Advanced Iodine-Catalyzed Synthesis Of 2-Phosphonomethyl Phenol Compounds For Commercial Scale-Up
The landscape of organophosphorus chemistry is undergoing a significant transformation driven by the demand for efficient, green, and cost-effective synthetic methodologies. Patent CN116237068A introduces a groundbreaking preparation method for 2-phosphonomethyl phenol compounds, a class of molecules critical for developing prostate phosphatase inhibitors, calcium ion antagonists, and antimicrobial agents. This innovation leverages a dual-catalyst system comprising elemental iodine and an alkaline reagent to construct benzylic carbon-phosphorus bonds with exceptional efficiency. By utilizing simple and easily obtained salicyl alcohol compounds alongside diaryl phosphine oxides, this technology addresses long-standing challenges in atom economy and reaction safety. For global procurement teams and R&D directors, this patent represents a pivotal shift towards more sustainable manufacturing processes that do not compromise on yield or purity standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of carbon-phosphorus bonds has relied heavily on classical reactions such as the Arbuzov reaction or transition metal-catalyzed couplings, which often suffer from significant drawbacks in an industrial setting. Traditional methods frequently require harsh reaction conditions, including high temperatures and pressures, which pose safety risks and increase energy consumption. Furthermore, many established protocols depend on expensive noble metal catalysts, such as barium triflimide complexes, which can cost upwards of 1900 yuan per gram, severely impacting the overall cost of goods sold. These metal-catalyzed routes are also sensitive to the electronic properties of substrates, often resulting in poor yields when applied to simple benzyl or allyl alcohols compared to specific structures like chalcone alcohols. The reliance on heavy metals also introduces complex downstream purification requirements to meet stringent residual metal specifications for pharmaceutical applications, adding time and cost to the production cycle.
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent utilizes a metal-free catalytic system that dramatically simplifies the synthetic workflow while enhancing economic viability. By employing elemental iodine and a base such as potassium tert-butoxide, the reaction proceeds smoothly under mild thermal conditions, typically between 80°C and 100°C. This approach exhibits remarkable substrate tolerance, successfully coupling various diaryl phosphine oxides with salicyl alcohol derivatives regardless of electron-donating or electron-withdrawing substituents. The process achieves high atom economy by directly dehydrating the alcohol and phosphine oxide without generating excessive waste streams. As illustrated in the general reaction scheme below, the transformation is direct and efficient, converting readily available starting materials into high-value intermediates with minimal operational complexity.
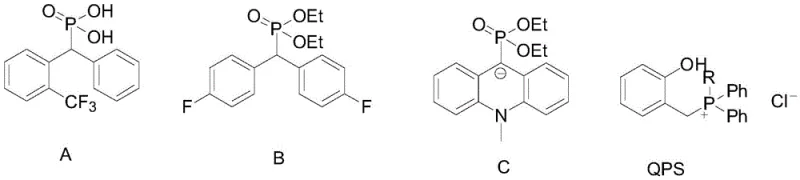
The versatility of this new route is further evidenced by its compatibility with diverse functional groups, including halogens, alkoxy groups, and heterocycles like thiophene. This broad scope ensures that a single standardized protocol can be adapted for the synthesis of a wide library of analogues, streamlining the development pipeline for new drug candidates. The elimination of transition metals not only reduces raw material costs but also simplifies the regulatory burden associated with heavy metal clearance, making it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Iodine-Catalyzed C-P Bond Formation
Understanding the mechanistic pathway is crucial for R&D teams aiming to optimize this process for large-scale manufacturing. The reaction initiates with the activation of the salicyl alcohol by elemental iodine, forming an iodinated intermediate and releasing hydrogen iodide. This intermediate subsequently undergoes dehydration in the presence of HI to regenerate elemental iodine, effectively creating a catalytic cycle while generating a highly reactive ortho-quinone methide species. This electrophilic intermediate is the key driver of the reaction, poised for nucleophilic attack. Simultaneously, the diaryl phosphine oxide is deprotonated by the alkaline reagent to form a nucleophilic diaryl phosphine anion. The convergence of these two activated species leads to the formation of the carbon-phosphorus bond, yielding the 2-phosphonomethyl phenol anion, which is finally protonated to afford the target product.
