Advanced Catalytic Decarboxylation for Scalable Biphenyl Carboxylic Acid Production
Advanced Catalytic Decarboxylation for Scalable Biphenyl Carboxylic Acid Production
The pharmaceutical industry continuously seeks robust synthetic routes for critical cardiovascular intermediates, specifically targeting the efficient production of 4'-halogenalkyl-biphenyl-2-carboxylic acids. Patent CN102190581A introduces a groundbreaking methodology that addresses long-standing inefficiencies in synthesizing these vital structures, such as the active constituent Xenalipin. This innovation leverages a sophisticated palladium and copper co-catalyzed decarboxylative cross-coupling strategy, fundamentally shifting the paradigm from traditional stoichiometric metal usage to a more sustainable catalytic cycle. By utilizing readily available phthalic anhydride derivatives and simple aryl halides, this process bypasses the need for hazardous organometallic reagents and complex protecting group strategies. The result is a streamlined pathway that offers superior atom economy and operational simplicity, positioning it as a preferred choice for modern API manufacturing. 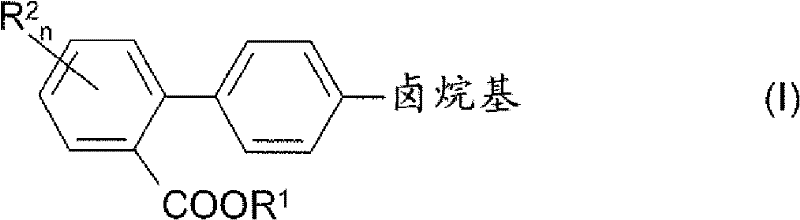
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4'-trifluoromethyl-biphenyl-2-carboxylic acid has been plagued by significant technical and economic hurdles inherent to classical cross-coupling methodologies. Traditional Suzuki-Miyaura approaches necessitate the preparation of arylboronic acids, which are notoriously unstable, moisture-sensitive, and often require cryogenic conditions for their synthesis from Grignard or organolithium precursors. Furthermore, the presence of free carboxylic acid functionalities typically demands additional protection and deprotection steps, drastically increasing the step count and reducing overall throughput. Alternative methods involving stoichiometric nickel coupling or Negishi reactions suffer from poor atom economy due to the requirement for equivalent amounts of expensive metals and generate substantial quantities of toxic metal waste. These legacy processes also face severe safety challenges in large-scale operations due to the handling of pyrophoric reagents and the generation of exothermic byproducts, making them economically unviable for cost-sensitive commercial production.
The Novel Approach
The novel approach detailed in the patent data revolutionizes this landscape by employing a direct decarboxylative cross-coupling of phthalate salts with aryl halides. This method elegantly circumvents the need for pre-formed organoboron or organozinc species, utilizing instead stable and inexpensive phthalic anhydride derivatives as the nucleophilic partner. The reaction proceeds through a concerted mechanism where the carboxylate group serves as a leaving group, driven by the thermodynamic stability of carbon dioxide evolution. This eliminates the necessity for protecting the carboxylic acid moiety during the coupling event, thereby collapsing multiple synthetic steps into a single transformative operation. The versatility of this system allows for the introduction of various halogenalkyl groups, including trifluoromethyl and pentafluoroethyl substituents, which are critical for modulating the metabolic stability and lipophilicity of the final drug candidate. 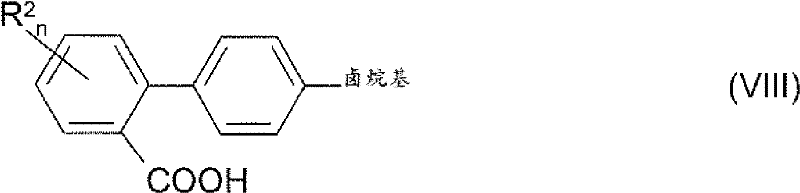
Mechanistic Insights into Pd/Cu Catalyzed Decarboxylative Coupling
The core of this technological breakthrough lies in the synergistic interaction between palladium and copper catalytic cycles, facilitated by specialized ligand systems. The reaction initiates with the oxidative addition of the aryl halide to the palladium center, forming a key aryl-palladium-halide intermediate. Concurrently, the copper source, often a copper(I) or copper(II) salt, coordinates with the phthalate carboxylate, promoting the decarboxylation event to generate an aryl-copper species. This transmetallation step is critical, as it transfers the aryl group from copper to palladium, setting the stage for reductive elimination. The choice of ligand is paramount; bulky, electron-rich phosphines such as 2-(di-t-butylphosphino)biphenyl (JohnPhos) or dicyclohexylphosphino-biphenyl derivatives stabilize the active palladium species and accelerate the reductive elimination step to release the biaryl product. 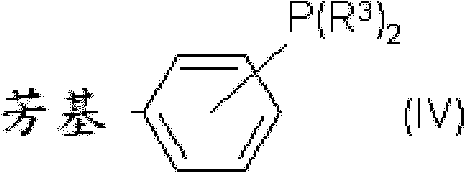
Impurity control in this system is achieved through the precise tuning of the catalyst loading and the selection of cyclic chelating amines like 1,10-phenanthroline. These additives help suppress homocoupling side reactions, which are common pitfalls in copper-mediated processes, ensuring a high purity profile for the resulting biphenyl ester. The use of polar aprotic solvents such as N-methylpyrrolidone (NMP) in combination with high-boiling hydrocarbons like mesitylene provides an optimal thermal environment for the reaction, typically running between 160°C and 210°C. This thermal robustness allows for the complete conversion of starting materials while minimizing the formation of脱卤 byproducts. The mechanistic elegance ensures that the carboxylate directing group is cleanly expelled as gas, simplifying downstream purification and reducing the burden on waste treatment facilities.
