Revolutionizing Cinacalcet Production: A Scalable Two-Step Synthetic Strategy for High-Purity Intermediates
Revolutionizing Cinacalcet Production: A Scalable Two-Step Synthetic Strategy for High-Purity Intermediates
The pharmaceutical landscape for calcimimetics continues to evolve, driven by the critical need for safer, more efficient manufacturing processes for active pharmaceutical ingredients (APIs). A pivotal advancement in this domain is detailed in patent CN109535006B, which introduces a novel intermediate compound and a streamlined preparation method for Cinacalcet Hydrochloride. This breakthrough addresses long-standing challenges in the synthesis of this vital medication, offering a pathway that significantly enhances both chemical efficiency and operational safety. As a leading entity in fine chemical manufacturing, understanding the nuances of this technology is essential for stakeholders aiming to optimize their supply chains and reduce production costs. The core of this innovation lies in the identification and utilization of a specific halogenated amine intermediate, which serves as a superior precursor compared to traditional aldehyde or alcohol-based starting materials.
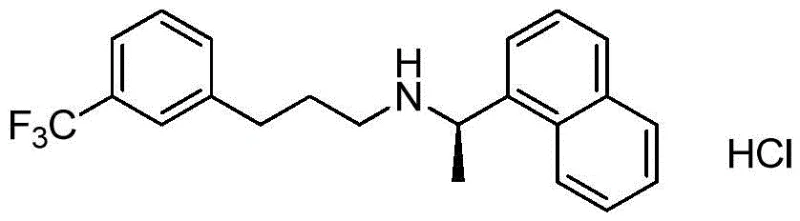
Cinacalcet Hydrochloride, chemically known as N-((1R)-1-(1-naphthyl)ethyl)-3-(3-(trifluoromethyl)phenyl)propane-1-amine hydrochloride, is a cornerstone therapy for secondary hyperparathyroidism. The molecular architecture, featuring a chiral naphthyl-ethyl amine linkage to a trifluoromethyl-substituted phenylpropyl chain, presents specific synthetic hurdles. Traditional approaches have often struggled with impurity profiles and the use of hazardous reagents. The new methodology described in the patent circumvents these issues by decoupling the formation of the amine backbone from the introduction of the aromatic trifluoromethyl group, allowing for tighter control over stereochemistry and purity at each stage of the synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of Cinacalcet Hydrochloride has relied on several distinct synthetic strategies, each fraught with significant drawbacks that hinder cost-effective and safe mass production. One prevalent method involves a direct reductive amination between m-trifluoromethyl phenylpropyl aldehyde and R-1-(1-naphthyl)ethylamine. While conceptually straightforward, this route typically employs titanium tetraisopropoxide as a catalyst and sodium cyanoborohydride as a reducing agent. The reliance on titanium species introduces complications regarding the removal of metal residues, which can be costly and technically demanding to achieve the stringent purity standards required for APIs. Furthermore, sodium cyanoborohydride is notoriously toxic, posing severe health risks to operators and creating complex waste disposal challenges.
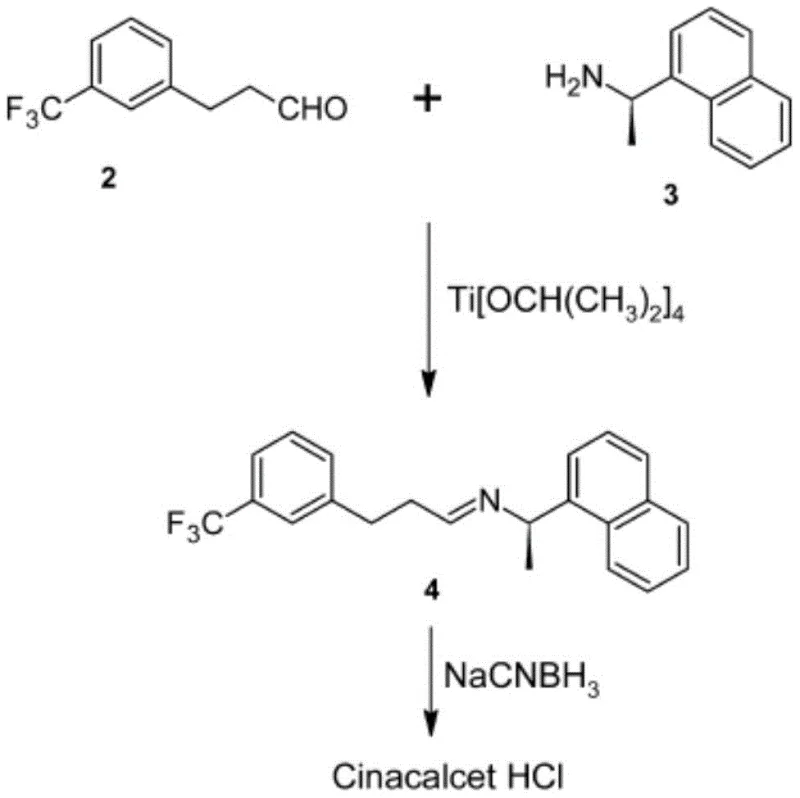
Alternative pathways have attempted to construct the carbon skeleton via Heck coupling reactions or by converting propanol derivatives into leaving groups for nucleophilic substitution. However, these methods often involve carcinogenic reagents like ethyl acrylate or require harsh conditions that lead to unstable intermediates. Another category of prior art focuses on reducing amide precursors using powerful reducing agents such as borane-THF, lithium aluminum hydride, or Red-Al. These reagents are not only expensive but also present substantial safety hazards; for instance, lithium aluminum hydride is pyrophoric and requires strictly anhydrous conditions, while borane complexes can release toxic gases during workup. The cumulative effect of these limitations is a manufacturing process that is fragile, expensive, and difficult to scale without compromising safety or environmental compliance.