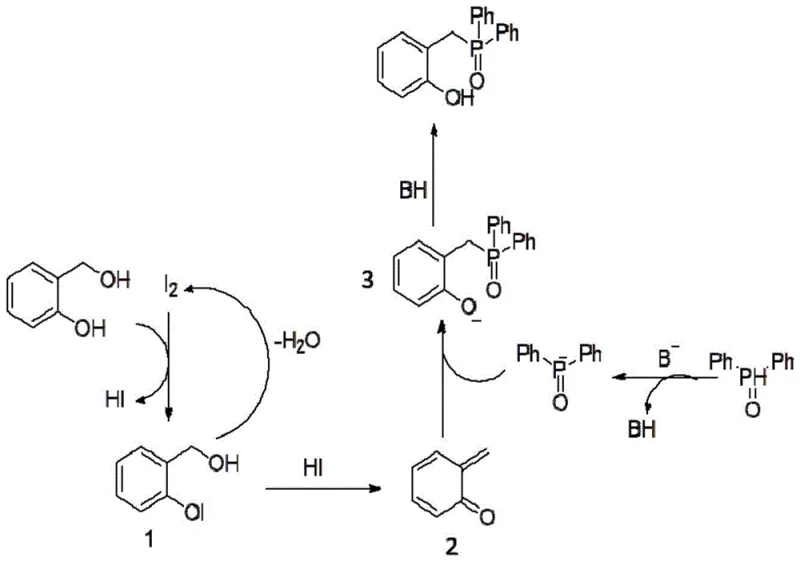
This mechanistic understanding highlights the elegance of the iodine-mediated activation, which avoids the need for external oxidants or harsh dehydrating agents. The regeneration of iodine ensures that only catalytic amounts are required, although the patent specifies a molar ratio of 0.9 to 1.1 relative to the alcohol to drive the equilibrium effectively. Impurity control is inherently managed by the specificity of the ortho-quinone methide formation, which minimizes side reactions common in radical-based or metal-catalyzed pathways. The use of anhydrous solvents like 1,4-dioxane or dichloromethane further stabilizes the reactive intermediates, ensuring consistent batch-to-batch reproducibility. For quality control laboratories, this predictable mechanism translates into a cleaner impurity profile, facilitating easier purification via standard column chromatography using ethyl acetate and petroleum ether mixtures.
How to Synthesize 2-Phosphonomethyl Phenol Efficiently
Implementing this synthesis route requires careful attention to anhydrous conditions and stoichiometric precision to maximize yield and purity. The process is divided into two primary stages: the preparation of the diaryl phosphine oxide precursor and the subsequent coupling reaction. The precursor is typically synthesized by reacting an aryl Grignard reagent with diethyl phosphonate, followed by acid quenching and purification. The core coupling step involves mixing the salicyl alcohol, the prepared phosphine oxide, potassium tert-butoxide, and iodine in a dry solvent under an inert atmosphere. Detailed standardized operating procedures for this synthesis, including specific workup and purification protocols, are outlined in the guide below.
- Prepare diaryl phosphine oxide by reacting aryl Grignard reagents with diethyl phosphonate in anhydrous conditions, followed by acid quenching and purification.
- Mix salicyl alcohol, diaryl phosphine oxide, potassium tert-butoxide, and elemental iodine in anhydrous 1,4-dioxane or dichloromethane under nitrogen protection.