How to Synthesize 4'-Trifluoromethyl-Biphenyl-2-Carboxylic Acid Efficiently
The practical execution of this synthesis begins with the preparation of the mono-ester potassium salt of phthalic acid, which serves as the stable nucleophilic precursor. This salt is then subjected to the cross-coupling conditions with 4-chlorobenzotrifluoride in the presence of the Pd/Cu catalyst system. The reaction mixture is heated under inert atmosphere to facilitate the decarboxylative coupling, yielding the corresponding biphenyl ester in high conversion. Following the coupling, a straightforward saponification step converts the ester into the final free acid, completing the sequence. The detailed standardized synthesis steps for this process are outlined in the guide below.
- Prepare the mono-ester potassium salt of phthalic acid by reacting phthalic anhydride with an alcohol and base.
- Perform decarboxylative cross-coupling between the phthalate salt and an aryl halide using a Pd/Cu catalyst system with a phosphine ligand.
- Hydrolyze the resulting biphenyl ester using alkali to obtain the final 4'-halogenalkyl-biphenyl-2-carboxylic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this decarboxylative coupling technology offers profound strategic advantages in terms of cost structure and supply reliability. By eliminating the dependency on volatile and expensive boronic acid building blocks, manufacturers can significantly reduce raw material costs and mitigate the risks associated with the supply chain instability of specialized organometallic reagents. The ability to utilize commodity chemicals like phthalic anhydride and simple aryl halides ensures a robust and diversified sourcing strategy, shielding production schedules from market fluctuations. Furthermore, the reduction in synthetic steps directly translates to lower operational expenditures, as fewer unit operations mean reduced energy consumption, solvent usage, and labor requirements. This efficiency gain is critical for maintaining competitiveness in the generic pharmaceutical market where margin pressure is intense.
- Cost Reduction in Manufacturing: The elimination of stoichiometric metal reagents and protecting group chemistry drastically simplifies the mass balance of the process. By avoiding the purchase of costly boronic acids and the reagents required for their protection, the overall cost of goods sold is substantially lowered. The catalytic nature of the palladium and copper system means that expensive metals are used in minute quantities, further enhancing the economic viability of the route. Additionally, the simplified workup procedures reduce the consumption of extraction solvents and chromatography media, leading to significant savings in waste disposal and material costs.
- Enhanced Supply Chain Reliability: Relying on stable, shelf-stable starting materials like phthalic anhydride salts removes the logistical challenges associated with transporting moisture-sensitive intermediates. This stability allows for bulk purchasing and long-term storage without degradation, ensuring continuous production capability even during supply disruptions. The robustness of the reaction conditions also means that the process is less susceptible to variations in raw material quality, providing a consistent output that meets stringent quality specifications. This reliability is essential for securing long-term contracts with major pharmaceutical partners who demand uninterrupted supply.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, with the potential for one-pot operations that minimize intermediate isolation and handling. This reduces the physical footprint required for manufacturing and lowers the risk of exposure to hazardous materials for plant personnel. From an environmental perspective, the generation of carbon dioxide as the primary byproduct is far preferable to the heavy metal waste streams associated with stoichiometric nickel or zinc processes. This aligns with modern green chemistry principles and facilitates easier regulatory approval for commercial manufacturing sites.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthetic route. These answers are derived directly from the experimental data and comparative analysis provided in the patent literature, offering clarity on the process capabilities. Understanding these details is crucial for technical teams evaluating the feasibility of technology transfer and for commercial teams assessing the value proposition.
Q: Why is this decarboxylative coupling preferred over Suzuki-Miyaura reactions?
A: Unlike Suzuki coupling which requires unstable boronic acids and protection of carboxylic acid groups, this method uses stable phthalate salts directly, eliminating complex protection/deprotection steps and improving safety.
Q: What are the safety advantages regarding organometallic reagents?
A: This process avoids the use of highly reactive and pyrophoric organolithium or Grignard reagents required in Negishi or Wurtz-Grignard pathways, significantly reducing industrial safety risks.
Q: Can this process be scaled for commercial production?
A: Yes, the patent demonstrates a one-pot procedure that minimizes isolation steps and utilizes robust catalyst systems, making it highly suitable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4'-Trifluoromethyl-Biphenyl-2-Carboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthetic routes in the development of life-saving cardiovascular medications. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our expertise in catalytic cross-coupling technologies allows us to optimize this specific decarboxylative process for maximum yield and minimal impurity formation, guaranteeing a supply chain that is both resilient and cost-effective.
We invite you to collaborate with us to leverage this advanced technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing excellence can drive value for your organization.