The Novel Approach
In stark contrast to the convoluted and hazardous legacy methods, the novel approach disclosed in CN109535006B offers a remarkably concise and robust solution. This strategy centers on the synthesis of a key intermediate, (R)-3-bromo-N-(1-(naphthalen-1-yl)ethyl)propan-1-amine, depicted in Formula (I). By establishing the chiral amine linkage first through a mild reductive amination of 3-halopropionaldehyde and (R)-1-naphthylethylamine, the process creates a stable platform for the subsequent introduction of the trifluoromethyl phenyl group. The second step utilizes a classic Friedel-Crafts alkylation, employing anhydrous aluminum trichloride to couple the intermediate with benzotrifluoride. This shift in strategy is transformative, replacing exotic and dangerous reducing agents with standard, well-understood Lewis acid chemistry.

The elegance of this new route lies in its modularity and safety profile. The initial formation of the bromo-amine intermediate proceeds under mild conditions in methanol, avoiding the extreme temperatures and pressures associated with other methods. The subsequent alkylation step is highly selective, minimizing the formation of regio-isomers and other byproducts that plague direct coupling methods. By isolating the intermediate, manufacturers gain a critical quality control checkpoint, ensuring that only material of the highest specification proceeds to the final step. This results in a final API with exceptional purity, reported to reach 99.9%, and significantly improved stability during storage, directly addressing the degradation issues observed in products synthesized via older reductive amination techniques.
Mechanistic Insights into the Friedel-Crafts Alkylation Strategy
The success of this novel synthesis hinges on the precise execution of the Friedel-Crafts alkylation in the second step. Mechanistically, this involves the activation of the alkyl halide intermediate by the Lewis acid, anhydrous aluminum trichloride (AlCl3). The aluminum center coordinates with the bromine atom of the intermediate, facilitating the generation of a carbocation-like species or a tight ion pair at the terminal carbon of the propyl chain. This electrophilic species is then attacked by the electron-rich aromatic ring of benzotrifluoride. Although the trifluoromethyl group is electron-withdrawing, the reaction conditions—specifically the use of dichloromethane as a solvent and strict temperature control between 0°C and 10°C—ensure that the alkylation occurs selectively at the meta-position relative to the trifluoromethyl group, preserving the desired structural integrity of the Cinacalcet molecule.
Impurity control is inherently built into this mechanistic pathway. In previous methods involving direct reduction of imines, over-reduction or incomplete reaction often led to complex mixtures of amines and alcohols that were difficult to separate. In the new process, the use of the halo-alkyl intermediate prevents these specific reduction-related impurities. Furthermore, the quenching process involves pouring the reaction mixture into ice water, which hydrolyzes the aluminum complexes and precipitates the product as the hydrochloride salt upon addition of concentrated HCl. This workup procedure is not only operationally simple but also effective at removing inorganic salts and residual starting materials, contributing to the high purity profile observed in the final product. The ability to recrystallize or pulp the final solid in an ethyl acetate-n-hexane mixture further refines the crystal form and removes trace organic impurities.
How to Synthesize (R)-3-bromo-N-(1-(naphthalen-1-yl)ethyl)propan-1-amine Efficiently
The preparation of the critical intermediate is the foundation of this entire synthetic strategy. The process begins with the condensation of 3-bromopropanal and (R)-1-naphthylethylamine in anhydrous methanol. To ensure high conversion and minimize side reactions, the reaction is conducted in an ice-water bath to maintain a low temperature. Sodium cyanoborohydride is added in batches—specifically in a 2:2:1 molar ratio split—to control the rate of hydrogen evolution and heat generation. Following the reduction, the reaction is quenched with dilute hydrochloric acid, and the product is extracted into ethyl acetate. This careful control of stoichiometry and temperature is crucial for achieving the reported yields of nearly 80% for the intermediate, setting the stage for the high-yielding final alkylation step.
