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the substitution of high-cost, high-risk reagents with commodity chemicals. By avoiding the need for cryogenic storage and specialized safety containment for toxic isocyanates, capital expenditure on facility upgrades is minimized. Additionally, the high yield of over 95% means that less raw material is required to produce the same amount of API, effectively stretching the purchasing budget. The recovery of the chiral amine further compounds these savings, as the most expensive component of the starting material loop is retained within the system rather than being lost to waste streams.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the availability of niche reagents or the shutdown of facilities due to safety incidents. This method relies on widely available bulk chemicals that are unlikely to face supply shortages. The mild reaction conditions (30-35°C) reduce the risk of thermal runaways or equipment failure, ensuring consistent batch-to-batch production schedules. For global buyers, this translates to a more dependable source of high-purity Fosfomycin Trometamol, reducing the risk of drug shortages caused by manufacturing delays or quality failures at the supplier level.
- Scalability and Environmental Compliance: From an environmental perspective, the reduction in "three wastes" (waste water, waste gas, waste residue) is a critical advantage. The anhydrous nature of the reaction minimizes wastewater generation, and the absence of toxic byproducts simplifies effluent treatment. This aligns perfectly with increasingly stringent global environmental regulations, allowing manufacturers to scale up production without facing prohibitive disposal costs or regulatory hurdles. The simplicity of the work-up procedure, involving basic filtration and drying, ensures that the process can be scaled from pilot plants to multi-ton commercial reactors with minimal engineering friction.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is essential for stakeholders evaluating its adoption for commercial production. The following questions address common inquiries regarding the stability, purity, and scalability of the process described in CN109608497B. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering a clear picture of why this method outperforms historical alternatives in both technical and economic metrics.
Q: How does the new process prevent the formation of diol impurities?
A: The process utilizes strictly anhydrous conditions and acetic anhydride, which acts as a dehydrating agent. This prevents the hydrolysis of the sensitive epoxide ring in the fosfomycin molecule, keeping diol content well below the pharmacopoeia limit of 0.3%.
Q: What are the cost advantages regarding raw materials?
A: Unlike prior art requiring toxic isocyanates or expensive low-temperature storage, this method uses low-cost acetic anhydride. Furthermore, the expensive dextro phenethylamine auxiliary can be quantitatively recovered and recycled from the mother liquor.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the reaction occurs at mild temperatures (30-35°C) without the need for complex low-temperature ion exchange columns. The simple operation and high yield (over 95%) make it highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fosfomycin Trometamol Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent to production requires a partner with deep technical expertise and a commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this anhydrous synthesis are fully realized in a GMP-compliant environment. We understand that for antibiotics like Fosfomycin Trometamol, stringent purity specifications are non-negotiable, which is why our rigorous QC labs employ advanced analytical techniques to verify that diol impurities remain well below the 0.3% threshold. Our infrastructure is designed to handle the specific solvent recovery and amine recycling loops required by this process, maximizing efficiency and minimizing environmental impact for our clients.
We invite global pharmaceutical partners to engage with our technical procurement team to discuss how this advanced manufacturing route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the potential reductions in raw material and waste disposal costs specific to your volume requirements. We encourage you to contact us today to obtain specific COA data from our recent batches and to receive comprehensive route feasibility assessments tailored to your project timelines. Let us demonstrate how our commitment to innovation and quality makes us the preferred choice for your critical API intermediate needs.