- Heat the reaction mixture to 80-100°C for 4-10 hours, then extract with water and dichloromethane, dry, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, this technology offers substantial benefits by decoupling production from volatile precious metal markets and complex logistics. The reliance on commodity chemicals like iodine and potassium tert-butoxide ensures a stable and resilient supply chain, mitigating risks associated with the sourcing of specialized catalysts. The mild reaction conditions reduce energy consumption and allow for the use of standard glass-lined or stainless steel reactors, lowering capital expenditure requirements for facility upgrades. Furthermore, the high yields reported in the patent examples, often exceeding 70% over two steps, contribute to a more efficient use of raw materials, directly impacting the bottom line through reduced waste disposal costs and improved material throughput.
- Cost Reduction in Manufacturing: The elimination of expensive barium or other noble metal catalysts results in a drastic reduction in raw material costs per kilogram of product. By avoiding the need for specialized metal scavengers and extensive purification steps to remove trace metals, the overall processing time and associated labor costs are significantly lowered. The high atom economy of the reaction ensures that a greater proportion of input materials are converted into the final product, minimizing waste generation and disposal fees. This economic efficiency makes the process highly competitive for the production of high-purity pharmaceutical intermediates where margin pressure is intense.
- Enhanced Supply Chain Reliability: The use of widely available starting materials such as salicyl alcohol and aryl bromides ensures that production schedules are not disrupted by shortages of exotic reagents. The robustness of the reaction against variations in substrate electronics means that suppliers can maintain consistent output even when switching between different grades of raw materials. This flexibility allows for better inventory management and faster response times to fluctuating market demands. Additionally, the simplified workflow reduces the number of unit operations, decreasing the potential for bottlenecks and enhancing overall supply continuity for critical drug substances.
- Scalability and Environmental Compliance: The absence of heavy metals simplifies environmental compliance and waste treatment, aligning with increasingly stringent global regulations on chemical manufacturing. The reaction can be safely scaled from laboratory benchtop quantities to multi-ton commercial production without encountering the heat transfer or mixing issues often associated with exothermic metal-catalyzed reactions. The use of common organic solvents that can be recovered and recycled further enhances the sustainability profile of the process. This green chemistry approach not only reduces the environmental footprint but also appeals to end-users seeking suppliers with strong ESG credentials.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this iodine-catalyzed synthesis method. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details is essential for evaluating the feasibility of adopting this technology for your specific production needs.
Q: What are the key advantages of the iodine-catalyzed method over traditional metal-catalyzed routes?
A: The iodine-catalyzed method eliminates the need for expensive noble metal catalysts like barium triflimide, significantly reducing raw material costs. It also operates under milder conditions (80-100°C) compared to harsh traditional methods, improving safety and environmental compliance while maintaining high atom economy.
Q: Is this synthesis method scalable for industrial production of pharmaceutical intermediates?
A: Yes, the process uses readily available raw materials such as salicyl alcohol and diaryl phosphine oxides. The reaction conditions are safe and mild, utilizing common solvents like 1,4-dioxane, which facilitates easy scale-up from laboratory to commercial tonnage without complex equipment requirements.
Q: What is the substrate scope for the aryl groups in the phosphine oxide component?
A: The method demonstrates strong universality, successfully accommodating various substituted aryl groups including thienyl, 3-methoxyphenyl, p-trifluoromethylphenyl, and unsubstituted phenyl groups. Electronic effects of substituents do not significantly impact the yield, ensuring consistent quality across different derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Phosphonomethyl Phenol Supplier
NINGBO INNO PHARMCHEM stands at the forefront of adopting innovative synthetic technologies to deliver superior chemical solutions to the global market. Our technical team has extensively evaluated the iodine-catalyzed route described in CN116237068A and confirmed its potential for robust commercial manufacturing. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and reliability. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 2-phosphonomethyl phenol meets the highest industry standards for pharmaceutical and agrochemical applications.
We invite you to collaborate with us to leverage this advanced synthesis method for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized processes can enhance your supply chain efficiency and reduce overall manufacturing costs.