- Prepare the key intermediate (R)-3-bromo-N-(1-(naphthalen-1-yl)ethyl)propan-1-amine by reacting 3-bromopropanal with (R)-1-naphthylethylamine using sodium cyanoborohydride reduction in methanol.
- Perform Friedel-Crafts alkylation by reacting the intermediate with benzotrifluoride in the presence of anhydrous aluminum trichloride in dichloromethane at 0-10°C.
- Quench the reaction with ice water, separate the organic layer, and treat with concentrated hydrochloric acid to precipitate the final Cinacalcet Hydrochloride product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel synthetic route represents a significant opportunity to de-risk operations and optimize expenditure. The primary advantage lies in the drastic simplification of the raw material portfolio. By eliminating the need for specialized, high-cost catalysts like titanium tetraisopropoxide and hazardous reducing agents like borane-THF or lithium aluminum hydride, the process relies on commodity chemicals that are widely available and competitively priced. This shift not only lowers the direct cost of goods sold (COGS) but also mitigates the risk of supply disruptions caused by the limited availability of niche reagents. The stability of the intermediate also allows for potential stockpiling, providing a buffer against market volatility.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and pyrophoric reagents leads to substantial cost savings. Traditional methods often require specialized equipment for handling air-sensitive materials and extensive downstream processing to remove metal traces, which adds significant operational overhead. The new method utilizes standard glass-lined or stainless steel reactors and common solvents like dichloromethane and methanol. Furthermore, the high yield of the final step reduces the amount of starting material required per kilogram of API, directly improving the material efficiency and lowering the overall production cost without compromising quality.
- Enhanced Supply Chain Reliability: Sourcing 3-bromopropanal and benzotrifluoride is significantly more reliable than sourcing complex, unstable intermediates required by other routes. These starting materials are produced on a large scale for various industrial applications, ensuring a steady supply. Additionally, the robustness of the synthesis means that production timelines are more predictable. There is less likelihood of batch failures due to reagent instability or sensitive reaction conditions, which translates to more consistent delivery schedules for downstream pharmaceutical customers and a stronger, more resilient supply chain.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route is vastly superior. Avoiding the use of cyanoborohydride in the final steps (it is used in the intermediate step but in a controlled manner) and completely removing borane and lithium aluminum hydride reduces the generation of toxic waste streams. The waste profile is simpler, consisting mainly of aluminum salts and organic solvents that can be recovered and recycled. This alignment with green chemistry principles facilitates easier regulatory approval and reduces the costs associated with waste disposal and environmental compliance, making the process highly scalable for commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel Cinacalcet synthesis pathway. These insights are derived directly from the comparative data and experimental results presented in the patent literature, highlighting the practical benefits for industrial adoption.
Q: What are the primary safety advantages of this new synthesis route compared to conventional methods?
A: This novel route eliminates the need for highly toxic and unstable reagents such as borane-THF complexes, lithium aluminum hydride, and titanium tetraisopropoxide. By utilizing standard Friedel-Crafts conditions with aluminum trichloride, the process significantly reduces hazardous waste generation and improves operational safety for large-scale manufacturing.
Q: How does the new intermediate improve the overall yield and purity of Cinacalcet Hydrochloride?
A: The use of the specific halo-alkyl amine intermediate allows for a highly selective alkylation step. Patent data indicates that this method achieves yields up to 95.6% with purity levels reaching 99.9%, effectively minimizing the formation of reducing impurities common in direct reductive amination pathways.
Q: Is this synthetic pathway suitable for industrial scale-up?
A: Yes, the process is designed for industrial viability. It utilizes commercially available starting materials like 3-bromopropanal and avoids complex multi-step sequences or carcinogenic reagents like ethyl acrylate found in older Heck coupling routes, ensuring a robust and scalable supply chain.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cinacalcet Intermediate Supplier
The technological advancements detailed in patent CN109535006B underscore the dynamic nature of pharmaceutical intermediate manufacturing. At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting such innovative pathways to meet the evolving demands of the global market. Our facility is equipped with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can seamlessly transition this novel two-step synthesis from the laboratory to full-scale manufacturing. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Cinacalcet intermediate meets the highest international standards, providing our partners with the confidence they need to advance their own drug development pipelines.
We invite pharmaceutical companies and contract manufacturers to explore the potential of this cost-effective and safe production route. By leveraging our expertise in Friedel-Crafts chemistry and reductive amination, we can offer a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can drive efficiency and reliability in your supply chain.
